Mouse OCILRP2/CLEC2i Antibody
R&D Systems | Catalog # AF3370

Key Product Details
Validated by
Biological Validation
Species Reactivity
Mouse
Applications
Western Blot
Label
Unconjugated
Antibody Source
Polyclonal Goat IgG
Loading...
Product Specifications
Immunogen
E. coli-derived recombinant mouse OCILRP2/CLEC2i
Thr77-Val217
Accession # Q9WVF9
Thr77-Val217
Accession # Q9WVF9
Specificity
Detects mouse OCILRP2/CLEC2i in direct ELISAs and Western blots. In Western blots, approximately 25% cross-reactivity with recombinant mouse OCIL is observed.
Clonality
Polyclonal
Host
Goat
Isotype
IgG
Scientific Data Images for Mouse OCILRP2/CLEC2i Antibody
Detection of Mouse OCILRP2/CLEC2i by Immunocytochemistry/Immunofluorescence
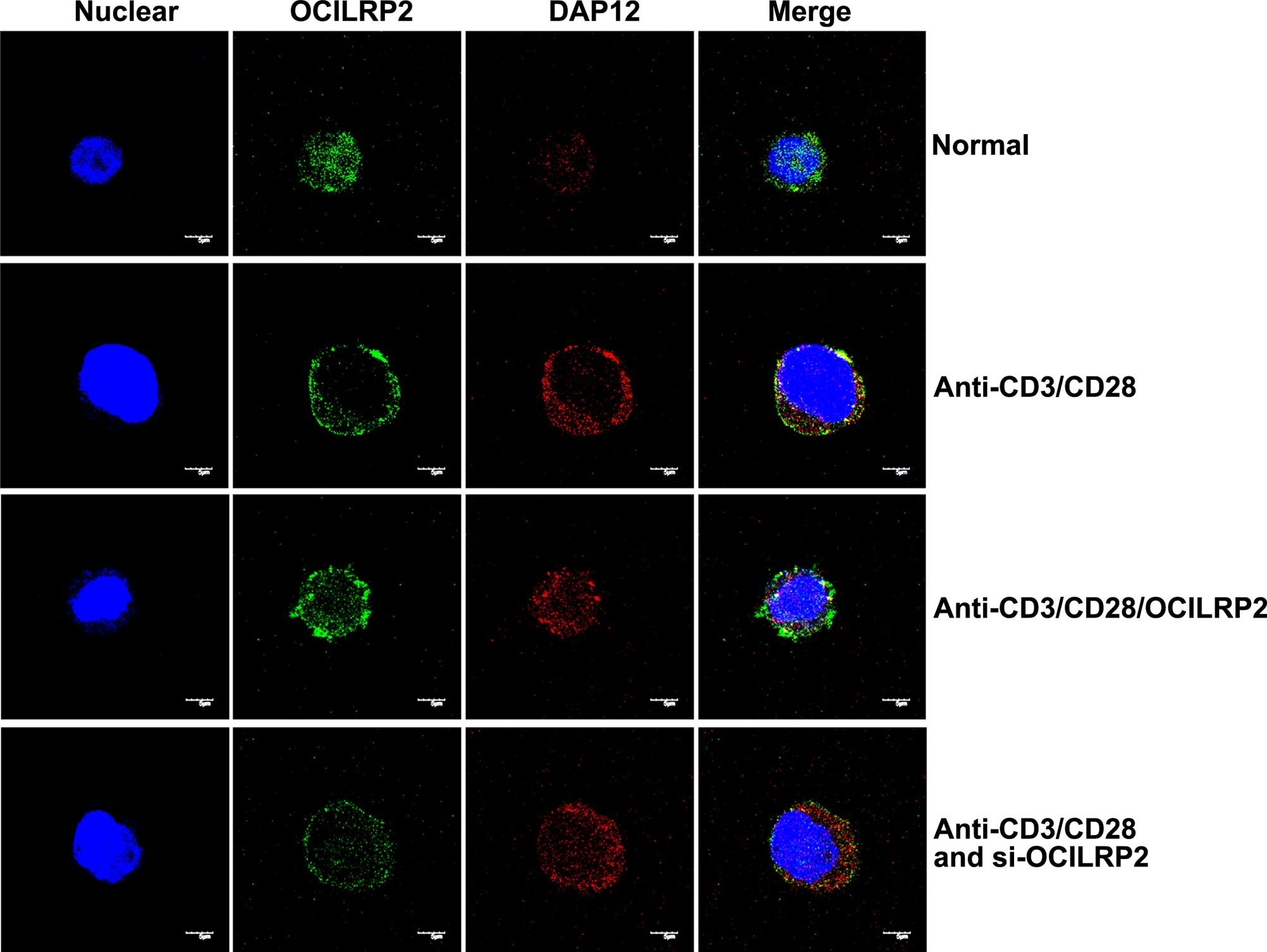
Membrane co-localization of OCILRP2 and DAP12 under anti-CD3/CD28 mAb treatment in EL4 T cells.EL4 T cells pre-transfected with pEGFP-siOCILRP2 or pre-coated with the OCILRP2 Ab were cultured overnight in the presence or absence of an anti-CD3/CD28 mAb and then stained for OCILRP2 (green) and nuclear stained with DAPI (blue) and DAP12 (red) to study OCILRP2 and DAP12 protein expression and localization. Unstimulated EL4 T cells exhibited OCILRP2 protein expression in the cytoplasm (upper panels). In contrast, CD3/CD28-activated EL4 T cells showed intracellular and membrane OCILRP2. OCILRP2/DAP12 co-localization appears in yellow. Each picture is representative of the vast majority of the observed cells on the slides. Image collected and cropped by CiteAb from the following publication (https://dx.plos.org/10.1371/journal.pone.0113218), licensed under a CC-BY license. Not internally tested by R&D Systems.Detection of Mouse OCILRP2/CLEC2i by Western Blot
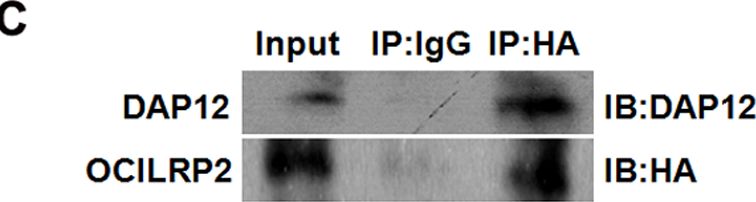
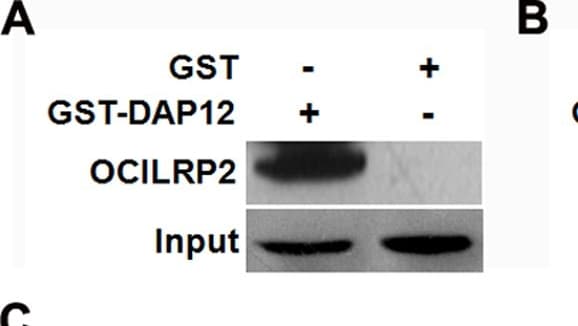
Interaction of OCILRP2 and DAP12 in GST pull-down and co-immunoprecipitation assays.The GST pull-down assay was carried out using purified beads that contained GST, GST-DAP12, or GST-OCILRP2. Precipitated OCILRP2 or DAP12 was detected by western blotting using an anti-OCILRP2 or anti-DAP12 antibody, respectively (a, b). 