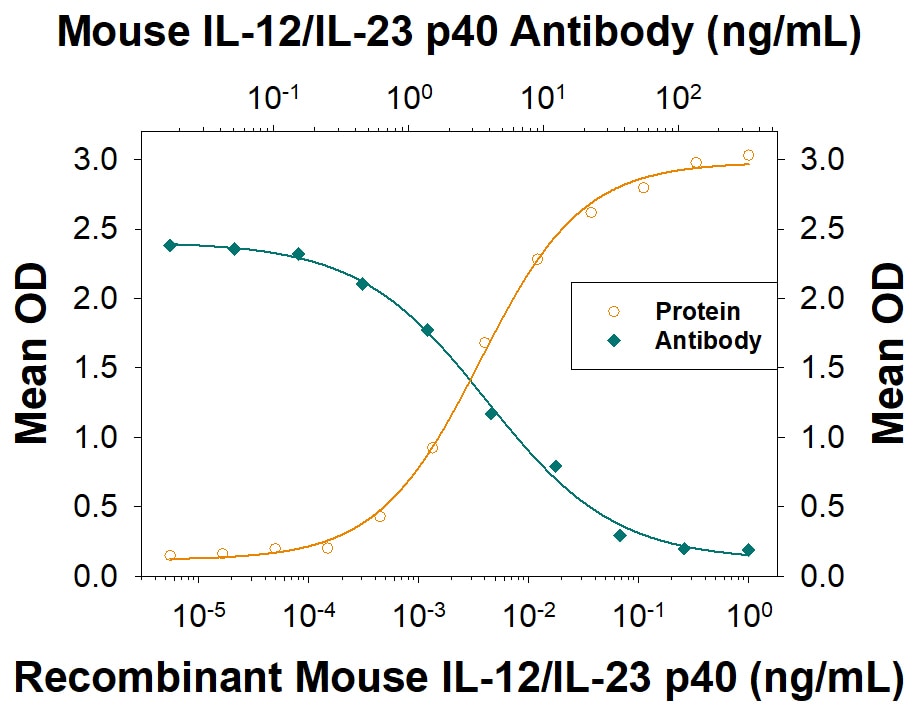
Interleukin 12, also known as natural killer cell stimulatory factor (NKSF) or cytotoxic lymphocyte maturation factor (CLMF), is a pleiotropic cytokine originally identified in the medium of activated human B lymphoblastoid cell lines. IL-12 has multiple effects on T cells and NK cells and is a key mediator in the development of Th1 cells.
IL-12 is a heterodimeric cytokine containing two disulfide-linked subunits, p35 and p40. Human and mouse IL-12 share 70% and 60% amino acid sequence identity in their p40 and p35 subunits, respectively. Although mouse IL-12 is active on human or mouse IL-12 responsive cells, human IL-12 is not active on mouse cells.
The disulfide-linked mouse p40 homodimer can bind to IL-12 receptors and is an antagonist of IL-12 activities in vitro. The mouse p40 monomer is at least ten times less active than the homodimer as an IL-12 antagonist. At the present time, the existence and the physiological role of mouse p40 homodimer in vivo remains to be determined.