Human PSGL-1 (P-Selectin Glycoprotein Ligand-1; also CD162), is a 120 kDa mucin-type glycoprotein that plays a key role in leukocyte adhesion (1-3). It is synthesized as a 412 amino acid (aa) preproprecursor that contains a 17 aa signal sequence, a 24 aa propeptide, a 279 aa extracellular domain (ECD), a 21 aa transmembrane segment and a 71 aa cytoplasmic region (4, 5). Following cleavage of the pre- and prosegments, it is expressed as a 240 kDa disulfide-linked homodimer. The extreme N-terminus (aa 1-16 of the mature molecule) contains one threonine (#16) and three tyrosines (#5, 7, and 10) that are involved in ligand binding. The Thr residue allows for O-linked glycosylation in the form of a core-2 structure (GalNAc-Gal) linked in a beta 1,6 bond to a sialylated Lewis X motif (GlcNAc linked to both Fuc and Gal with a terminal sialic acid residue) (1, 2, 5, 6, 7). The three tyrosine residues allow for sulfation (8, 9). When binding to P-selectin, Tyr sulfation and glycosylation are essential. Tyr7 provides the most efficient sulfate moiety, while Fuc and sialic acid are essentially mandatory (7). When binding to
E‑Selectin, only carbohydrate is needed, while both carbohydrate and Tyr10 are used for L-selectin binding (6, 8). There are 16 decameric aa repeats in the ECD of the longform of PSGL-1. This form is referred to as the A allele, and represents 65-80% of the population. Alleles B and C show deletions of decameric repeats #2 (aa 132‑141) plus #9 and 10 (aa 222-241), respectively. Shorter forms may show weaker binding to P-selectin (9, 10). Soluble forms of PSGL-1 are also known. Neutrophil elastase will cleave somewhere within repeats #5-9, while cathepsin G cleaves after Tyr7 (11). The loss of Tyr5 and 7 should impact binding affinity.
PSGL‑1 is found on virtually all leukocytes and macrophages/DC’s (1). Although there is similarity in the organization of the ECD between species, there is little aa identity. Human PSGL-1 ECD is 51%, 52% and 43% aa identical to equine, canine and mouse ECD, respectively.
Human PSGL‑1/CD162 Antibody
R&D Systems | Catalog # AF3345

Key Product Details
Species Reactivity
Validated:
Cited:
Applications
Validated:
Cited:
Label
Antibody Source
Product Specifications
Immunogen
Thr44-Val295
Accession # NP_002997
Specificity
Clonality
Host
Isotype
Scientific Data Images for Human PSGL‑1/CD162 Antibody
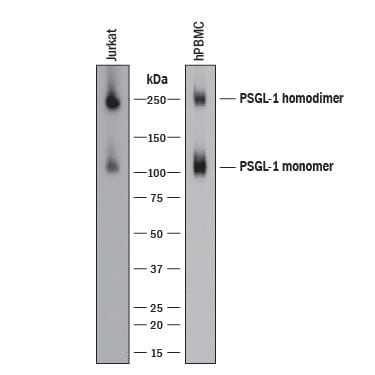
Detection of Human PSGL‑1/CD162 by Western Blot.
Western blot shows lysates of Jurkat human acute T cell leukemia cell line and human peripheral blood mononuclear cells (PBMCs). PVDF membrane was probed with 0.2 µg/mL of Sheep Anti-Human PSGL-1/CD162 Antigen Affinity-purified Polyclonal Antibody (Catalog # AF3345) followed by HRP-conjugated Anti-Sheep IgG Secondary Antibody (Catalog # HAF016). Specific bands were detected for PSGL-1/CD162 homodimer at approximately 250 kDa and PSGL-1/CD162 monomer at approximately 110 kDa (as indicated). This experiment was conducted under reducing conditions and using Immunoblot Buffer Group 8.PSGL‑1/CD162 in Human Tonsil.
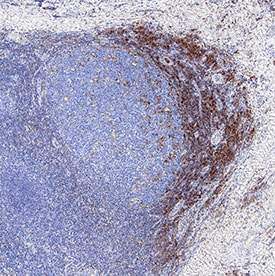
PSGL-1/CD162 was detected in immersion fixed paraffin-embedded sections of human tonsil using Sheep Anti-Human PSGL-1/CD162 Antigen Affinity-purified Polyclonal Antibody (Catalog # AF3345) at 3 µg/mL overnight at 4 °C. Tissue was stained using the Anti-Sheep HRP-DAB Cell & Tissue Staining Kit (brown; Catalog # CTS019) and counterstained with hematoxylin (blue). Specific staining was localized to lymphocytes. View our protocol for Chromogenic IHC Staining of Paraffin-embedded Tissue Sections.Human PSGL‑1/CD162 ELISA Standard Curve.