293T cells were grown in 6-cm dishes and transfected with the pCDNA3-HA-OCILRP2 and pDsRed-C1-DAP12 plasmids, respectively. OCILRP2 and DAP12 were detected by western blotting using an anti-HA antibody or an anti-DAP12 antibody (c). Schematic diagram of OCILRP2 predicted by SMART software. The green column represents the transmembrane region (amino acids 57–80) (d). The GST pull-down assay was carried out using purified beads that contained GST, full-length GST-OCILRP2, GST-OCILRP2i (aa 1–57), or GST-OCILRP2e (aa 80–221). Precipitated DAP12 was detected by western blotting using an anti-DAP12 antibody (e). Image collected and cropped by CiteAb from the following publication (https://dx.plos.org/10.1371/journal.pone.0113218), licensed under a CC-BY license. Not internally tested by R&D Systems.Detection of Mouse OCILRP2/CLEC2i by Western Blot
Interaction of OCILRP2 and DAP12 in GST pull-down and co-immunoprecipitation assays.The GST pull-down assay was carried out using purified beads that contained GST, GST-DAP12, or GST-OCILRP2. Precipitated OCILRP2 or DAP12 was detected by western blotting using an anti-OCILRP2 or anti-DAP12 antibody, respectively (a, b). 293T cells were grown in 6-cm dishes and transfected with the pCDNA3-HA-OCILRP2 and pDsRed-C1-DAP12 plasmids, respectively. OCILRP2 and DAP12 were detected by western blotting using an anti-HA antibody or an anti-DAP12 antibody (c). Schematic diagram of OCILRP2 predicted by SMART software. The green column represents the transmembrane region (amino acids 57–80) (d). The GST pull-down assay was carried out using purified beads that contained GST, full-length GST-OCILRP2, GST-OCILRP2i (aa 1–57), or GST-OCILRP2e (aa 80–221). Precipitated DAP12 was detected by western blotting using an anti-DAP12 antibody (e). Image collected and cropped by CiteAb from the following publication (https://dx.plos.org/10.1371/journal.pone.0113218), licensed under a CC-BY license. Not internally tested by R&D Systems.Detection of Mouse OCILRP2/CLEC2i by Flow Cytometry
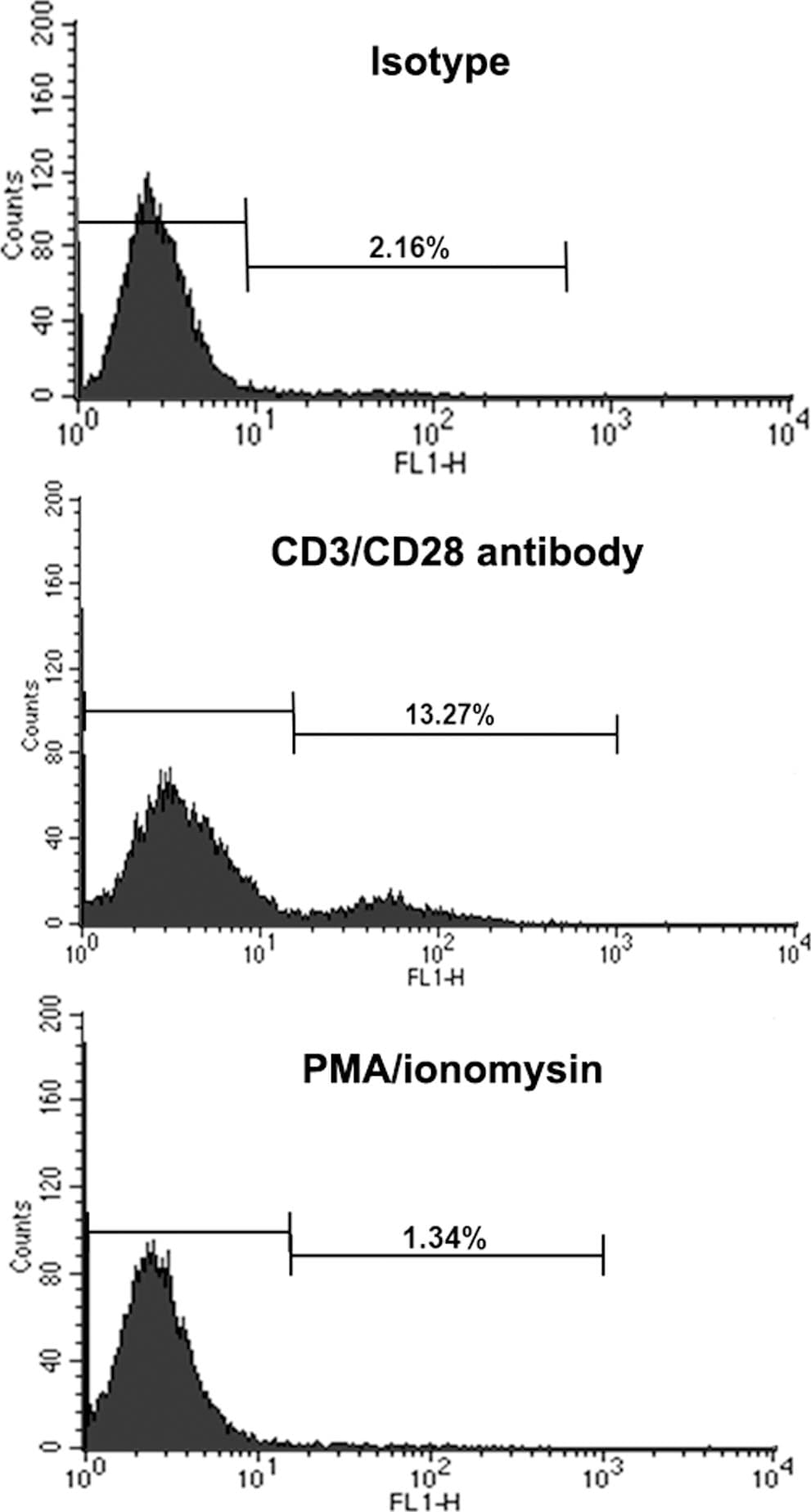
