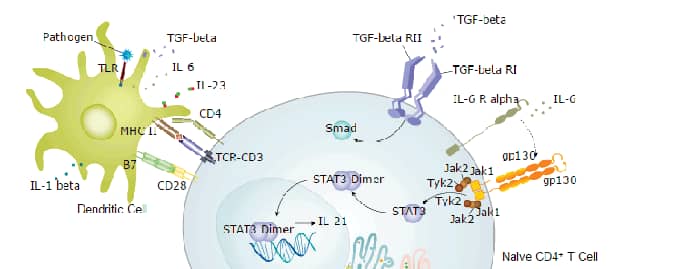
IL-6 induces signaling through a cell surface heterodimeric receptor complex composed of a ligand binding subunit (IL-6 R alpha) and a signal transducing subunit (gp130). IL-6 binds to IL-6 Ra, triggering IL-6 Ra association with gp130 and gp130 dimerization. gp130 is also a component of the receptors for CLC, CNTF, CT-1, IL-11, IL-27, LIF, and OSM. Soluble forms of IL-6 Ra are generated by both alternative splicing and proteolytic cleavage. In a mechanism known as trans-signaling, complexes of soluble IL-6 and IL-6 Ra elicit responses from gp130-expressing cells that lack cell surface IL-6 Ra. Trans-signaling enables a wider range of cell types to respond to IL-6, as the expression of gp130 is ubiquitous, while that of IL-6 Ra is predominantly restricted to hepatocytes, monocytes, and resting lymphocytes. Soluble splice forms of gp130 block trans-signaling from IL-6/IL-6 Ra but not from other cytokines that use gp130 as a co-receptor.
IL-6, along with TNF-a and IL-1, drives the acute inflammatory response. IL-6 is almost solely responsible for fever and the acute phase response in the liver, and it is important in the transition from acute inflammation to either acquired immunity or chronic inflammatory disease. When dysregulated, it contributes to chronic inflammation in conditions such as obesity, insulin resistance, inflammatory bowel disease, arthritis, and sepsis. IL-6 modulates bone resorption and is a major effector of inflammatory joint destruction in rheumatoid arthritis through its promotion of Th17 cell development and activity. It contributes to atherosclerotic plaque development and destabilization as well as the development of inflammation-associated carcinogenesis.