-
Q: an the N-2 MAX Media Supplement be stored frozen as 1 mL aliquots?
A: Yes. The N-2 MAX Media Supplement may be stored frozen as 1 mL aliquots. The frozen aliquots should not undergo multiple freeze-thaws before use.
-
Q: Does N-2 MAX Media Supplement (Catalog # AR009) contain any components that are calcium salts?
-
Q: Does N-2 MAX Media Supplement (Catalog # AR009) contain any proteins that are produced using Baculovirus?
-
Q: Does the N-2 MAX Media Supplement contain any animal-derived components other than Human Transferrin?
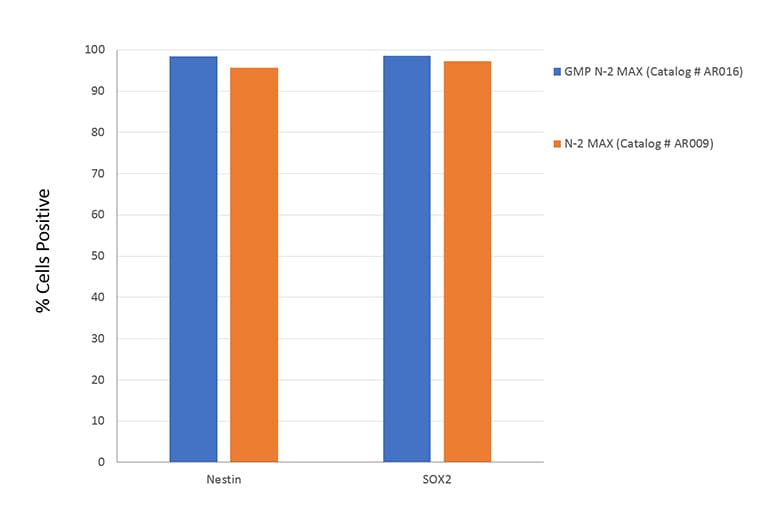
A: No. The N-2 MAX Media Supplement does not contain any animal-derived components other than Human Transferrin. GMP N-2 MAX Media Supplement is an animal-free supplement (Catalog # AR016).
-
Q: How do you recommend thawing this media supplement prior to use?
A: We recommend thawing the product overnight at 2-8 °C, or on the benchtop at room temperature with gently mixing and monitoring until it has fully thawed, prior to use or preparing any single-use aliquots.
-
Q: How many freeze/thaw cycles can N-2 MAX Media Supplement (100X), Catalog # AR009, undergo?
A: The N-2 MAX Media Supplement should be able to undergo one freeze/thaw cycyle as long as the product is used witin the expiration date.
-
Q: What is the difference between N-2 Plus Media Supplement (Catalog # AR003) and N-2 MAX Media Supplement (Catalog # AR009)?
A:
The N-2 Plus Media Supplement (Catalog # AR003) contains Bovine Insulin whereas N-2 MAX Media Supplement (Catalog # AR009) contains Recombinant Human Insulin. All other media components and concentrations in N-2 Plus and N-2 MAX are the same. These two products perform comparably in side-by-side testing. Due to a limited supply of bovine insulin, the products are priced differently.
-
Q: an the N-2 MAX Media Supplement be stored frozen as 1 mL aliquots?
A: Yes. The N-2 MAX Media Supplement may be stored frozen as 1 mL aliquots. The frozen aliquots should not undergo multiple freeze-thaws before use.
-
Q: Does N-2 MAX Media Supplement (Catalog # AR009) contain any components that are calcium salts?
-
Q: Does N-2 MAX Media Supplement (Catalog # AR009) contain any proteins that are produced using Baculovirus?
-
Q: Does the N-2 MAX Media Supplement contain any animal-derived components other than Human Transferrin?
A: No. The N-2 MAX Media Supplement does not contain any animal-derived components other than Human Transferrin. GMP N-2 MAX Media Supplement is an animal-free supplement (Catalog # AR016).
-
Q: How do you recommend thawing this media supplement prior to use?
A: We recommend thawing the product overnight at 2-8 °C, or on the benchtop at room temperature with gently mixing and monitoring until it has fully thawed, prior to use or preparing any single-use aliquots.
-
Q: How many freeze/thaw cycles can N-2 MAX Media Supplement (100X), Catalog # AR009, undergo?
A: The N-2 MAX Media Supplement should be able to undergo one freeze/thaw cycyle as long as the product is used witin the expiration date.
-
Q: What is the difference between N-2 Plus Media Supplement (Catalog # AR003) and N-2 MAX Media Supplement (Catalog # AR009)?