Membrane re-localization of OCILRP2 via treatment with an anti-CD3/CD28 mAb.EL4 cells were incubated with plate-bound CD3/CD28 antibodies, isotype-matched mIgGs, or PMA/ionomycin for 48 h. The cells (1×106 cells) were then incubated with an anti-OCILRP2 antagonist antibody for 30 min on ice, followed by staining with an FITC-labeled second antibody. The cells were analyzed with Cellquest software using a FACS Calibur flow cytometer. Image collected and cropped by CiteAb from the following publication (https://dx.plos.org/10.1371/journal.pone.0113218), licensed under a CC-BY license. Not internally tested by R&D Systems.Detection of OCILRP2/CLEC2i by Western Blot
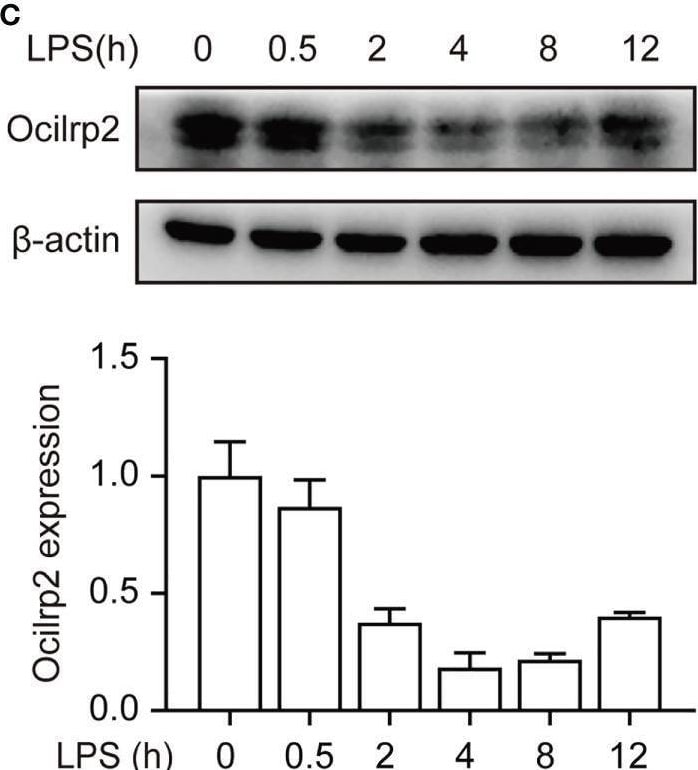
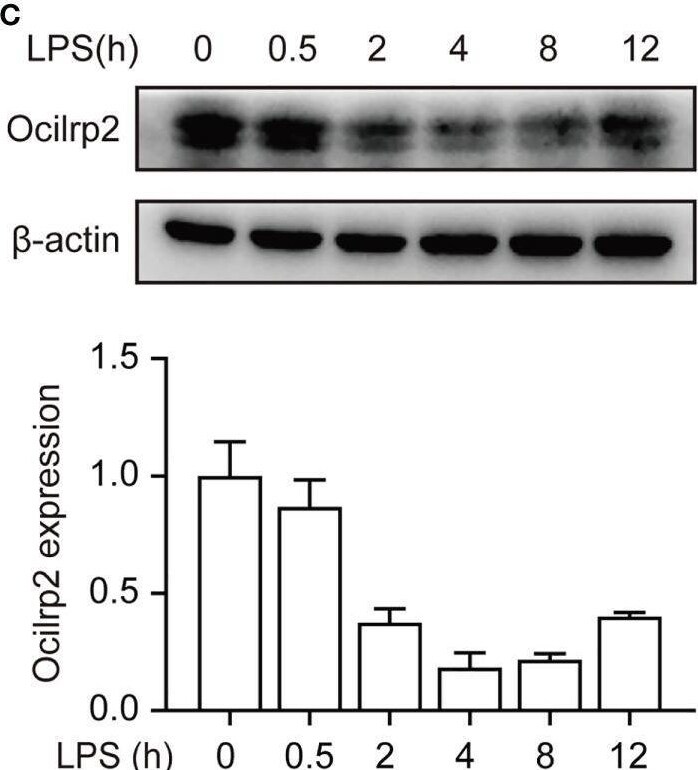
Downregulation of Ocilrp2 transcription in macrophages upon innate stimuli. (A) qRT-PCR analysis of Ocilrp2 mRNA levels in mouse peritoneal macrophages following LPS, Poly (I:C) or CpG stimulation or VSV, and SeV infection for 4 h. Data were normalized to the expression of GAPDH. (B) qRT-PCR analysis of Ocilrp2 mRNA levels after LPS stimulation on 2, 4, 8, and 12 h in mouse peritoneal macrophages. Untreated macrophages were used as a control. Data were normalized to the expression of GAPDH. (C) Western Blot analysis of Ocilrp2 protein levels after stimulated by LPS in mouse peritoneal macrophages. Ocilrp2 expression was analyzed by western blotting. Western blots and immunofluorescence images are representative of three independent experiments. (D, E) qRT-PCR analysis of Ocilrp2 mRNA level in macrophages treated with NF-kappa B inhibitor (BAY11-7082) or Erk inhibitor (SCH772984) for 1 h and then stimulated with LPS for 4 h or infected with VSV for 4 h. Data were normalized to the expression of GAPDH. Data were calculated from at least 3 independent experiments (means ± s.e.m.), **P< 0.01 (The two-tailed Student’s t-test). Image collected and cropped by CiteAb from the following open publication (https://pubmed.ncbi.nlm.nih.gov/36300111), licensed under a CC-BY license. Not internally tested by R&D Systems.Detection of OCILRP2/CLEC2i by Western Blot
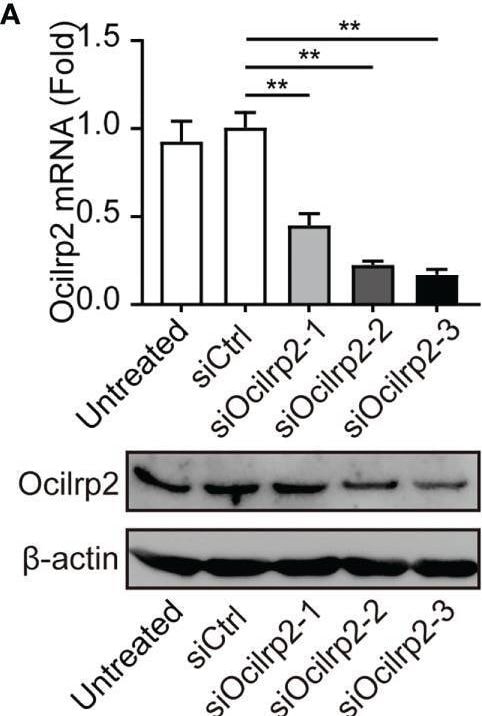
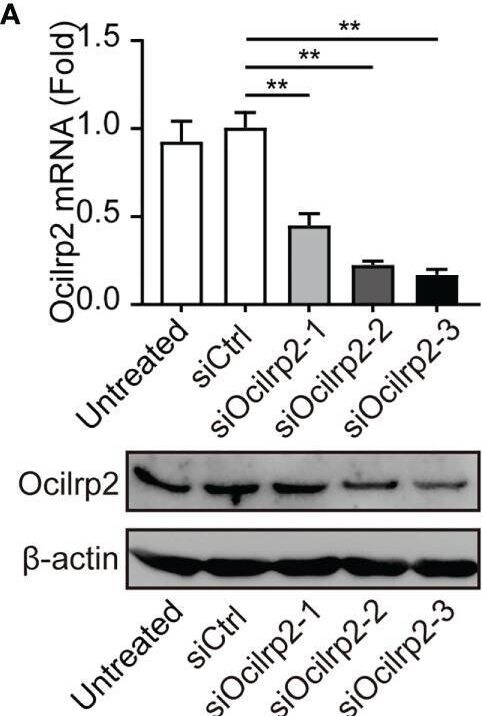
