MuSK (Muscle-Specific Kinase) is a receptor tyrosine kinase expressed in muscle that regulates the formation of the neuromuscular junction. It binds the heparin sulfate proteoglycan Agrin to promote Acetylcholine Receptor clustering.


Key Product Details
Validated by
Biological Validation
Species Reactivity
Validated:
Rat
Cited:
Human, Mouse, Rat, Transgenic Mouse
Applications
Validated:
Western Blot, Neutralization
Cited:
Immunohistochemistry, Western Blot, Neutralization, Immunocytochemistry, Immunoprecipitation
Label
Unconjugated
Antibody Source
Polyclonal Goat IgG
Loading...
Product Specifications
Immunogen
Mouse myeloma cell line NS0-derived recombinant rat MuSK
Specificity
Detects rat MuSK in direct ELISAs and Western blots.
Clonality
Polyclonal
Host
Goat
Isotype
IgG
Endotoxin Level
<0.10 EU per 1 μg of the antibody by the LAL method.
Scientific Data Images for Rat MuSK Antibody
Acetylcholine Receptor Clustering Mediated by MuSK and Neutralization by Rat MuSK Antibody.
Recombinant Rat Agrin (Catalog # 550-AG) induces MuSK-dependent acetylcholine receptor clustering on myotubes differentiated from the C2C12 mouse myoblast cell line in a dose-dependent manner (orange line). Acetylcholine Receptor Clustering elicited by Recombinant Rat Agrin (16 ng/mL) is neutralized (green line) by increasing concentrations of Goat Anti-Rat MuSK Antigen Affinity-purified Polyclonal Antibody (Catalog # AF562). The ND50 is typically 0.5-2 µg/mL.Detection of Human MuSK by Immunocytochemistry/ Immunofluorescence
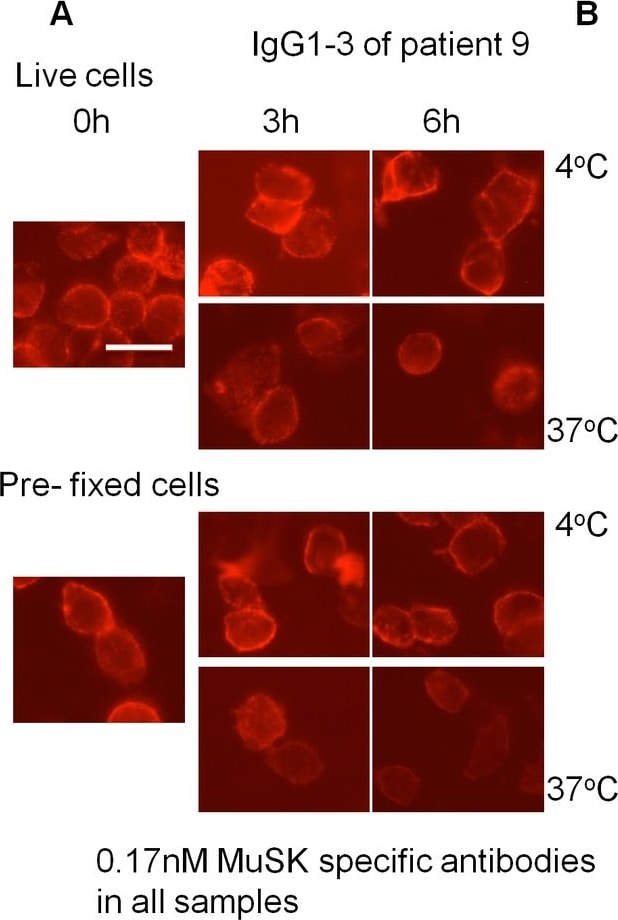
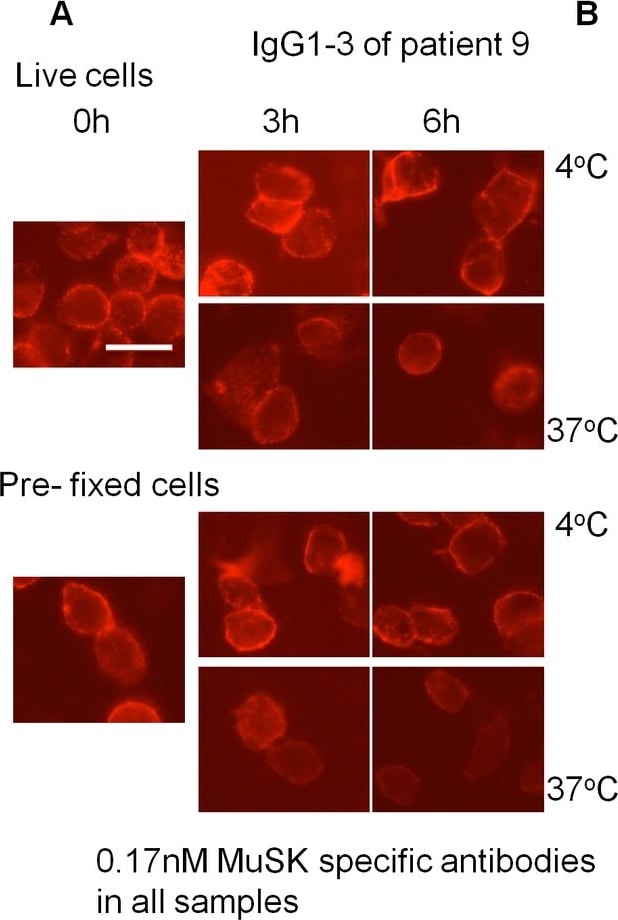
MuSK patient IgG4 or IgG1-3 do not induce endocytosis of MuSK.Patient plasma or purified IgG1-3 or IgG4 were applied at 0.17nM final concentration of MuSK antibody to HEK293 cells expressing MuSK at the cell surface. Cells were either incubated at 4°C to prevent, or at 37°C to allow, endocytosis. Patient antibody binding was visualised by addition of a secondary fluorescent anti-human antibody at the end of the experiment. (A) An example of cells that were incubated with IgG1-3 from patient 9. Robust staining was observed after 6 hours incubation at both temperatures. Scale bar=25µm. (B) Staining was scored by two individuals as described in methods, and the scores were normalised to the score at 0 hours for each condition. A