Mouse IFN‑ gamma Antibody
R&D Systems | Catalog # MAB485

Key Product Details
Species Reactivity
Validated:
Cited:
Applications
Validated:
Cited:
Label
Antibody Source
Product Specifications
Immunogen
Specificity
Clonality
Host
Isotype
Endotoxin Level
Scientific Data Images for Mouse IFN‑ gamma Antibody
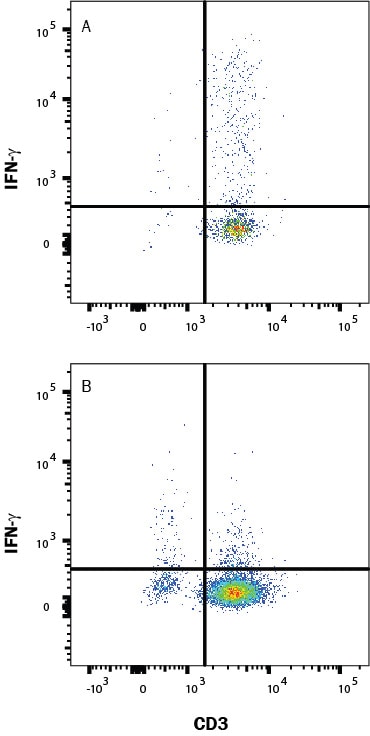
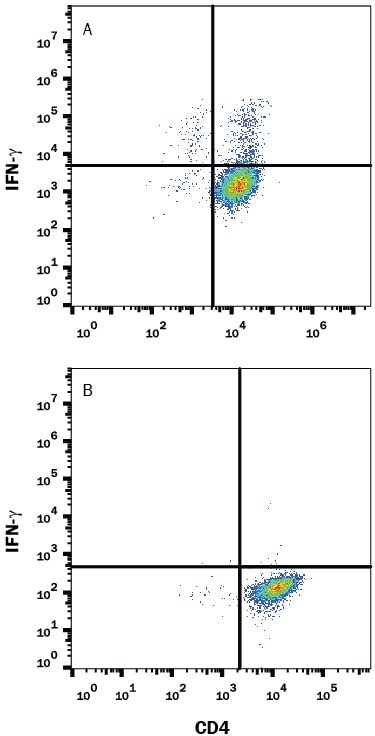
Detection of IFN‑ gamma in Mouse Splenoctyes by Flow Cytometry.
Mouse splenocytes either (A) stimulated to induce Th1 cells or (B) unstimulated were stained with Rat Anti-Mouse IFN-gamma Monoclonal Antibody (Catalog # MAB485) followed by Phycoerythrin-conjugated Anti-Rat IgG Secondary Antibody (F0105B) and Rat Anti-Mouse CD4 APC-conjugated Monoclonal Antibody (FAB554A). Quadrant markers were set based on control antibody staining (MAB006). To facilitate intracellular staining, cells were fixed with Flow Cytometry Fixation Buffer (FC004) and permeabilized with Flow Cytometry Permeabilization/Wash Buffer I (FC005). View our protocol for Staining Intracellular Molecules.Detection of IFN‑ gamma in Mouse Splenoctyes by Flow Cytometry.
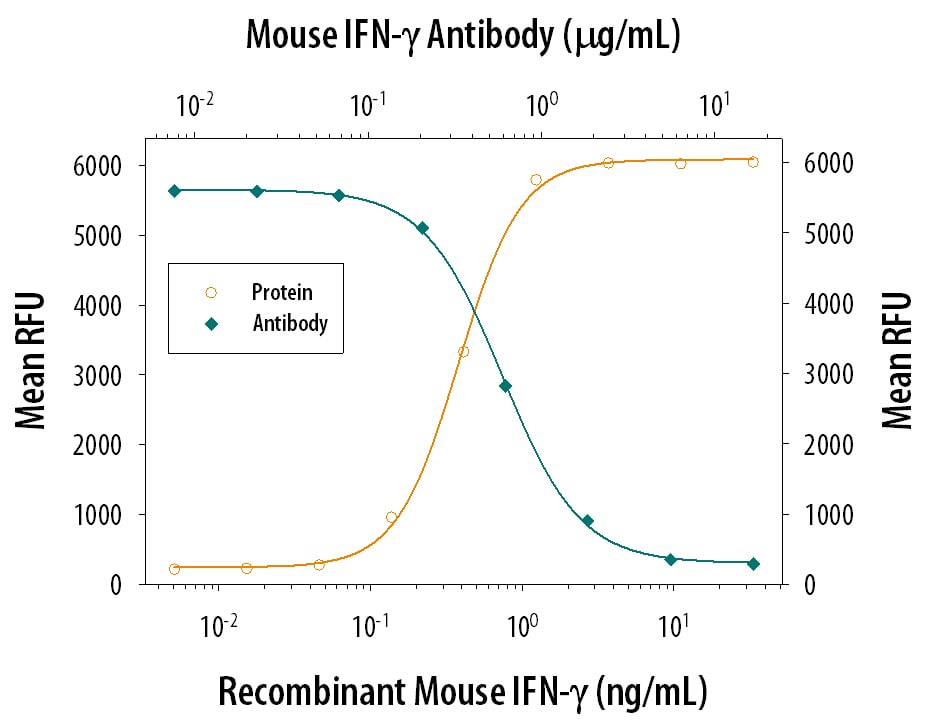
Mouse splenocytes either (A) stimulated for 4 hours with Cell Activation Cocktail 500x (5476) or (B) unstimulated were stained with Rat Anti-Mouse IFN-gamma Monoclonal Antibody (Catalog # MAB485) followed by Phycoerythrin-conjugated Anti-Rat IgG Secondary Antibody (F0105B) and Rat Anti-Mouse CD4 APC-conjugated Monoclonal Antibody (FAB554A). Quadrant markers were set based on control antibody staining (MAB006). To facilitate intracellular staining, cells were fixed with Flow Cytometry Fixation Buffer (FC004) and permeabilized with Flow Cytometry Permeabilization/Wash Buffer I (FC005). Staining was performed using our Staining Intracellular Molecules protocol.IFN‑ gamma Inhibition of EMCV-induced Cytopathy and Neutralization by Mouse IFN‑ gamma Antibody.
Recombinant Mouse IFN-gamma 485-MI) reduces the Encephalomyocarditis Virus (EMCV)-induced cytopathy in the L-929 mouse fibroblast cell line in a dose-dependent manner (orange line), as measured by crystal violet staining. Inhibition of EMCV activity elicited by Recombinant Mouse IFN-gamma (2.5 ng/mL) is neutralized (green line) by increasing concentrations of Rat Anti-Mouse IFN-gamma Monoclonal Antibody (Catalog # MAB485). The ND50 is typically 0.075-0.3 µg/mL.Detection of IFN‑ gamma in Mouse Spleen.