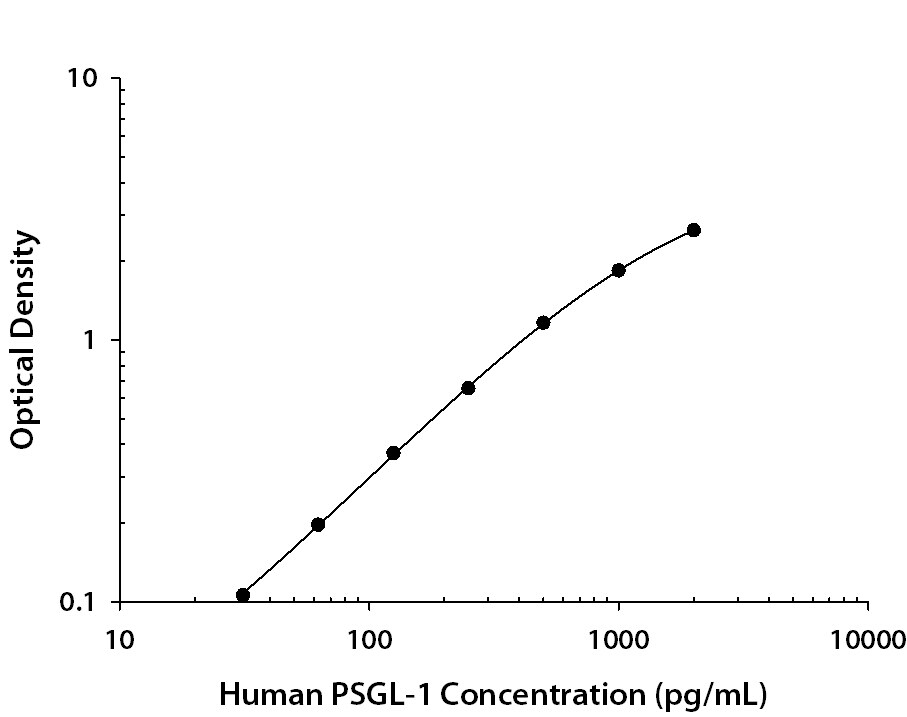
Recombinant Human PSGL-1/CD162 protein was serially diluted 2-fold and captured by Mouse Anti-Human PSGL-1/CD162 Monoclonal Antibody(Catalog # MAB9962) coated on a Clear Polystyrene Microplate (Catalog # DY990). Sheep Anti-Human PSGL-1/CD162 Antigen Affinity-purified Polyclonal Antibody (Catalog # AF3345) was biotinylated and incubated with the protein captured on the plate. Detection of the standard curve was achieved by incubating Streptavidin-HRP (Catalog # DY998) followed by Substrate Solution (Catalog # DY999) and stopping the enzymatic reaction with Stop Solution (Catalog # DY994).Applications for Human PSGL‑1/CD162 Antibody
ELISA
This antibody functions as an ELISA detection antibody when paired with Mouse Anti-Human PSGL‑1/CD162 Monoclonal Antibody(Catalog # MAB9962).
This product is intended for assay development on various assay platforms requiring antibody pairs. We recommend the Human PSGL-1/CD162 DuoSet ELISA (Catalog # DY3345-05) for convenient development of a sandwich ELISA.
Immunohistochemistry
Sample: Immersion fixed paraffin-embedded sections of human tonsils
Western Blot
Sample: Jurkat human acute T cell leukemia cell line and human peripheral blood mononuclear cells (PBMCs)
Formulation, Preparation, and Storage
Purification
Reconstitution
Sterile PBS to a final concentration of 0.2 mg/mL. For liquid material, refer to CoA for concentration.
Formulation
Shipping
Stability & Storage
- 12 months from date of receipt, -20 to -70 °C as supplied.
- 1 month, 2 to 8 °C under sterile conditions after reconstitution.
- 6 months, -20 to -70 °C under sterile conditions after reconstitution.
Calculators
Background: PSGL-1/CD162
References
- Yang, J. et al. (1999) Thromb. Haemost. 81:1.
- Cummings, R.D. (1999) Braz. J. Med. Biol. Res. 32:519.
- McEver, R.P. and R.D. Cummings (1997) J. Clin. Invest. 100:485.
- Sako, D. et al. (1993) Cell 75:1179.
- Veldman, G.M. et al. (1995) J. Biol. Chem. 270:16470.
- Bernimoulin, M.P. et al. (2003) J. Biol. Chem. 278:37.
- Leppanen, A. et al. (2000) J. Biol. Chem. 275:39569.
- Sako, D. et al. (1995) Cell 83:323.
- Afshar-Kharghan, V. et al. (2001) Blood 97:3306.
- Lozano, M.L. et al. (2001) Br. J. Haematol. 115:969.
