Mouse CD68/SR‑D1 Antibody
R&D Systems | Catalog # MAB10114

Key Product Details
Species Reactivity
Applications
Label
Antibody Source
Product Specifications
Immunogen
Asp21-Pro282
Accession # P31996
Specificity
Clonality
Host
Isotype
Scientific Data Images for Mouse CD68/SR‑D1 Antibody
CD68/SR‑D1 in RAW 264.7 and NIH-3T3 Mouse Cell Lines.
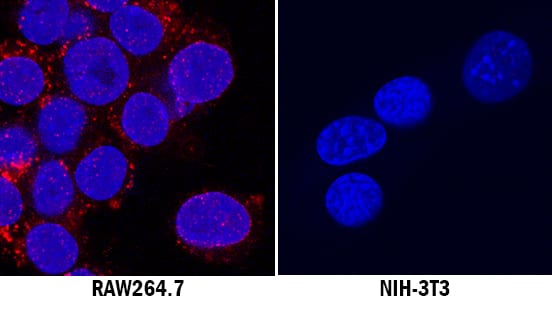
CD68/SR-D1 was detected in immersion fixed RAW 264.7 mouse monocyte/macrophage cell line (positive stain) and NIH-3T3 mouse embryonic fibroblast cell line (negative stain) using Rabbit Anti-Mouse CD68/SR-D1 Monoclonal Antibody (Catalog # MAB10114) at 3 µg/mL for 3 hours at room temperature. Cells were stained using the NorthernLights™ 557-conjugated Anti-Rabbit IgG Secondary Antibody (red; Catalog # NL004) and counterstained with DAPI (blue). Specific staining was localized to cytoplasm. View our protocol for Fluorescent ICC Staining of Non-adherent Cells.Detection of CD68 in Raw264.7 Mouse Cell line by Flow Cytometry.
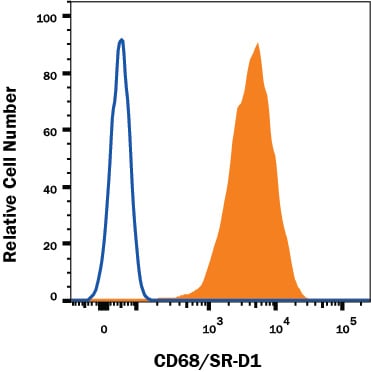
Raw264.7 mouse cell line was stained with Rabbit Anti-Mouse CD68 Monoclonal Antibody (Catalog # MAB10114, filled histogram) or Normal Rabbit IgG Control (Catalog # MAB1050, open histogram) followed by Phycoerythrin-conjugated Anti-Rabbit IgG Secondary Antibody (Catalog # F0110). To facilitate intracellular staining, cells were fixed with Flow Cytometry Fixation Buffer (1x) (Catalog # FC004) and permeabilized with ice-cold methanol. View our protocol for Staining Intracellular Molecules.Detection of Mouse CD68/SR-D1 by Immunohistochemistry
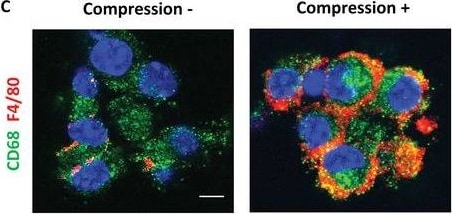
Mechanical compression stimulated piezo‐type mechanosensitive ion channel component 1 (PIEZO1) signaling for CD68+F4/80+ macrophages differentiation in whole periosteal cells (WPCs) and bone marrow‐derived macrophages (BMDMs). A–I) Primary WPCs or BMDMs were harvested from WT mice and subjected to 10 or 0 kPa of mechanical compression. A) mRNA expression of Cd68 and adhesion G protein‐coupled receptor E1 (Adgre1) by RT‐qPCR in WPCs after compression. B) mRNA expression of Adgre1 in BMDMs after compression. C) Immunofluorescent staining of CD68+ (green) and F4/80+ (red) cells in BMDMs after compression. Blue indicates DAPI staining of nuclei. Scale bar, 5 µm. D,E) mRNA expression of colony‐stimulating factor 1 (Csf1) in WPCs and BMDMs after compression, respectively. F,G) mRNA expression of Piezo1 in WPCs and BMDMs after compression, respectively. H,I) BMDMs were pretreated with the PIEZO1 inhibitor GsMTx4 or vehicle for 24 h before compression. H) mRNA expression of Csf1 and Adgre1 in BMDMs after compression. I) Immunofluorescent staining of CD68+ (green) and F4/80+ (red) cells after compression. Blue indicates DAPI staining of nuclei. Scale bar, 5 µm. Data are presented as mean ± SEM. n = 3 per group. A,B,D–G) * p < 0.05 compared with 0 kPa. Statistical significance was determined by unpaired, two‐tailed Student's t‐test. H) *p < 0.05 compared with vehicle 10‐kPa compression group; †p < 0.05. Statistical significance was determined by two‐way ANOVA with Bonferroni post hoc test. Image collected and cropped by CiteAb from the following open publication (https://pubmed.ncbi.nlm.nih.gov/34854257), licensed under a CC-BY license. Not internally tested by R&D Systems.Applications for Mouse CD68/SR‑D1 Antibody
CyTOF-ready
Immunocytochemistry
Sample: Immersion fixed RAW264.7 mouse monocyte/macrophage cell line
Intracellular Staining by Flow Cytometry
Sample: RAW264.7 mouse monocyte/macrophage cell line fixed with Flow Cytometry Fixation Buffer (Catalog # FC004) and permeabilized with ice-cold methanol.