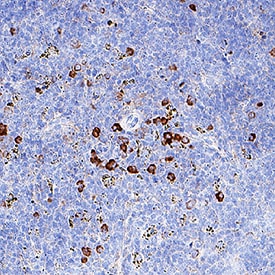
IFN‑ gamma was detected in immersion fixed paraffin-embedded sections of Mouse Spleen using Rat Anti-Mouse IFN‑ gamma Monoclonal Antibody (Catalog # MAB485) at 10 µg/mL for 1 hour at room temperature followed by incubation with the Anti-Rat IgG VisUCyte™ HRP Polymer Antibody (Catalog # VC005). Before incubation with the primary antibody, tissue was subjected to heat-induced epitope retrieval using VisUCyte Antigen Retrieval Reagent-Basic (Catalog # VCTS021). Tissue was stained using DAB (brown) and counterstained with hematoxylin (blue). Specific staining was localized to cytoplasm in lymphocytes. View our protocol for IHC Staining with VisUCyte HRP Polymer Detection Reagents.Detection of Mouse IFN-gamma by Flow Cytometry
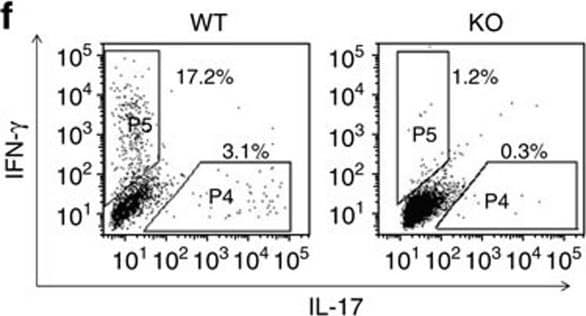
EAE induction in KO mice.(a) Means±s.e.m. of EAE clinical scores of KO and WT mice. *P<0.05 (two-tailed Student's t-test). (b) EAE incidence in KO and WT mice. *P<0.05 (chi-square test). (c) Means±s.e.m. of body weight of KO and WT mice during EAE induction. Body weight of mice on day 10 post-immunization was considered as 100%. *P<0.05 (two-tailed Student's t-test). (d) Means±s.e.m. of cellularity in draining LN and of cells infiltrating the CNS of mice 14 days after MOG immunization. Mouse numbers (n) and P values (paired two-tailed Student's t-test) are indicated. (e,f) Cytokine-producing cells among CD4 cells from draining LN (e) and CNS (f) on days 13–18 after MOG immunization. Left panels: representative dot plots; right panel: bar graphs (means±s.e.m.) summarizing all the results, with mouse numbers and P values (two-tailed Student's t-test) indicated. (g) HE (left column) or Luxol Fast Blue (right column) staining of spinal cords 30 days after MOG immunization. Asterisks indicate cell infiltration. Arrows point to demyelination. (h) Means±s.e.m. of mononuclear cell infiltration scores, demyelination scores and total pathological scores, which is the sum of the first two scores. Mouse numbers (n) and P values (two-tailed Student's t-test) are indicated. (i) Treg cells in naive KO mice on day 17 during EAE induction. Left panel: representative dot plots; right panel: means±s.e.m. of data from three experiments. NS: not significant (two-tailed Student's t-test). Image collected and cropped by CiteAb from the following publication (https://pubmed.ncbi.nlm.nih.gov/28169274), licensed under a CC-BY license. Not internally tested by R&D Systems.Detection of Mouse IFN-gamma by Flow Cytometry