A:
The N-2 Plus Media Supplement (Catalog # AR003) contains Bovine Insulin whereas N-2 MAX Media Supplement (Catalog # AR009) contains Recombinant Human Insulin. All other media components and concentrations in N-2 Plus and N-2 MAX are the same. These two products perform comparably in side-by-side testing. Due to a limited supply of bovine insulin, the products are priced differently.
-
Q: an the N-2 MAX Media Supplement be stored frozen as 1 mL aliquots?
A: Yes. The N-2 MAX Media Supplement may be stored frozen as 1 mL aliquots. The frozen aliquots should not undergo multiple freeze-thaws before use.
-
Q: Does N-2 MAX Media Supplement (Catalog # AR009) contain any components that are calcium salts?
-
Q: Does N-2 MAX Media Supplement (Catalog # AR009) contain any proteins that are produced using Baculovirus?
-
Q: Does the N-2 MAX Media Supplement contain any animal-derived components other than Human Transferrin?
A: No. The N-2 MAX Media Supplement does not contain any animal-derived components other than Human Transferrin. GMP N-2 MAX Media Supplement is an animal-free supplement (Catalog # AR016).
-
Q: How do you recommend thawing this media supplement prior to use?
A: We recommend thawing the product overnight at 2-8 °C, or on the benchtop at room temperature with gently mixing and monitoring until it has fully thawed, prior to use or preparing any single-use aliquots.
-
Q: How many freeze/thaw cycles can N-2 MAX Media Supplement (100X), Catalog # AR009, undergo?
A: The N-2 MAX Media Supplement should be able to undergo one freeze/thaw cycyle as long as the product is used witin the expiration date.
-
Q: What is the difference between N-2 Plus Media Supplement (Catalog # AR003) and N-2 MAX Media Supplement (Catalog # AR009)?
A:
The N-2 Plus Media Supplement (Catalog # AR003) contains Bovine Insulin whereas N-2 MAX Media Supplement (Catalog # AR009) contains Recombinant Human Insulin. All other media components and concentrations in N-2 Plus and N-2 MAX are the same. These two products perform comparably in side-by-side testing. Due to a limited supply of bovine insulin, the products are priced differently.
-
Q: an the N-2 MAX Media Supplement be stored frozen as 1 mL aliquots?
A: Yes. The N-2 MAX Media Supplement may be stored frozen as 1 mL aliquots. The frozen aliquots should not undergo multiple freeze-thaws before use.
-
Q: Does N-2 MAX Media Supplement (Catalog # AR009) contain any components that are calcium salts?
-
Q: Does N-2 MAX Media Supplement (Catalog # AR009) contain any proteins that are produced using Baculovirus?
-
Q: Does the N-2 MAX Media Supplement contain any animal-derived components other than Human Transferrin?
A: No. The N-2 MAX Media Supplement does not contain any animal-derived components other than Human Transferrin. GMP N-2 MAX Media Supplement is an animal-free supplement (Catalog # AR016).
-
Q: How do you recommend thawing this media supplement prior to use?
A: We recommend thawing the product overnight at 2-8 °C, or on the benchtop at room temperature with gently mixing and monitoring until it has fully thawed, prior to use or preparing any single-use aliquots.
-
Q: How many freeze/thaw cycles can N-2 MAX Media Supplement (100X), Catalog # AR009, undergo?
A: The N-2 MAX Media Supplement should be able to undergo one freeze/thaw cycyle as long as the product is used witin the expiration date.
-
Q: What is the difference between N-2 Plus Media Supplement (Catalog # AR003) and N-2 MAX Media Supplement (Catalog # AR009)?
A:
The N-2 Plus Media Supplement (Catalog # AR003) contains Bovine Insulin whereas N-2 MAX Media Supplement (Catalog # AR009) contains Recombinant Human Insulin. All other media components and concentrations in N-2 Plus and N-2 MAX are the same. These two products perform comparably in side-by-side testing. Due to a limited supply of bovine insulin, the products are priced differently.
-
Q: an the N-2 MAX Media Supplement be stored frozen as 1 mL aliquots?
A: Yes. The N-2 MAX Media Supplement may be stored frozen as 1 mL aliquots. The frozen aliquots should not undergo multiple freeze-thaws before use.
-
Q: Does N-2 MAX Media Supplement (Catalog # AR009) contain any components that are calcium salts?
-
Q: Does N-2 MAX Media Supplement (Catalog # AR009) contain any proteins that are produced using Baculovirus?
-
Q: Does the N-2 MAX Media Supplement contain any animal-derived components other than Human Transferrin?
A: No. The N-2 MAX Media Supplement does not contain any animal-derived components other than Human Transferrin. GMP N-2 MAX Media Supplement is an animal-free supplement (Catalog # AR016).
