LPS-induced CXC chemokine (LIX) is a chemokine originally cloned from LPS-stimulated mouse fibroblasts. Rat LIX shares approximately 74% amino acid (aa) sequence identity with mouse LIX and is likely the rat orthologue to mouse LIX. Rat LIX cDNA encodes a 130 aa residue precursor with a predicted 37 aa residue signal peptide and a 93 aa residue mature protein. Among human CXC chemokines, rat LIX is most closely related to human GCP-2 and ENA-78. Rat LIX also differs from these two human proteins by having an extended carboxy-terminus. The amino-terminal 115 residues of rat LIX shares 59% and 53% aa sequence homology with human GCP-2 and ENA-78, respectively. It is not clear if LIX should be considered an orthologue of GCP-2 or ENA-78. Yet, mouse LIX was alternatively named mouse GCP-2.
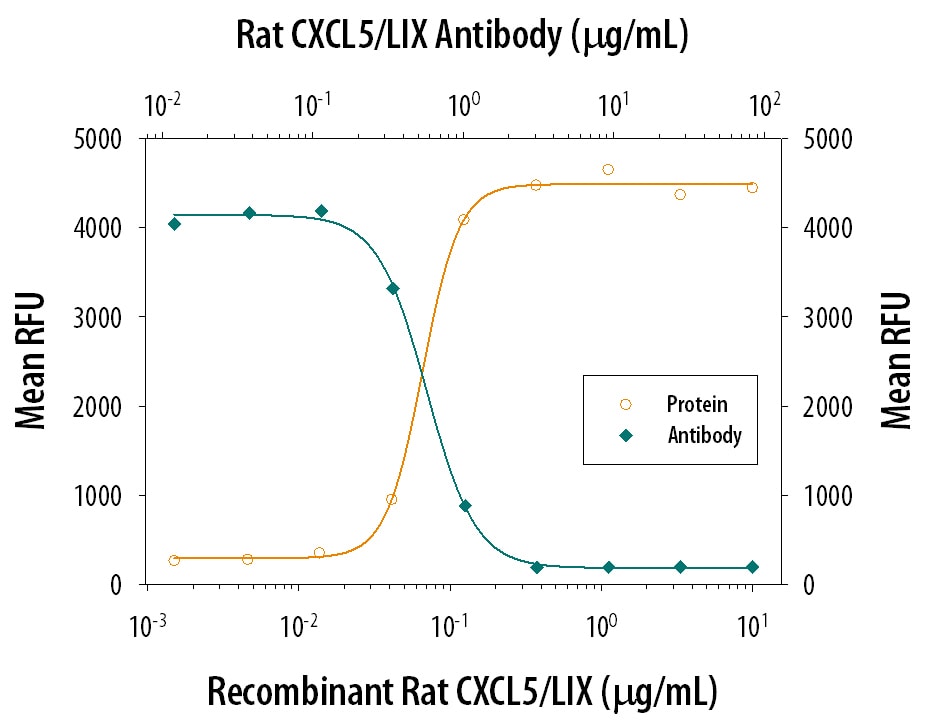
Purified recombinant rat LIX is C-terminally truncated. This is consistent with the observation that natural mouse LIX purified from fibroblasts and epithelial cells also contains multiple amino-terminal and carboxy-terminal truncated isoforms. The shorter isoforms of the natural protein were reported to be more active than the longer forms. The bioactivity of rat LIX can be mediated through CXCR2.