EAE induction in KO mice.(a) Means±s.e.m. of EAE clinical scores of KO and WT mice. *P<0.05 (two-tailed Student's t-test). (b) EAE incidence in KO and WT mice. *P<0.05 (chi-square test). (c) Means±s.e.m. of body weight of KO and WT mice during EAE induction. Body weight of mice on day 10 post-immunization was considered as 100%. *P<0.05 (two-tailed Student's t-test). (d) Means±s.e.m. of cellularity in draining LN and of cells infiltrating the CNS of mice 14 days after MOG immunization. Mouse numbers (n) and P values (paired two-tailed Student's t-test) are indicated. (e,f) Cytokine-producing cells among CD4 cells from draining LN (e) and CNS (f) on days 13–18 after MOG immunization. Left panels: representative dot plots; right panel: bar graphs (means±s.e.m.) summarizing all the results, with mouse numbers and P values (two-tailed Student's t-test) indicated. (g) HE (left column) or Luxol Fast Blue (right column) staining of spinal cords 30 days after MOG immunization. Asterisks indicate cell infiltration. Arrows point to demyelination. (h) Means±s.e.m. of mononuclear cell infiltration scores, demyelination scores and total pathological scores, which is the sum of the first two scores. Mouse numbers (n) and P values (two-tailed Student's t-test) are indicated. (i) Treg cells in naive KO mice on day 17 during EAE induction. Left panel: representative dot plots; right panel: means±s.e.m. of data from three experiments. NS: not significant (two-tailed Student's t-test). Image collected and cropped by CiteAb from the following publication (https://pubmed.ncbi.nlm.nih.gov/28169274), licensed under a CC-BY license. Not internally tested by R&D Systems.Detection of Mouse IFN-gamma by Flow Cytometry
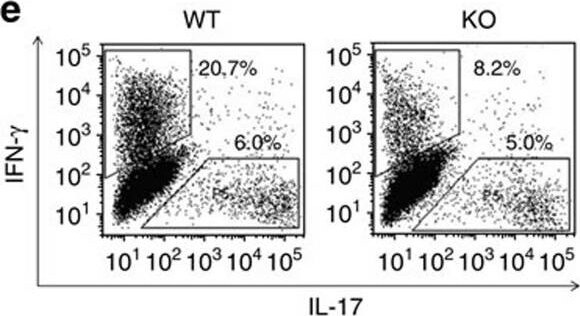
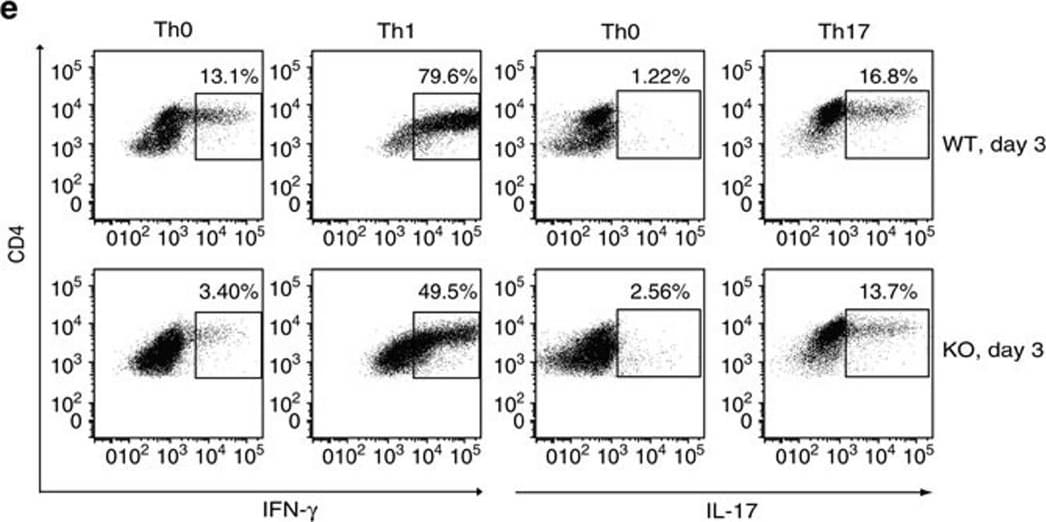
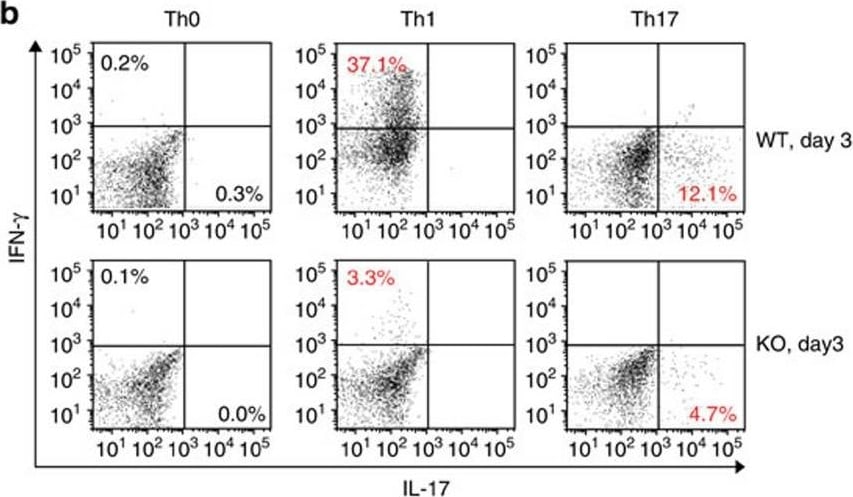
Proliferation and differentiation of naive KO CD4 cells into Th1 and Th17 cells.