Silencing of Ocilrp2 promotes IL-6 expression in vitro and in vivo. (A) Peritoneal macrophages were transfected with scrambled siRNA (siCtrl) or three Ocilrp2 siRNAs (siOcilrp2-1, siOcilrp2-2, siOcilrp2-3) for 48 h, Ocilrp2 mRNA levels were detected with qRT-PCR, and Ocilrp2 protein levels were analyzed by Western Blot. The experiment was repeated three times. (B) qRT-PCR analysis of Ocilrp2 mRNA level in mouse peritoneal macrophages transfected with Ocilrp2 siRNA and 48 h later stimulated with LPS or infected with VSV for the indicated time.(C) qRT-PCR analysis of IL-6 mRNA level in macrophages transfected as in (A), and 48 h later stimulated with LPS for the indicated time and (D) ELISA of IL-6 in supernatants of macrophages stimulated with LPS for 8 h. (E) qRT-PCR analysis of IL-6 mRNA level in macrophages transfected as in (A), and 48 h later after VSV infection for the indicated time. (F) ELISA of IL-6 in supernatants of macrophages transfected as in (A), and 48 h later infection with VSV for 8 h. (G, H) Six C57BL/6 mice were transfected in vivo with Ocilrp2 siRNA or Control siRNA (2.5 mg/kg) for 2 days, respectively. Then, peritoneal macrophages and serum were collected at 12 h after intraperitoneal injection of LPS (10 mg/kg). qRT-PCR analysis of Ocilrp2 and IL-6 mRNA level in mouse peritoneal macrophages and (I) ELISA of IL-6 in serum. Data were calculated from at least 3 independent experiments (means ± s.e.m.), *P< 0.05, **P< 0.01 (The two-tailed Student’s t-test). Image collected and cropped by CiteAb from the following open publication (https://pubmed.ncbi.nlm.nih.gov/36300111), licensed under a CC-BY license. Not internally tested by R&D Systems.Detection of OCILRP2/CLEC2i by Western Blot
Silencing of Ocilrp2 promotes IL-6 expression in vitro and in vivo. (A) Peritoneal macrophages were transfected with scrambled siRNA (siCtrl) or three Ocilrp2 siRNAs (siOcilrp2-1, siOcilrp2-2, siOcilrp2-3) for 48 h, Ocilrp2 mRNA levels were detected with qRT-PCR, and Ocilrp2 protein levels were analyzed by Western Blot. The experiment was repeated three times. (B) qRT-PCR analysis of Ocilrp2 mRNA level in mouse peritoneal macrophages transfected with Ocilrp2 siRNA and 48 h later stimulated with LPS or infected with VSV for the indicated time.