-
Q: How do you recommend thawing this media supplement prior to use?
A: We recommend thawing the product overnight at 2-8 °C, or on the benchtop at room temperature with gently mixing and monitoring until it has fully thawed, prior to use or preparing any single-use aliquots.
-
Q: How many freeze/thaw cycles can N-2 MAX Media Supplement (100X), Catalog # AR009, undergo?
A: The N-2 MAX Media Supplement should be able to undergo one freeze/thaw cycyle as long as the product is used witin the expiration date.
-
Q: What is the difference between N-2 Plus Media Supplement (Catalog # AR003) and N-2 MAX Media Supplement (Catalog # AR009)?
A:
The N-2 Plus Media Supplement (Catalog # AR003) contains Bovine Insulin whereas N-2 MAX Media Supplement (Catalog # AR009) contains Recombinant Human Insulin. All other media components and concentrations in N-2 Plus and N-2 MAX are the same. These two products perform comparably in side-by-side testing. Due to a limited supply of bovine insulin, the products are priced differently.
-
Q: an the N-2 MAX Media Supplement be stored frozen as 1 mL aliquots?
A: Yes. The N-2 MAX Media Supplement may be stored frozen as 1 mL aliquots. The frozen aliquots should not undergo multiple freeze-thaws before use.
-
Q: Does N-2 MAX Media Supplement (Catalog # AR009) contain any components that are calcium salts?
-
Q: Does N-2 MAX Media Supplement (Catalog # AR009) contain any proteins that are produced using Baculovirus?
-
Q: Does the N-2 MAX Media Supplement contain any animal-derived components other than Human Transferrin?
A: No. The N-2 MAX Media Supplement does not contain any animal-derived components other than Human Transferrin. GMP N-2 MAX Media Supplement is an animal-free supplement (Catalog # AR016).
-
Q: How do you recommend thawing this media supplement prior to use?
A: We recommend thawing the product overnight at 2-8 °C, or on the benchtop at room temperature with gently mixing and monitoring until it has fully thawed, prior to use or preparing any single-use aliquots.
-
Q: How many freeze/thaw cycles can N-2 MAX Media Supplement (100X), Catalog # AR009, undergo?
A: The N-2 MAX Media Supplement should be able to undergo one freeze/thaw cycyle as long as the product is used witin the expiration date.
-
Q: What is the difference between N-2 Plus Media Supplement (Catalog # AR003) and N-2 MAX Media Supplement (Catalog # AR009)?
A:
The N-2 Plus Media Supplement (Catalog # AR003) contains Bovine Insulin whereas N-2 MAX Media Supplement (Catalog # AR009) contains Recombinant Human Insulin. All other media components and concentrations in N-2 Plus and N-2 MAX are the same. These two products perform comparably in side-by-side testing. Due to a limited supply of bovine insulin, the products are priced differently.
-
Q: an the N-2 MAX Media Supplement be stored frozen as 1 mL aliquots?
A: Yes. The N-2 MAX Media Supplement may be stored frozen as 1 mL aliquots. The frozen aliquots should not undergo multiple freeze-thaws before use.
-
Q: Does N-2 MAX Media Supplement (Catalog # AR009) contain any components that are calcium salts?
-
Q: Does N-2 MAX Media Supplement (Catalog # AR009) contain any proteins that are produced using Baculovirus?
-
Q: Does the N-2 MAX Media Supplement contain any animal-derived components other than Human Transferrin?
A: No. The N-2 MAX Media Supplement does not contain any animal-derived components other than Human Transferrin. GMP N-2 MAX Media Supplement is an animal-free supplement (Catalog # AR016).
-
Q: How do you recommend thawing this media supplement prior to use?
A: We recommend thawing the product overnight at 2-8 °C, or on the benchtop at room temperature with gently mixing and monitoring until it has fully thawed, prior to use or preparing any single-use aliquots.
-
Q: How many freeze/thaw cycles can N-2 MAX Media Supplement (100X), Catalog # AR009, undergo?
A: The N-2 MAX Media Supplement should be able to undergo one freeze/thaw cycyle as long as the product is used witin the expiration date.
-
Q: What is the difference between N-2 Plus Media Supplement (Catalog # AR003) and N-2 MAX Media Supplement (Catalog # AR009)?
A:
The N-2 Plus Media Supplement (Catalog # AR003) contains Bovine Insulin whereas N-2 MAX Media Supplement (Catalog # AR009) contains Recombinant Human Insulin. All other media components and concentrations in N-2 Plus and N-2 MAX are the same. These two products perform comparably in side-by-side testing. Due to a limited supply of bovine insulin, the products are priced differently.