Flow Cytometry Panel Builder
Bio-Techne Knows Flow Cytometry
Save time and reduce costly mistakes by quickly finding compatible reagents using the Panel Builder Tool.
Advanced Features
- Spectra Viewer - Custom analysis of spectra from multiple fluorochromes
- Spillover Popups - Visualize the spectra of individual fluorochromes
- Antigen Density Selector - Match fluorochrome brightness with antigen density
Formulation, Preparation, and Storage
Purification
Reconstitution
Reconstitute at 0.5 mg/mL in sterile PBS. For liquid material, refer to CoA for concentration.
Formulation
Shipping
Stability & Storage
- 12 months from date of receipt, -20 to -70 °C as supplied.
- 1 month, 2 to 8 °C under sterile conditions after reconstitution.
- 6 months, -20 to -70 °C under sterile conditions after reconstitution.
Calculators
Background: CD68/SR-D1
Alternate Names
Gene Symbol
UniProt
Additional CD68/SR-D1 Products
Product Documents for Mouse CD68/SR‑D1 Antibody
Certificate of Analysis
To download a Certificate of Analysis, please enter a lot or batch number in the search box below.
Note: Certificate of Analysis not available for kit components.
Product Specific Notices for Mouse CD68/SR‑D1 Antibody
For research use only
Citations for Mouse CD68/SR‑D1 Antibody
Customer Reviews for Mouse CD68/SR‑D1 Antibody
There are currently no reviews for this product. Be the first to review Mouse CD68/SR‑D1 Antibody and earn rewards!
Have you used Mouse CD68/SR‑D1 Antibody?
Submit a review and receive an Amazon gift card!
$25/€18/£15/$25CAN/¥2500 Yen for a review with an image
$10/€7/£6/$10CAN/¥1110 Yen for a review without an image
Submit a review
Protocols
Find general support by application which include: protocols, troubleshooting, illustrated assays, videos and webinars.
- 7-Amino Actinomycin D (7-AAD) Cell Viability Flow Cytometry Protocol
- Appropriate Fixation of IHC/ICC Samples
- Cellular Response to Hypoxia Protocols
- ClariTSA™ Fluorophore Kits
- Detection & Visualization of Antibody Binding
- Extracellular Membrane Flow Cytometry Protocol
- Flow Cytometry Protocol for Cell Surface Markers
- Flow Cytometry Protocol for Staining Membrane Associated Proteins
- Flow Cytometry Staining Protocols
- Flow Cytometry Troubleshooting Guide
- ICC Cell Smear Protocol for Suspension Cells
- ICC Immunocytochemistry Protocol Videos
- ICC for Adherent Cells
- Immunocytochemistry (ICC) Protocol
- Immunocytochemistry Troubleshooting
- Immunofluorescence of Organoids Embedded in Cultrex Basement Membrane Extract
- Immunohistochemistry (IHC) and Immunocytochemistry (ICC) Protocols
- Intracellular Flow Cytometry Protocol Using Alcohol (Methanol)
- Intracellular Flow Cytometry Protocol Using Detergents
- Intracellular Nuclear Staining Flow Cytometry Protocol Using Detergents
- Intracellular Staining Flow Cytometry Protocol Using Alcohol Permeabilization
- Intracellular Staining Flow Cytometry Protocol Using Detergents to Permeabilize Cells
- Preparing Samples for IHC/ICC Experiments
- Preventing Non-Specific Staining (Non-Specific Binding)
- Primary Antibody Selection & Optimization
- Propidium Iodide Cell Viability Flow Cytometry Protocol
- Protocol for Liperfluo
- Protocol for VisUCyte™ HRP Polymer Detection Reagent
- Protocol for the Characterization of Human Th22 Cells
- Protocol for the Characterization of Human Th9 Cells
- Protocol for the Fluorescent ICC Staining of Cell Smears - Graphic
- Protocol for the Fluorescent ICC Staining of Cultured Cells on Coverslips - Graphic
- Protocol for the Preparation and Fluorescent ICC Staining of Cells on Coverslips
- Protocol for the Preparation and Fluorescent ICC Staining of Non-adherent Cells
- Protocol for the Preparation and Fluorescent ICC Staining of Stem Cells on Coverslips
- Protocol for the Preparation of a Cell Smear for Non-adherent Cell ICC - Graphic
- Protocol: Annexin V and PI Staining by Flow Cytometry
- Protocol: Annexin V and PI Staining for Apoptosis by Flow Cytometry
- TUNEL and Active Caspase-3 Detection by IHC/ICC Protocol
- The Importance of IHC/ICC Controls
- Troubleshooting Guide: Fluorokine Flow Cytometry Kits
- View all Protocols, Troubleshooting, Illustrated assays and Webinars
Associated Pathways