(C) qRT-PCR analysis of IL-6 mRNA level in macrophages transfected as in (A), and 48 h later stimulated with LPS for the indicated time and (D) ELISA of IL-6 in supernatants of macrophages stimulated with LPS for 8 h. (E) qRT-PCR analysis of IL-6 mRNA level in macrophages transfected as in (A), and 48 h later after VSV infection for the indicated time. (F) ELISA of IL-6 in supernatants of macrophages transfected as in (A), and 48 h later infection with VSV for 8 h. (G, H) Six C57BL/6 mice were transfected in vivo with Ocilrp2 siRNA or Control siRNA (2.5 mg/kg) for 2 days, respectively. Then, peritoneal macrophages and serum were collected at 12 h after intraperitoneal injection of LPS (10 mg/kg). qRT-PCR analysis of Ocilrp2 and IL-6 mRNA level in mouse peritoneal macrophages and (I) ELISA of IL-6 in serum. Data were calculated from at least 3 independent experiments (means ± s.e.m.), *P< 0.05, **P< 0.01 (The two-tailed Student’s t-test). Image collected and cropped by CiteAb from the following open publication (https://pubmed.ncbi.nlm.nih.gov/36300111), licensed under a CC-BY license. Not internally tested by R&D Systems.Detection of OCILRP2/CLEC2i by Western Blot
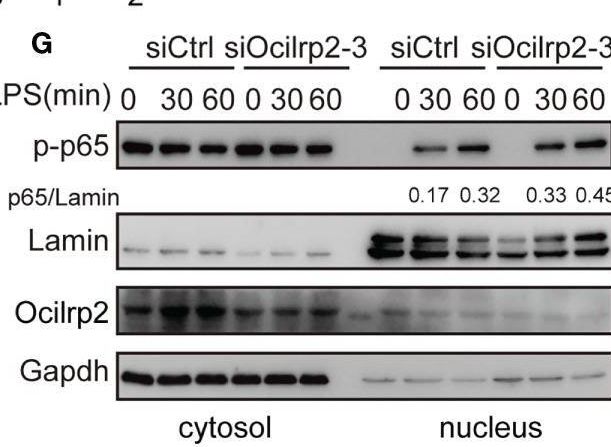
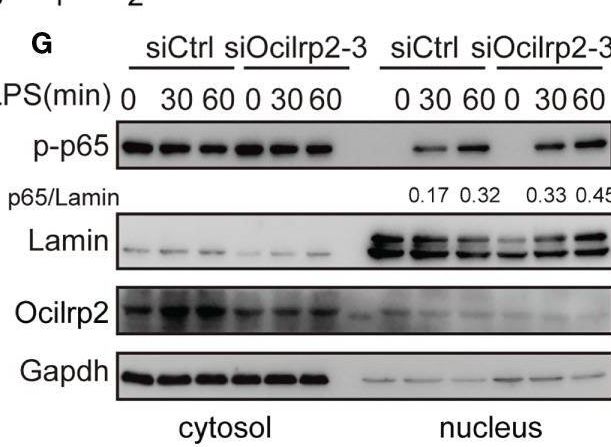
Silencing of Ocilrp2 accelerates the activation of the TLR4 signaling. Mouse peritoneal macrophages were treated with or without LPS (1 µg/ml) for different periods, as indicated, after transfection with siCtrl or siOcilrp2-3. (A)Then subjected to Western blot analyses using anti-Myd88, anti-Traf6, and anti- phosphorylated (p-) or total proteins of IKK alpha / beta, I kappa B alpha, and p65. (B) Densitometric quantification of the ratio of Myd88, Traf6, p-IKK alpha / beta, p-I kappa B alpha and p-p65. Data were analyzed by three independent experiments. (C) Immunoblot analysis of phosphorylated (p-) or total proteins of pErk, Jnk, and p38 for the indicated time. The beta -actin was detected as a loading control. (D) Densitometric quantification of the ratio of p-Erk, p-Jnk and p-p38. Data were analyzed by three independent experiments. (E) Mouse peritoneal macrophages were transfected with Ocilrp2 siRNA and 48hr later stimulated with LPS for the indicated times. Immunofluorescence analysis of the nuclear translocation of p65 and the co-localization between p65 (green) and cell nucleus (blue) (Bar, 50 