(a) Proliferation of WT and KO naive spleen CD4 cells under Th1 and Th17 conditions was assessed based on CFSE content according to flow cytometry. Experiments were conducted three times, and representative histograms are shown. Grey peaks represent the CFSE content of CD4 cells at day 0. (b) These cells' differentiation into Th1 and Th17 cells was also determined by flow cytometry according to intracellular IFN-gamma and IL-17 positivity (gated on total CD4+). Representative dot plots are shown in the left panel. Means±s.e.m. of data from three experiments are presented as bar graphs in the right panel. Mouse numbers (n) per group are indicated. P values are reported in the bar graphs (two-tailed Student's t-test). (c,d) T-bet and ROR gamma t expression in CD4 cells cultured under Th1 and Th17 conditions or in IFN gamma + or IL-17+ cells was determined by flow cytometry. Experiments were conducted three times. Representative histograms are shown. (e) Th1 and Th17 differentiation of naive spleen CD4 cells (CD45.2 single-positive) derived from WT and KO donors in chimeric mice was analysed by flow cytometry based on their intracellular IFN-gamma and IL-17 expression. Representative dot plots are shown in the left panel. Means±s.e.m. of data from three experiments are presented as bar graphs in the right panel. Mouse numbers (n) per group are indicated. p values are reported in the bar graphs (two-tailed Student's t-test). Image collected and cropped by CiteAb from the following publication (https://pubmed.ncbi.nlm.nih.gov/28169274), licensed under a CC-BY license. Not internally tested by R&D Systems.Detection of Mouse IFN-gamma by Flow Cytometry
Proliferation and differentiation of naive KO CD4 cells into Th1 and Th17 cells.(a) Proliferation of WT and KO naive spleen CD4 cells under Th1 and Th17 conditions was assessed based on CFSE content according to flow cytometry. Experiments were conducted three times, and representative histograms are shown. Grey peaks represent the CFSE content of CD4 cells at day 0. (b) These cells' differentiation into Th1 and Th17 cells was also determined by flow cytometry according to intracellular IFN-gamma and IL-17 positivity (gated on total CD4+). Representative dot plots are shown in the left panel. Means±s.e.m. of data from three experiments are presented as bar graphs in the right panel. Mouse numbers (n) per group are indicated. P values are reported in the bar graphs (two-tailed Student's t-test). (c,d) T-bet and ROR gamma t expression in CD4 cells cultured under Th1 and Th17 conditions or in IFN gamma + or IL-17+ cells was determined by flow cytometry. Experiments were conducted three times. Representative