slight decrease in staining was observed for cells incubated at 37°C, and pre-fixed cells also showed this reduction. (C) As a positive control for endocytosis, IgG1-3 or IgG4 was cross-linked by addition of Alexa Fluor 568-conjugated anti-human IgG prior to incubation. Example images show cells treated with patient 9 IgG4 and IgG1-3 in the presence and absence of cross-linking anti-human IgG after 6 hours incubation at 37°C. There is a clear difference when the cross-linking secondary antibody is present. Scale bar =50µm (D) Cross-linking by the secondary antibody induced internalisation of human anti-MuSK antibodies as well as MuSK-EGFP as early as 30 minutes, as observed by confocal microscopy. An example image of a cell from a confocal z-stack with orthogonal side-views is shown. The arrow shows internalised secondary Alexa Fluor 568-conjugated anti-human IgG (red) colocalised with EGFP-tagged MuSK (green). Scale bar =5µm. Image collected and cropped by CiteAb from the following open publication (https://dx.plos.org/10.1371/journal.pone.0080695), licensed under a CC-BY license. Not internally tested by R&D Systems.Detection of Human MuSK by Immunocytochemistry/ Immunofluorescence
MuSK patient IgG4 or IgG1-3 do not induce endocytosis of MuSK.Patient plasma or purified IgG1-3 or IgG4 were applied at 0.17nM final concentration of MuSK antibody to HEK293 cells expressing MuSK at the cell surface. Cells were either incubated at 4°C to prevent, or at 37°C to allow, endocytosis. Patient antibody binding was visualised by addition of a secondary fluorescent anti-human antibody at the end of the experiment. (A) An example of cells that were incubated with IgG1-3 from patient 9. Robust staining was observed after 6 hours incubation at both temperatures. Scale bar=25µm. (B) Staining was scored by two individuals as described in methods, and the scores were normalised to the score at 0 hours for each condition. A slight decrease in staining was observed for cells incubated at 37°C, and pre-fixed cells also showed this reduction. (C) As a positive control for endocytosis, IgG1-3 or IgG4 was cross-linked by addition of Alexa Fluor 568-conjugated anti-human IgG prior to incubation. Example images show cells treated with patient 9 IgG4 and IgG1-3 in the presence and absence of cross-linking anti-human IgG