Superoxide Dismutases (SODs), originally identified as Indophenoloxidase (IPO), are enzymes that catalyze the converversion of naturally-occuring but harmful superoxide radicals into molecular oxygen and hydrogen peroxide. Superoxide Dismutases 1, SOD1, also known as Cu/Zn SOD, soluble SOD, and IPO-A, is a soluble, cytoplasmic 16 kDa homodimer. Each SOD1 monomer binds one Cu2+ and Zn2+ ion. Three isozymes of SOD have been identified and are functionally related but have very modest sequence homology. SOD1 shares 23% and 27% sequence identity with SOD2 and SOD3, respectively. Mouse SOD1 is 97% aa identcal to rat SOD1. Mutations in SOD1 have been suggested to be the cause of familial amyotrophic lateral sclerosis (ALS). The ALS-causing mutations of SOD1 are scattered throughout the protein and provide no clear functional or structural clues to the underlying disease mechanism. The oligomerization hypothesis suggests that mutant SOD1 proteins become misfolded and consequently oligomerize into high molecular weight aggregates that result in the death of motor neurons. The oxidative damage hypothesis suggests that loss of function mutations in SOD1 result in the intracellular accumulation of the superoxide radical, leading to free radical-mediated damage, the release of cytochrome c, and apoptosis.
Mouse/Rat SOD1/Cu‑Zn SOD Antibody
R&D Systems | Catalog # AF3787


Key Product Details
Species Reactivity
Validated:
Mouse, Rat
Cited:
Human, Mouse, Rat, Naked mole-rat
Applications
Validated:
Western Blot
Cited:
Immunohistochemistry, Western Blot
Label
Unconjugated
Antibody Source
Polyclonal Goat IgG
Loading...
Product Specifications
Immunogen
E. coli-derived recombinant mouse SOD1
Met1-Gln154
Accession # P08228
Met1-Gln154
Accession # P08228
Specificity
Detects endogenous mouse and rat SOD1 in Western blots. In Western blots, this antibody shows no cross‑reacivity with recombinant human SOD2 or SOD3.
Clonality
Polyclonal
Host
Goat
Isotype
IgG
Scientific Data Images for Mouse/Rat SOD1/Cu‑Zn SOD Antibody
Detection of Mouse/Rat SOD1/Cu-Zn SOD by Western Blot.
Western blot shows lysates of NIH-3T3 mouse embryonic fibroblast cell line and NRK rat normal kidney cell line. PVDF membrane was probed with 0.2 µg/mL of Goat Anti-Mouse/Rat SOD1/Cu-Zn SOD Antigen Affinity-purified Polyclonal Antibody (Catalog # AF3787) followed by HRP-conjugated Anti-Goat IgG Secondary Antibody (Catalog # HAF017). A specific band was detected for SOD1/Cu-Zn SOD at approximately 19 kDa (as indicated). This experiment was conducted using Immunoblot Buffer Group 2.Detection of SOD1/Cu-Zn SOD by Western Blot
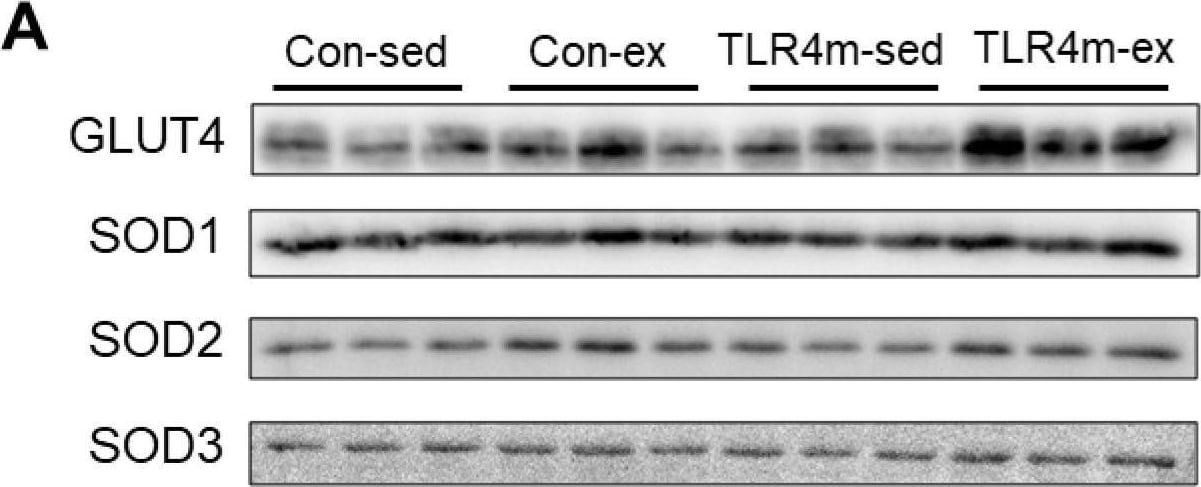
