SOST, also known as sclerostin, is a member of the cerberus/DAN family, a group of secreted glycoproteins characterized by a cysteine-knot motif. Cerberus/DAN family members are putative BMP antagonists, and include Dan, Cerberus, Gremlin, PRDC, and Caronte. While the overall sequence identity between members of the family is low, they have conserved spacing of six cysteine residues. Cerberus and dan have an additional cysteine residue used for dimerization; however, SOST does not and is secreted as a monomer. SOST was originally identified as an important regulator of bone homesotasis. Positional cloning studies identified that mutations in the SOST gene can cause sclerosteosis and van Buchem disease, bone dysplasia disorders characterized by progressive skeletal overgrowth. Significant levels of SOST expression are detected in bone, cartilage, kidney, and liver. SOST is expressed by osteoclasts in developing bones of mouse embryos, including both intramembranously forming skull bones and endochondrally forming long bones. SOST plays a physiological role as a negative regulator of bone formation by repressing BMP-induced osteogenesis. SOST has been shown to have unique ligand specificity, binding BMP-5, -6, and -7 with high affinity and BMP-2 and -4 with low affinity. This seems to be the first example of a BMP antagonist being localized to osteoclasts, cells derived from the hematopoietic lineage, that function to degrade bone matrix. Human and mouse SOST share 88% amino acid identity (1-3).
Human SOST/Sclerostin Antibody
R&D Systems | Catalog # AF1406

Key Product Details
Species Reactivity
Validated:
Human
Cited:
Human
Applications
Validated:
Western Blot
Cited:
Immunohistochemistry, Western Blot, Flow Cytometry, Immunocytochemistry
Label
Unconjugated
Antibody Source
Polyclonal Goat IgG
Loading...
Product Specifications
Immunogen
Mouse myeloma cell line NS0-derived recombinant human SOST
Gln24-Tyr213
Accession # Q9BQB4
Gln24-Tyr213
Accession # Q9BQB4
Specificity
Detects human SOST in direct ELISAs and Western blots. In direct ELISAs, approximately 15% cross‑reactivity with recombinant mouse SOST is observed.
Clonality
Polyclonal
Host
Goat
Isotype
IgG
Scientific Data Images for Human SOST/Sclerostin Antibody
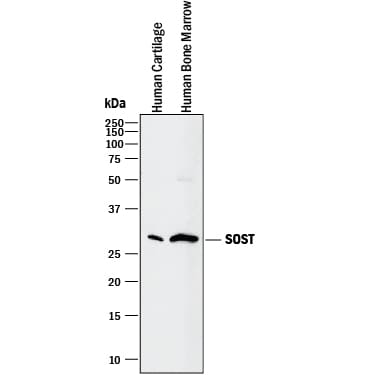
Detection of Human SOST/Sclerostin by Western Blot.
Western blot shows lysates of human cartilage tissue and human bone marrow. PVDF membrane was probed with 2 µg/mL of Goat Anti-Human SOST/Sclerostin Antigen Affinity-purified Polyclonal Antibody (Catalog # AF1406) followed by HRP-conjugated Anti-Goat IgG Secondary Antibody (Catalog # HAF017). A specific band was detected for SOST/Sclerostin at approximately 28 kDa (as indicated). This experiment was conducted under reducing conditions and using Immunoblot Buffer Group 1.Applications for Human SOST/Sclerostin Antibody
Application
Recommended Usage
Western Blot
2 µg/mL
Sample: Human cartilage tissue and human bone marrow
Sample: Human cartilage tissue and human bone marrow
Formulation, Preparation, and Storage
Purification
Antigen Affinity-purified
Reconstitution
Reconstitute at 0.2 mg/mL in sterile PBS. For liquid material, refer to CoA for concentration.
Loading...
Formulation
Lyophilized from a 0.2 μm filtered solution in PBS with Trehalose. *Small pack size (SP) is supplied either lyophilized or as a 0.2 µm filtered solution in PBS.
Shipping
Lyophilized product is shipped at ambient temperature. Liquid small pack size (-SP) is shipped with polar packs. Upon receipt, store immediately at the temperature recommended below.
Stability & Storage
Use a manual defrost freezer and avoid repeated freeze-thaw cycles.
- 12 months from date of receipt, -20 to -70 °C as supplied.
- 1 month, 2 to 8 °C under sterile conditions after reconstitution.
- 6 months, -20 to -70 °C under sterile conditions after reconstitution.
Calculators
Background: SOST/Sclerostin
References
- Kusu, N. et al. (2003) J. Biol. Chem. 278:24113.
- Balemans, W. et al. (2001) Hum. Mol. Genet. 10:537.
- Brunkow, M.E. et al. (2001) Am. J. Hum. Genet. 68:577.
Alternate Names
sclerostin, VBCHsclerosteosis
Gene Symbol
SOST
UniProt
Additional SOST/Sclerostin Products
Product Documents for Human SOST/Sclerostin Antibody
Certificate of Analysis
To download a Certificate of Analysis, please enter a lot or batch number in the search box below.
Note: Certificate of Analysis not available for kit components.
Product Specific Notices for Human SOST/Sclerostin Antibody
For research use only
Related Research Areas
Citations for Human SOST/Sclerostin Antibody
Customer Reviews for Human SOST/Sclerostin Antibody
There are currently no reviews for this product. Be the first to review Human SOST/Sclerostin Antibody and earn rewards!
Have you used Human SOST/Sclerostin Antibody?
Submit a review and receive an Amazon gift card!
$25/€18/£15/$25CAN/¥2500 Yen for a review with an image
$10/€7/£6/$10CAN/¥1110 Yen for a review without an image
Submit a review
Protocols
Find general support by application which include: protocols, troubleshooting, illustrated assays, videos and webinars.
- Cellular Response to Hypoxia Protocols
- R&D Systems Quality Control Western Blot Protocol
- Troubleshooting Guide: Western Blot Figures
- Western Blot Conditions
- Western Blot Protocol
- Western Blot Protocol for Cell Lysates
- Western Blot Troubleshooting
- Western Blot Troubleshooting Guide
- View all Protocols, Troubleshooting, Illustrated assays and Webinars
Loading...