after 6 hours incubation at 37°C. There is a clear difference when the cross-linking secondary antibody is present. Scale bar =50µm (D) Cross-linking by the secondary antibody induced internalisation of human anti-MuSK antibodies as well as MuSK-EGFP as early as 30 minutes, as observed by confocal microscopy. An example image of a cell from a confocal z-stack with orthogonal side-views is shown. The arrow shows internalised secondary Alexa Fluor 568-conjugated anti-human IgG (red) colocalised with EGFP-tagged MuSK (green). Scale bar =5µm. Image collected and cropped by CiteAb from the following open publication (https://dx.plos.org/10.1371/journal.pone.0080695), licensed under a CC-BY license. Not internally tested by R&D Systems.Detection of Mouse MuSK by Western Blot
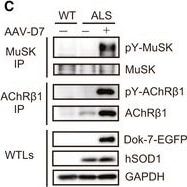
DOK7 gene therapy suppresses motor nerve terminal degeneration at the NMJ in ALS miceAForelimb grip strength of wild‐type (WT) mice&that of control&AAV‐D7 treatment groups of ALS mice at P84 (WT, n = 5 mice; control group, n = 5 mice; treatment group, n = 6 mice). Values are means ± SEM. **P = 0.0016 (WT vs. control group)&0.0003 (WT vs. treatment group) by one‐way analysis of variance (ANOVA) with Bonferroni's post hoc test. n.s., not significant.BThe difference in cycle threshold ( delta Ct) between the human SOD1‐G93A transgene&the reference mouse apob gene. To calculate the human transgene level, the delta Ct value of hSOD1 was subtracted from the delta Ct value of apob (control group, n = 5 mice; treatment group, n = 6 mice).C–GWT or ALS mice treated or not with 1.2 × 1012 vg of AAV‐D7 at P90 subjected to the following assays at P120. Tyrosine phosphorylation of MuSK or AChR in the hind‐limb muscle. MuSK or AChR beta 1 subunit (AChR beta 1) immunoprecipitates (IP) from whole‐tissue lysates (WTLs) of the hind‐limb muscle immunoblotted for phosphotyrosine (pY), MuSK,&AChR beta 1. WTLs blotted for Dok‐7‐EGFP, human SOD1,&GAPDH (C). Whole‐mount staining of NMJs on the diaphragm muscle. Motor nerve terminals (green)&postsynaptic AChRs (red) stained with anti‐synapsin‐1 antibodies& alpha ‐bungarotoxin, respectively. Expression of Dok‐7‐EGFP fusion protein (gray) was monitored by EGFP. Scale bars, 50 μm. NT, not treated (D). The size of AChR cluster (E), the size of motor nerve terminal (F),&innervation ratio (G) (WT‐NT, n = 5 mice; ALS‐NT, n = 5 mice; ALS‐AAV‐D7, n = 6 mice). Values are means ± SD. (E) ***P < 0.0001; (F) **P = 0.0077, ***P = 0.0001; (G) *P = 0.0134, ***P < 0.0001 by one‐way ANOVA with Dunnett's post hoc test.Source data are available online for this figure. Image collected&cropped by CiteAb from the following open publication (https://pubmed.ncbi.nlm.nih.gov/28490573), licensed under a CC-BY license. Not internally tested by R&D Systems.Detection of MuSK by Western Blot
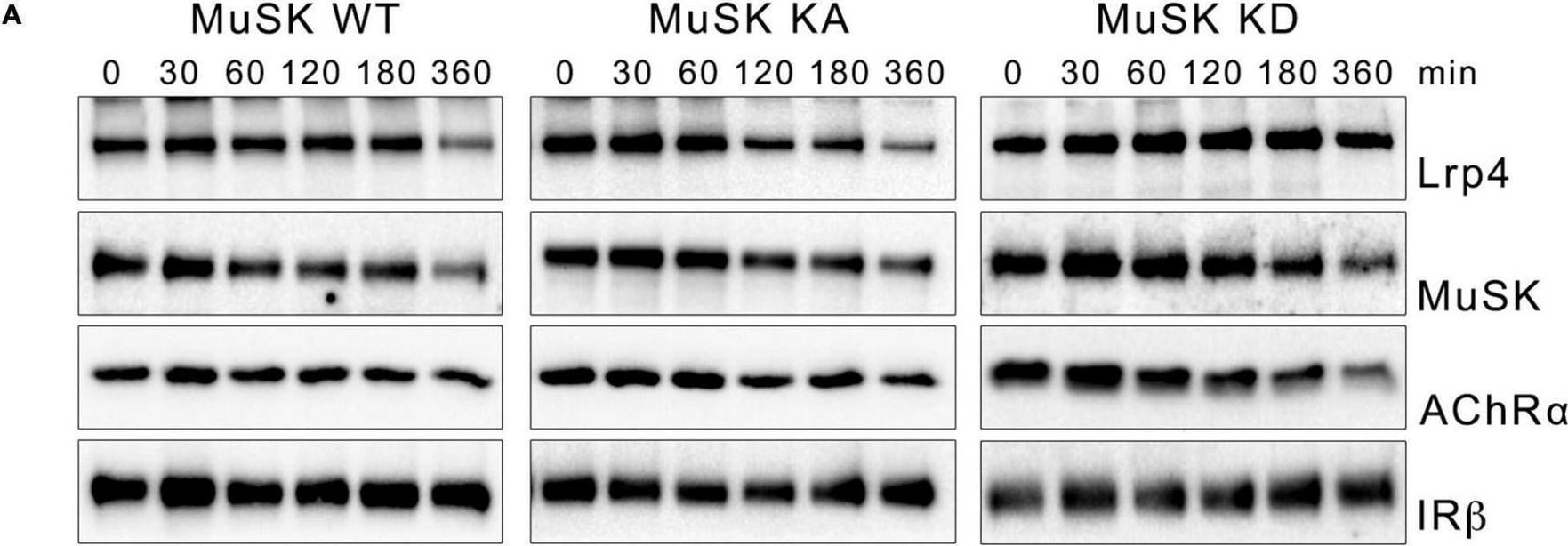
MuSK kinase activity fails to induce MuSK internalization. (A) Muscle cells expressing MuSK wildtype (WT), MuSK kinase-active (KA) or MuSK kinase-dead (KD) were treated with cycloheximide for the indicated time periods (in minutes). Surface proteins were isolated using biotinylation followed by streptavidin pulldown. Protein expression was determined by immunoblotting using antibodies against Lrp4, MuSK, AChR alpha and IR beta. (B) Quantification of surface protein expression in muscle cells expressing MuSK wildtype (blue), kinase-active (gray) and kinase-dead (orange) is shown over a time course of 6 h. Isolated surface proteins were normalized against IR beta. Timepoint 0 was set to 1. Data are presented as means ± SEM, n > 5. (C) Quantification of total protein expression as function of time. Total protein samples of MuSK wildtype (blue), kinase-active (gray) and kinase-dead (orange) were normalized against Actin. Timepoint 0 was set to 1. Data are presented as means ± SEM; n > 5. Image collected and cropped by CiteAb from the following open publication (https://pubmed.ncbi.nlm.nih.gov/35370548), licensed under a CC-BY license. Not internally tested by R&D Systems.Detection of MuSK by Western Blot
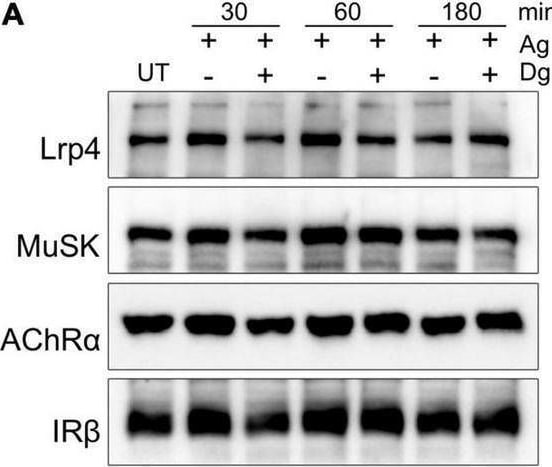
Lrp4, MuSK and AChR internalization takes place independent of Dynamin. Myotubes were pretreated with cycloheximide with or without Dyngo-4a and subsequently stimulated with 5 nM Agrin for the indicated time periods (in minutes). Surface proteins were isolated using biotinylation followed by streptavidin pulldown. (A) Samples were subjected to SDS-PAGE and analyzed by immunoblotting. Surface expression was determined using antibodies against Lrp4, MuSK, AChR alpha and IR beta. (B) Quantification of surface protein expression