Supervillin ("Largest" villin [family member]; also Archvillin and p250) is a 240-250 kDa member of the villin/gelsolin family of proteins. It is expressed in select cell types, including platelets, neutrophils, renal epithelium and striated muscle cells. Supervillin is found intracellularly in both the nuclear and cytoplasmic compartments. Its principal function is to serve as a scaffold for molecules that interact to oversee changes in the actin cytoskeleton at the cell membrane. It is able to both crosslink F-actin and initiate actin bundle formation. It also binds nonmuscle myosin and negatively regulates focal adhesion complexes. Human supervillin is 2214 amino acids (aa) in length. It contains an NLS (aa 927-931), five gelsolin-like repeats that interact with F-actin (aa 1441-2122), and a C-terminal villin headpiece domain (aa 2179-2214). The 250 kDa form of supervillin (archvillin) is striated muscle-specific. There is also one general nonmuscle form of supervillin (p205) that is 200-210 kDa in size and contains two blocks of deletions between aa 276-669 and 749-781. In ferret, a third 220-230 kDa intermediate isoform has been reported that is unique to smooth muscle. Over aa 1801-2214, human supervillin shares 96% aa sequence identity with mouse supervillin.
Human Supervillin Antibody
R&D Systems | Catalog # AF7338

Key Product Details
Species Reactivity
Human
Applications
Western Blot
Label
Unconjugated
Antibody Source
Polyclonal Goat IgG
Loading...
Product Specifications
Immunogen
E. coli-derived recombinant human Supervillin
His1801-Thr2214
Accession # O95425
His1801-Thr2214
Accession # O95425
Specificity
Detects human Supervillin in direct ELISAs and Western blots.
Clonality
Polyclonal
Host
Goat
Isotype
IgG
Scientific Data Images for Human Supervillin Antibody
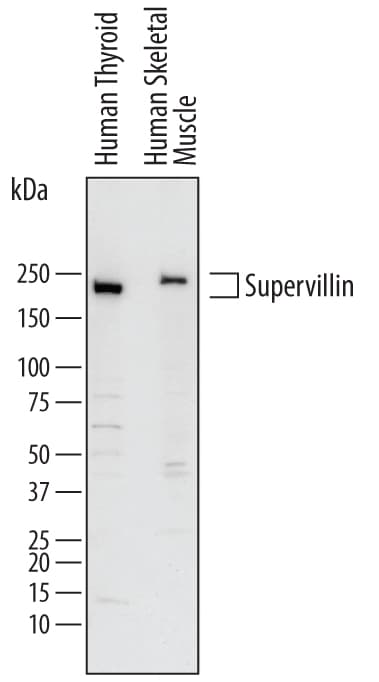
Detection of Human Supervillin by Western Blot.
Western blot shows lysates of human thyroid tissue and human skeletal muscle tissue. PVDF membrane was probed with 1 µg/mL of Goat Anti-Human Supervillin Antigen Affinity-purified Polyclonal Antibody (Catalog # AF7338) followed by HRP-conjugated Anti-Goat IgG Secondary Antibody (Catalog # HAF017). Specific bands were detected for Supervillin at approximately 240-250 kDa (as indicated). This experiment was conducted under reducing conditions and using Immunoblot Buffer Group 1.Applications for Human Supervillin Antibody
Application
Recommended Usage
Western Blot
1 µg/mL
Sample: Human thyroid tissue and human skeletal muscle tissue
Sample: Human thyroid tissue and human skeletal muscle tissue
Formulation, Preparation, and Storage
Purification
Antigen Affinity-purified
Reconstitution
Sterile PBS to a final concentration of 0.2 mg/mL. For liquid material, refer to CoA for concentration.
Loading...
Formulation
Lyophilized from a 0.2 μm filtered solution in PBS with Trehalose. *Small pack size (SP) is supplied either lyophilized or as a 0.2 µm filtered solution in PBS.
Shipping
Lyophilized product is shipped at ambient temperature. Liquid small pack size (-SP) is shipped with polar packs. Upon receipt, store immediately at the temperature recommended below.
Stability & Storage
Use a manual defrost freezer and avoid repeated freeze-thaw cycles.
- 12 months from date of receipt, -20 to -70 °C as supplied.
- 1 month, 2 to 8 °C under sterile conditions after reconstitution.
- 6 months, -20 to -70 °C under sterile conditions after reconstitution.
Calculators
Background: Supervillin
Alternate Names
Archvillin, p205/p250, SVIL
Gene Symbol
SVIL
UniProt
Additional Supervillin Products
Product Documents for Human Supervillin Antibody
Certificate of Analysis
To download a Certificate of Analysis, please enter a lot or batch number in the search box below.
Note: Certificate of Analysis not available for kit components.
Product Specific Notices for Human Supervillin Antibody
For research use only
Citations for Human Supervillin Antibody
Customer Reviews for Human Supervillin Antibody
There are currently no reviews for this product. Be the first to review Human Supervillin Antibody and earn rewards!
Have you used Human Supervillin Antibody?
Submit a review and receive an Amazon gift card!
$25/€18/£15/$25CAN/¥2500 Yen for a review with an image
$10/€7/£6/$10CAN/¥1110 Yen for a review without an image
Submit a review
Protocols
Find general support by application which include: protocols, troubleshooting, illustrated assays, videos and webinars.
- Cellular Response to Hypoxia Protocols
- R&D Systems Quality Control Western Blot Protocol
- Troubleshooting Guide: Western Blot Figures
- Western Blot Conditions
- Western Blot Protocol
- Western Blot Protocol for Cell Lysates
- Western Blot Troubleshooting
- Western Blot Troubleshooting Guide
- View all Protocols, Troubleshooting, Illustrated assays and Webinars
Loading...