- Gardiner, E.E. et al. (2001) Blood 98:1440.
Long Name
Alternate Names
Gene Symbol
UniProt
Additional PSGL-1/CD162 Products
Product Documents for Human PSGL‑1/CD162 Antibody
Certificate of Analysis
To download a Certificate of Analysis, please enter a lot or batch number in the search box below.
Note: Certificate of Analysis not available for kit components.
Product Specific Notices for Human PSGL‑1/CD162 Antibody
For research use only
Citations for Human PSGL‑1/CD162 Antibody
Customer Reviews for Human PSGL‑1/CD162 Antibody
There are currently no reviews for this product. Be the first to review Human PSGL‑1/CD162 Antibody and earn rewards!
Have you used Human PSGL‑1/CD162 Antibody?
Submit a review and receive an Amazon gift card!
$25/€18/£15/$25CAN/¥2500 Yen for a review with an image
$10/€7/£6/$10CAN/¥1110 Yen for a review without an image
Submit a review
Protocols
Find general support by application which include: protocols, troubleshooting, illustrated assays, videos and webinars.
- Antigen Retrieval Protocol (PIER)
- Antigen Retrieval for Frozen Sections Protocol
- Appropriate Fixation of IHC/ICC Samples
- Cellular Response to Hypoxia Protocols
- Chromogenic IHC Staining of Formalin-Fixed Paraffin-Embedded (FFPE) Tissue Protocol
- Chromogenic Immunohistochemistry Staining of Frozen Tissue
- ClariTSA™ Fluorophore Kits
- Detection & Visualization of Antibody Binding
- ELISA Sample Preparation & Collection Guide
- ELISA Troubleshooting Guide
- Fluorescent IHC Staining of Frozen Tissue Protocol
- Graphic Protocol for Heat-induced Epitope Retrieval
- Graphic Protocol for the Preparation and Fluorescent IHC Staining of Frozen Tissue Sections
- Graphic Protocol for the Preparation and Fluorescent IHC Staining of Paraffin-embedded Tissue Sections
- Graphic Protocol for the Preparation of Gelatin-coated Slides for Histological Tissue Sections
- How to Run an R&D Systems DuoSet ELISA
- How to Run an R&D Systems Quantikine ELISA
- How to Run an R&D Systems Quantikine™ QuicKit™ ELISA
- IHC Sample Preparation (Frozen sections vs Paraffin)
- Immunofluorescent IHC Staining of Formalin-Fixed Paraffin-Embedded (FFPE) Tissue Protocol
- Immunohistochemistry (IHC) and Immunocytochemistry (ICC) Protocols
- Immunohistochemistry Frozen Troubleshooting
- Immunohistochemistry Paraffin Troubleshooting
- Preparing Samples for IHC/ICC Experiments
- Preventing Non-Specific Staining (Non-Specific Binding)
- Primary Antibody Selection & Optimization
- Protocol for Heat-Induced Epitope Retrieval (HIER)
- Protocol for Making a 4% Formaldehyde Solution in PBS
- Protocol for VisUCyte™ HRP Polymer Detection Reagent
- Protocol for the Preparation & Fixation of Cells on Coverslips
- Protocol for the Preparation and Chromogenic IHC Staining of Frozen Tissue Sections
- Protocol for the Preparation and Chromogenic IHC Staining of Frozen Tissue Sections - Graphic
- Protocol for the Preparation and Chromogenic IHC Staining of Paraffin-embedded Tissue Sections
- Protocol for the Preparation and Chromogenic IHC Staining of Paraffin-embedded Tissue Sections - Graphic
- Protocol for the Preparation and Fluorescent IHC Staining of Frozen Tissue Sections
- Protocol for the Preparation and Fluorescent IHC Staining of Paraffin-embedded Tissue Sections
- Protocol for the Preparation of Gelatin-coated Slides for Histological Tissue Sections
- Quantikine HS ELISA Kit Assay Principle, Alkaline Phosphatase
- Quantikine HS ELISA Kit Principle, Streptavidin-HRP Polymer
- R&D Systems Quality Control Western Blot Protocol
- Sandwich ELISA (Colorimetric) – Biotin/Streptavidin Detection Protocol
- Sandwich ELISA (Colorimetric) – Direct Detection Protocol
- TUNEL and Active Caspase-3 Detection by IHC/ICC Protocol
- The Importance of IHC/ICC Controls
- Troubleshooting Guide: ELISA
- Troubleshooting Guide: Immunohistochemistry
- Troubleshooting Guide: Western Blot Figures
- Western Blot Conditions
- Western Blot Protocol
- Western Blot Protocol for Cell Lysates
- Western Blot Troubleshooting
- Western Blot Troubleshooting Guide
- View all Protocols, Troubleshooting, Illustrated assays and Webinars