histograms are shown. (e) Th1 and Th17 differentiation of naive spleen CD4 cells (CD45.2 single-positive) derived from WT and KO donors in chimeric mice was analysed by flow cytometry based on their intracellular IFN-gamma and IL-17 expression. Representative dot plots are shown in the left panel. Means±s.e.m. of data from three experiments are presented as bar graphs in the right panel. Mouse numbers (n) per group are indicated. p values are reported in the bar graphs (two-tailed Student's t-test). Image collected and cropped by CiteAb from the following publication (https://pubmed.ncbi.nlm.nih.gov/28169274), licensed under a CC-BY license. Not internally tested by R&D Systems.Detection of Mouse IFN-gamma by Flow Cytometry
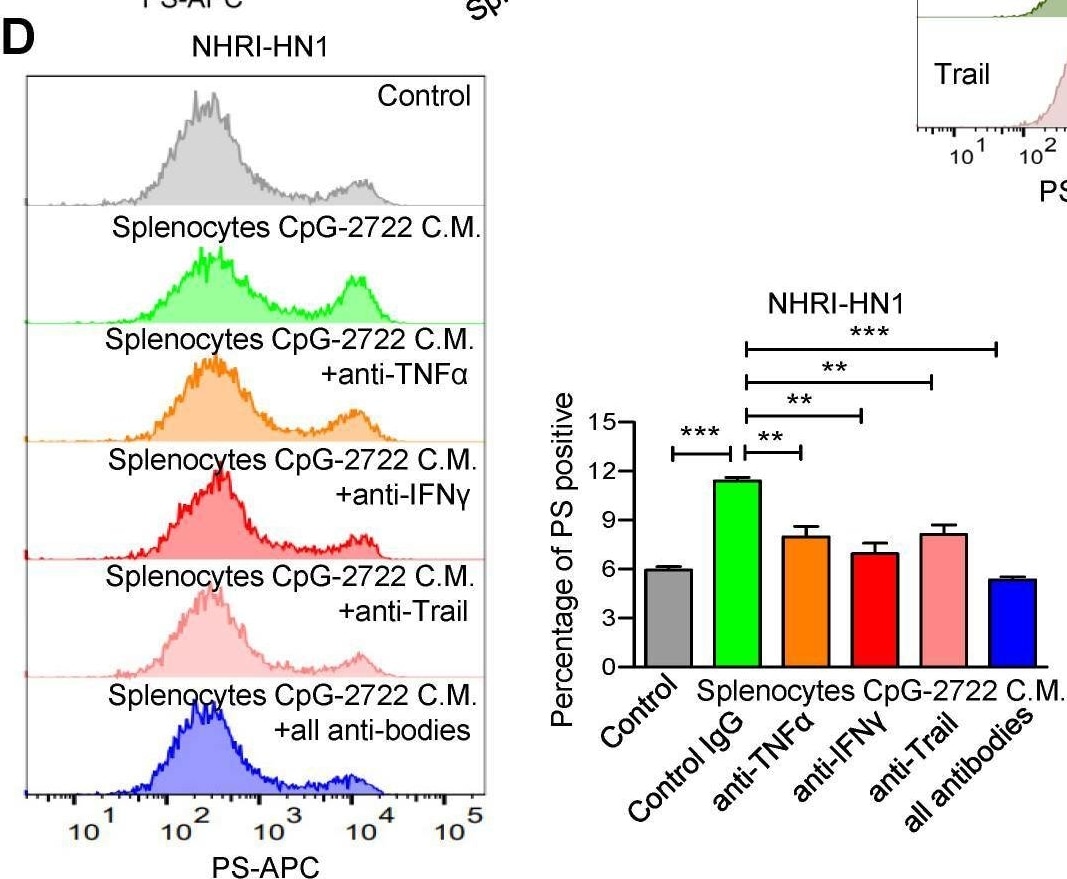
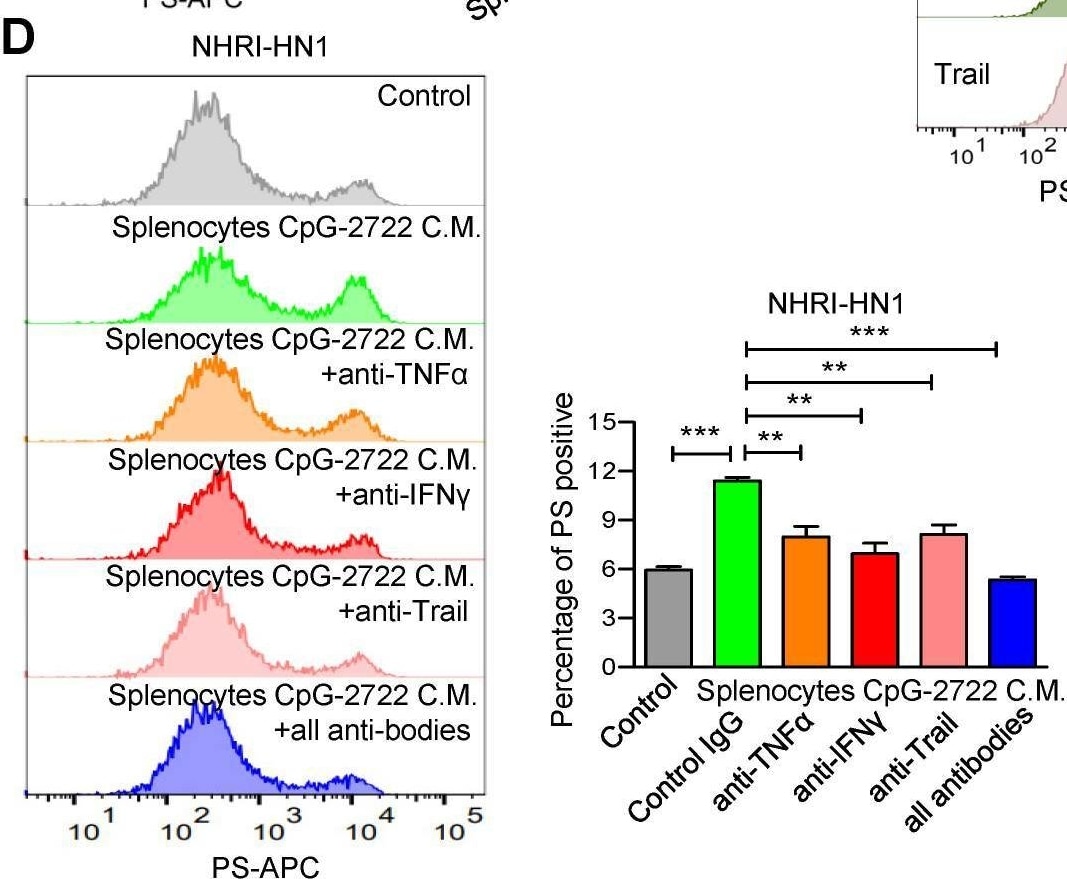
CpG-2722 treatment increases PS exposure on cancer through induction of cytokines from immune. (A) Mouse splenocytes were stimulated with/without 5 μg/ml of CpG-2722 for 48 h, & collected the condition medium (C.M.). NHRI-HN1 were treatment with/without 5 μg/ml of CpG-2722 or with/without medium containing 25% C.M. for 24 h as illustrated in the top panel. Phosphatidylserine (PS) exposure on NHRI-HN1 were stained by annexin V conjugated APC antibody, & acquired on a FACS Canto II & analyzed using FlowJo software. Bottom left panel: a representative set of histograms. Bottom right panel: Data represent the mean ± SEM (n = 3). (B) Cytokine levels of TNF-alpha, IFN-gamma, & Trail in tumors from the experiment in Figure 5 were measured by ELISA. Data represent the mean ± SEM (n = 5). (C) NHRI-HN1 were treated with 100 ng/ml of TNF-alpha, IFN-gamma, or Trail for 24 h. Levels of PS exposure were stained by annexin V conjugated APC antibody & acquired on a FACS Canto II. Left panel: a representative set of histograms. Right panel: Data represent the mean ± SEM (n = 3). (D) Mouse splenocytes were stimulated with/without 5 μg/ml of CpG-2722 for 48 h, & the C.M. was collected. NHRI-HN1 were incubated with medium containing 25% of the C.M. in the presence of 1 μg/ml of neutralizing antibody to TNF-alpha, IFN-gamma, Trail or their combination as indicated for 24 h. Phosphatidylserine (PS) exposure on NHRI-HN1 were stained by annexin V conjugated APC antibody, & acquired on a FACS Canto II & analyzed using FlowJo software. Bottom left panel: a representative set of