The relative expressions of molecules related to glucose metabolism and oxidative capacity in the plantaris. Glucose transporter 4 (GLUT4) (B), superoxide dismutase 1 (SOD1) (C), SOD2 (D), and SOD3 (E) were measured. Representative images of immunoblots are shown (A). Values are means ± standard error; n = 6 per group. ** p < 0.01 vs. sed within each strain; † p < 0.05 vs. controls within each condition. Image collected and cropped by CiteAb from the following open publication (https://pubmed.ncbi.nlm.nih.gov/35163799), licensed under a CC-BY license. Not internally tested by R&D Systems.Applications for Mouse/Rat SOD1/Cu‑Zn SOD Antibody
Application
Recommended Usage
Western Blot
0.2 µg/mL
Sample: NIH-3T3 mouse embryonic fibroblast cell line and NRK rat normal kidney cell line
Sample: NIH-3T3 mouse embryonic fibroblast cell line and NRK rat normal kidney cell line
Formulation, Preparation, and Storage
Purification
Antigen Affinity-purified
Reconstitution
Reconstitute at 0.2 mg/mL in sterile PBS. For liquid material, refer to CoA for concentration.
Loading...
Formulation
Lyophilized from a 0.2 μm filtered solution in PBS with Trehalose. *Small pack size (SP) is supplied either lyophilized or as a 0.2 µm filtered solution in PBS.
Shipping
Lyophilized product is shipped at ambient temperature. Liquid small pack size (-SP) is shipped with polar packs. Upon receipt, store immediately at the temperature recommended below.
Stability & Storage
Use a manual defrost freezer and avoid repeated freeze-thaw cycles.
- 12 months from date of receipt, -20 to -70 °C as supplied.
- 1 month, 2 to 8 °C under sterile conditions after reconstitution.
- 6 months, -20 to -70 °C under sterile conditions after reconstitution.
Calculators
Background: SOD1/Cu-Zn SOD
Long Name
Superoxide Dismutase-1
Alternate Names
Cu-Zn SOD, CuZn SOD, Ipo1, IPOA, SOD, cytosolic, SOD, Soluble
Gene Symbol
SOD1
UniProt
Additional SOD1/Cu-Zn SOD Products
Product Documents for Mouse/Rat SOD1/Cu‑Zn SOD Antibody
Certificate of Analysis
To download a Certificate of Analysis, please enter a lot or batch number in the search box below.
Note: Certificate of Analysis not available for kit components.
Product Specific Notices for Mouse/Rat SOD1/Cu‑Zn SOD Antibody
For research use only
Citations for Mouse/Rat SOD1/Cu‑Zn SOD Antibody
Customer Reviews for Mouse/Rat SOD1/Cu‑Zn SOD Antibody
There are currently no reviews for this product. Be the first to review Mouse/Rat SOD1/Cu‑Zn SOD Antibody and earn rewards!
Have you used Mouse/Rat SOD1/Cu‑Zn SOD Antibody?
Submit a review and receive an Amazon gift card!
$25/€18/£15/$25CAN/¥2500 Yen for a review with an image
$10/€7/£6/$10CAN/¥1110 Yen for a review without an image
Submit a review
Protocols
Find general support by application which include: protocols, troubleshooting, illustrated assays, videos and webinars.
- Cellular Response to Hypoxia Protocols
- R&D Systems Quality Control Western Blot Protocol
- Troubleshooting Guide: Western Blot Figures
- Western Blot Conditions
- Western Blot Protocol
- Western Blot Protocol for Cell Lysates
- Western Blot Troubleshooting
- Western Blot Troubleshooting Guide
- View all Protocols, Troubleshooting, Illustrated assays and Webinars
Loading...