μm). (F) Quantifying of p65 protein translocation based on the visual images in c, ~300 cells for each time point. (G) Immunoblot analysis of p65 protein in cytoplasm and nucleus of mouse peritoneal macrophages transfected with Ocilrp2 siRNA and 48 h later stimulated by LPS for the indicated time. Lamin A/C is used as an internal nuclear control. GAPDH is shown as a cytoplasm internal control. Data represent of the results of three independent experiments (means ± s.e.m). Significant differences compared to the control group are denoted by *P < 0.05, **P < 0.01 (two-tailed Student’s t-test). Image collected and cropped by CiteAb from the following open publication (https://pubmed.ncbi.nlm.nih.gov/36300111), licensed under a CC-BY license. Not internally tested by R&D Systems.Detection of OCILRP2/CLEC2i by Western Blot
Silencing of Ocilrp2 accelerates the activation of the TLR4 signaling. Mouse peritoneal macrophages were treated with or without LPS (1 µg/ml) for different periods, as indicated, after transfection with siCtrl or siOcilrp2-3. (A)Then subjected to Western blot analyses using anti-Myd88, anti-Traf6, and anti- phosphorylated (p-) or total proteins of IKK alpha / beta, I kappa B alpha, and p65. (B) Densitometric quantification of the ratio of Myd88, Traf6, p-IKK alpha / beta, p-I kappa B alpha and p-p65. Data were analyzed by three independent experiments. (C) Immunoblot analysis of phosphorylated (p-) or total proteins of pErk, Jnk, and p38 for the indicated time. The beta -actin was detected as a loading control. (D) Densitometric quantification of the ratio of p-Erk, p-Jnk and p-p38. Data were analyzed by three independent experiments. (E) Mouse peritoneal macrophages were transfected with Ocilrp2 siRNA and 48hr later stimulated with LPS for the indicated times. Immunofluorescence analysis of the nuclear translocation of p65 and the co-localization between p65 (green) and cell nucleus (blue) (Bar, 50 μm). (F) Quantifying of p65 protein translocation based on the visual images in c, ~300 cells for each time point. (G) Immunoblot analysis of p65 protein in cytoplasm and nucleus of mouse peritoneal macrophages transfected with Ocilrp2 siRNA and 48 h later stimulated