histograms. Bottom right panel: Data represent the mean ± SEM (n = 3). *, **, & *** represent statistically significant differences p < 0.05, p < 0.01, & p < 0.001, respectively, compared with the control or as indicated. Image collected & cropped by CiteAb from the following open publication (https://pubmed.ncbi.nlm.nih.gov/37324949), licensed under a CC-BY license. Not internally tested by R&D Systems.Detection of Mouse IFN-gamma by Flow Cytometry
CpG-2722 treatment increases PS exposure on cancer through induction of cytokines from immune. (A) Mouse splenocytes were stimulated with/without 5 μg/ml of CpG-2722 for 48 h, & collected the condition medium (C.M.). NHRI-HN1 were treatment with/without 5 μg/ml of CpG-2722 or with/without medium containing 25% C.M. for 24 h as illustrated in the top panel. Phosphatidylserine (PS) exposure on NHRI-HN1 were stained by annexin V conjugated APC antibody, & acquired on a FACS Canto II & analyzed using FlowJo software. Bottom left panel: a representative set of histograms. Bottom right panel: Data represent the mean ± SEM (n = 3). (B) Cytokine levels of TNF-alpha, IFN-gamma, & Trail in tumors from the experiment in Figure 5 were measured by ELISA. Data represent the mean ± SEM (n = 5). (C) NHRI-HN1 were treated with 100 ng/ml of TNF-alpha, IFN-gamma, or Trail for 24 h. Levels of PS exposure were stained by annexin V conjugated APC antibody & acquired on a FACS Canto II. Left panel: a representative set of histograms. Right panel: Data represent the mean ± SEM (n = 3). (D) Mouse splenocytes were stimulated with/without 5 μg/ml of CpG-2722 for 48 h, & the C.M. was collected. NHRI-HN1 were incubated with medium containing 25% of the C.M. in the presence of 1 μg/ml of neutralizing antibody to TNF-alpha, IFN-gamma, Trail or their combination as indicated for 24 h. Phosphatidylserine (PS) exposure on NHRI-HN1 were stained by annexin V conjugated APC antibody, & acquired on a FACS Canto II & analyzed using FlowJo software. Bottom left panel: a representative set of histograms. Bottom right panel: Data represent the mean ± SEM (n = 3). *, **, & *** represent statistically significant differences p < 0.05, p < 0.01, & p < 0.001, respectively, compared with the control or as indicated. Image collected & cropped by CiteAb from the following open publication (https://pubmed.ncbi.nlm.nih.gov/37324949), licensed under a CC-BY license. Not internally tested by R&D Systems.Applications for Mouse IFN‑ gamma Antibody