by LPS for the indicated time. Lamin A/C is used as an internal nuclear control. GAPDH is shown as a cytoplasm internal control. Data represent of the results of three independent experiments (means ± s.e.m). Significant differences compared to the control group are denoted by *P < 0.05, **P < 0.01 (two-tailed Student’s t-test). Image collected and cropped by CiteAb from the following open publication (https://pubmed.ncbi.nlm.nih.gov/36300111), licensed under a CC-BY license. Not internally tested by R&D Systems.Detection of OCILRP2/CLEC2i by Western Blot
Downregulation of Ocilrp2 transcription in macrophages upon innate stimuli. (A) qRT-PCR analysis of Ocilrp2 mRNA levels in mouse peritoneal macrophages following LPS, Poly (I:C) or CpG stimulation or VSV, and SeV infection for 4 h. Data were normalized to the expression of GAPDH. (B) qRT-PCR analysis of Ocilrp2 mRNA levels after LPS stimulation on 2, 4, 8, and 12 h in mouse peritoneal macrophages. Untreated macrophages were used as a control. Data were normalized to the expression of GAPDH. (C) Western Blot analysis of Ocilrp2 protein levels after stimulated by LPS in mouse peritoneal macrophages. Ocilrp2 expression was analyzed by western blotting. Western blots and immunofluorescence images are representative of three independent experiments. (D, E) qRT-PCR analysis of Ocilrp2 mRNA level in macrophages treated with NF-kappa B inhibitor (BAY11-7082) or Erk inhibitor (SCH772984) for 1 h and then stimulated with LPS for 4 h or infected with VSV for 4 h. Data were normalized to the expression of GAPDH. Data were calculated from at least 3 independent experiments (means ± s.e.m.), **P< 0.01 (The two-tailed Student’s t-test). Image collected and cropped by CiteAb from the following open publication (https://pubmed.ncbi.nlm.nih.gov/36300111), licensed under a CC-BY license. Not internally tested by R&D Systems.Applications for Mouse OCILRP2/CLEC2i Antibody
Application
Recommended Usage
Western Blot
0.1 µg/mL
Sample: Recombinant Mouse OCILRP2/CLEC2i
Sample: Recombinant Mouse OCILRP2/CLEC2i
Formulation, Preparation, and Storage
Purification
Antigen Affinity-purified
Reconstitution
Reconstitute at 0.2 mg/mL in sterile PBS. For liquid material, refer to CoA for concentration.