CyTOF-ready
Immunohistochemistry
Sample: Immersion fixed paraffin-embedded sections of Mouse Spleen
Intracellular Staining by Flow Cytometry
Sample: Mouse splenocytes stimulated to induce Th1 cells or for 4 hours with Cell Activation Cocktail 500x (5476) were fixed with Flow Cytometry Fixation Buffer (Catalog # FC004) and permeabilized with Flow Cytometry Permeabilization/Wash Buffer I (Catalog # FC005)
Western Blot
Sample: Recombinant Mouse IFN-gamma (Catalog # 485-MI)
Neutralization
Flow Cytometry Panel Builder
Bio-Techne Knows Flow Cytometry
Save time and reduce costly mistakes by quickly finding compatible reagents using the Panel Builder Tool.
Advanced Features
- Spectra Viewer - Custom analysis of spectra from multiple fluorochromes
- Spillover Popups - Visualize the spectra of individual fluorochromes
- Antigen Density Selector - Match fluorochrome brightness with antigen density
Formulation, Preparation, and Storage
Purification
Reconstitution
Reconstitute at 0.5 mg/mL in sterile PBS. For liquid material, refer to CoA for concentration.
Formulation
Shipping
Stability & Storage
- 12 months from date of receipt, -20 to -70 °C as supplied.
- 1 month, 2 to 8 °C under sterile conditions after reconstitution.
- 6 months, -20 to -70 °C under sterile conditions after reconstitution.
Calculators
Background: IFN-gamma
Long Name
Alternate Names
Entrez Gene IDs
Gene Symbol
Additional IFN-gamma Products
Product Documents for Mouse IFN‑ gamma Antibody
Certificate of Analysis
To download a Certificate of Analysis, please enter a lot or batch number in the search box below.
Note: Certificate of Analysis not available for kit components.
Product Specific Notices for Mouse IFN‑ gamma Antibody
For research use only
Related Research Areas
Citations for Mouse IFN‑ gamma Antibody
Customer Reviews for Mouse IFN‑ gamma Antibody
There are currently no reviews for this product. Be the first to review Mouse IFN‑ gamma Antibody and earn rewards!
Have you used Mouse IFN‑ gamma Antibody?
Submit a review and receive an Amazon gift card!
$25/€18/£15/$25CAN/¥2500 Yen for a review with an image
$10/€7/£6/$10CAN/¥1110 Yen for a review without an image
Submit a review
Protocols
Find general support by application which include: protocols, troubleshooting, illustrated assays, videos and webinars.