Loading...
Formulation
Lyophilized from a 0.2 μm filtered solution in PBS with Trehalose. *Small pack size (SP) is supplied either lyophilized or as a 0.2 µm filtered solution in PBS.
Shipping
Lyophilized product is shipped at ambient temperature. Liquid small pack size (-SP) is shipped with polar packs. Upon receipt, store immediately at the temperature recommended below.
Stability & Storage
Use a manual defrost freezer and avoid repeated freeze-thaw cycles.
- 12 months from date of receipt, -20 to -70 °C as supplied.
- 1 month, 2 to 8 °C under sterile conditions after reconstitution.
- 6 months, -20 to -70 °C under sterile conditions after reconstitution.
Calculators
Background: OCILRP2/CLEC2i
Long Name
Osteoclast Inhibitory Lectin Related Protein 2
Alternate Names
CLEC2i, CLRG
Entrez Gene IDs
93675 (Mouse)
Gene Symbol
CLEC2I
UniProt
Additional OCILRP2/CLEC2i Products
Product Documents for Mouse OCILRP2/CLEC2i Antibody
Certificate of Analysis
To download a Certificate of Analysis, please enter a lot or batch number in the search box below.
Note: Certificate of Analysis not available for kit components.
Product Specific Notices for Mouse OCILRP2/CLEC2i Antibody
For research use only
Related Research Areas
Citations for Mouse OCILRP2/CLEC2i Antibody
Customer Reviews for Mouse OCILRP2/CLEC2i Antibody
There are currently no reviews for this product. Be the first to review Mouse OCILRP2/CLEC2i Antibody and earn rewards!
Have you used Mouse OCILRP2/CLEC2i Antibody?
Submit a review and receive an Amazon gift card!
$25/€18/£15/$25CAN/¥2500 Yen for a review with an image
$10/€7/£6/$10CAN/¥1110 Yen for a review without an image
Submit a review
Protocols
Find general support by application which include: protocols, troubleshooting, illustrated assays, videos and webinars.
- Cellular Response to Hypoxia Protocols
- R&D Systems Quality Control Western Blot Protocol
- Troubleshooting Guide: Western Blot Figures
- Western Blot Conditions
- Western Blot Protocol
- Western Blot Protocol for Cell Lysates
- Western Blot Troubleshooting
- Western Blot Troubleshooting Guide
- View all Protocols, Troubleshooting, Illustrated assays and Webinars
Loading...