- 7-Amino Actinomycin D (7-AAD) Cell Viability Flow Cytometry Protocol
- Antigen Retrieval Protocol (PIER)
- Antigen Retrieval for Frozen Sections Protocol
- Appropriate Fixation of IHC/ICC Samples
- Cellular Response to Hypoxia Protocols
- Chromogenic IHC Staining of Formalin-Fixed Paraffin-Embedded (FFPE) Tissue Protocol
- Chromogenic Immunohistochemistry Staining of Frozen Tissue
- ClariTSA™ Fluorophore Kits
- Detection & Visualization of Antibody Binding
- Extracellular Membrane Flow Cytometry Protocol
- Flow Cytometry Protocol for Cell Surface Markers
- Flow Cytometry Protocol for Staining Membrane Associated Proteins
- Flow Cytometry Staining Protocols
- Flow Cytometry Troubleshooting Guide
- Fluorescent IHC Staining of Frozen Tissue Protocol
- Graphic Protocol for Heat-induced Epitope Retrieval
- Graphic Protocol for the Preparation and Fluorescent IHC Staining of Frozen Tissue Sections
- Graphic Protocol for the Preparation and Fluorescent IHC Staining of Paraffin-embedded Tissue Sections
- Graphic Protocol for the Preparation of Gelatin-coated Slides for Histological Tissue Sections
- IHC Sample Preparation (Frozen sections vs Paraffin)
- Immunofluorescent IHC Staining of Formalin-Fixed Paraffin-Embedded (FFPE) Tissue Protocol
- Immunohistochemistry (IHC) and Immunocytochemistry (ICC) Protocols
- Immunohistochemistry Frozen Troubleshooting
- Immunohistochemistry Paraffin Troubleshooting
- Intracellular Flow Cytometry Protocol Using Alcohol (Methanol)
- Intracellular Flow Cytometry Protocol Using Detergents
- Intracellular Nuclear Staining Flow Cytometry Protocol Using Detergents
- Intracellular Staining Flow Cytometry Protocol Using Alcohol Permeabilization
- Intracellular Staining Flow Cytometry Protocol Using Detergents to Permeabilize Cells
- Preparing Samples for IHC/ICC Experiments
- Preventing Non-Specific Staining (Non-Specific Binding)
- Primary Antibody Selection & Optimization
- Propidium Iodide Cell Viability Flow Cytometry Protocol
- Protocol for Heat-Induced Epitope Retrieval (HIER)
- Protocol for Liperfluo
- Protocol for Making a 4% Formaldehyde Solution in PBS
- Protocol for VisUCyte™ HRP Polymer Detection Reagent
- Protocol for the Characterization of Human Th22 Cells
- Protocol for the Characterization of Human Th9 Cells
- Protocol for the Preparation & Fixation of Cells on Coverslips
- Protocol for the Preparation and Chromogenic IHC Staining of Frozen Tissue Sections
- Protocol for the Preparation and Chromogenic IHC Staining of Frozen Tissue Sections - Graphic
- Protocol for the Preparation and Chromogenic IHC Staining of Paraffin-embedded Tissue Sections
- Protocol for the Preparation and Chromogenic IHC Staining of Paraffin-embedded Tissue Sections - Graphic
- Protocol for the Preparation and Fluorescent IHC Staining of Frozen Tissue Sections
- Protocol for the Preparation and Fluorescent IHC Staining of Paraffin-embedded Tissue Sections
- Protocol for the Preparation of Gelatin-coated Slides for Histological Tissue Sections
- Protocol: Annexin V and PI Staining by Flow Cytometry
- Protocol: Annexin V and PI Staining for Apoptosis by Flow Cytometry
- R&D Systems Quality Control Western Blot Protocol
- TUNEL and Active Caspase-3 Detection by IHC/ICC Protocol
- The Importance of IHC/ICC Controls
- Troubleshooting Guide: Fluorokine Flow Cytometry Kits
- Troubleshooting Guide: Immunohistochemistry
- Troubleshooting Guide: Western Blot Figures
- Western Blot Conditions
- Western Blot Protocol
- Western Blot Protocol for Cell Lysates
- Western Blot Troubleshooting
- Western Blot Troubleshooting Guide
- View all Protocols, Troubleshooting, Illustrated assays and Webinars
Associated Pathways
 Hematopoietic Stem Cell Differentiation Pathways & Lineage-specific Markers
Hematopoietic Stem Cell Differentiation Pathways & Lineage-specific Markers
 IL-15 Signaling Pathways and their Primary Biological Effects in Different Immune Cell Types
IL-15 Signaling Pathways and their Primary Biological Effects in Different Immune Cell Types
 IL-21 Signaling Pathways and their Primary Biological Effects in Different Immune Cell Types
IL-21 Signaling Pathways and their Primary Biological Effects in Different Immune Cell Types
 Innate Lymphoid Cell Differentiation Pathways
Innate Lymphoid Cell Differentiation Pathways