as function of time and Dyngo-4a treatment (− Dyngo-4a, blue; + Dyngo-4a, orange) is shown. Isolated surface proteins were normalized against IR beta. Timepoint 0 was set to 1. Data are presented as means ± SEM; n > 4. Ag, Agrin; Dg, Dyngo-4a; UT, untreated. Image collected and cropped by CiteAb from the following open publication (https://pubmed.ncbi.nlm.nih.gov/35370548), licensed under a CC-BY license. Not internally tested by R&D Systems.Detection of MuSK by Western Blot
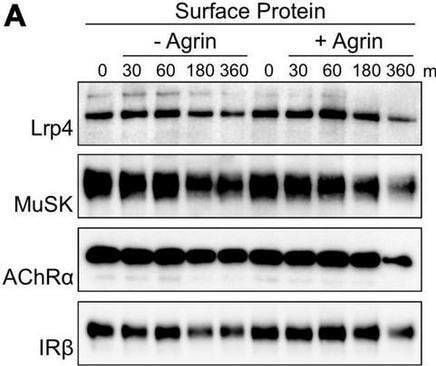
Agrin stimulation increases MuSK and AChR internalization but does not affect Lrp4 internalization. Muscle cells were pretreated with cycloheximide and incubated with or without 5 nM Agrin for the indicated time periods (in minutes). Surface proteins were isolated using biotinylation followed by streptavidin pulldown. (A) Samples were subjected to SDS-PAGE and analyzed by immunoblotting. Surface and total protein expression was determined using antibodies against Lrp4, MuSK, AChR alpha, IR beta and Actin. (B) Quantification of surface protein expression as function of time and Agrin treatment (− Agrin, blue; + Agrin, orange) is shown. Isolated surface proteins were normalized against IR beta. Timepoint 0 was set to 1. Data are presented as means ± SEM; n > 9. (C) Quantification of total protein expression as function of time and Agrin treatment (− Agrin, blue; + Agrin, orange). Total protein samples were normalized against Actin. Timepoint 0 was set to 1. Data are presented as means ± SEM; n > 5. (D) Muscle cells were pretreated with cycloheximide in the presence or absence of Primaquine (Pq) followed by stimulation with 5 nM Agrin for the indicated time periods (in minutes). Surface proteins were isolated using biotinylation followed by streptavidin pulldown. Samples were subjected to SDS-PAGE and analyzed by immunoblotting. Quantification of surface protein expression as function of time and Primaquine treatment (− Primaquine, orange; + Primaquine, gray) is shown. Isolated surface proteins were normalized against IR beta. Timepoint 0 was set to 1. Data are presented as means ± SEM; n > 3. *p < 0.05; **p < 0.01; ***p < 0.001. Image collected and cropped by CiteAb from the following open publication (https://pubmed.ncbi.nlm.nih.gov/35370548), licensed under a CC-BY license. Not internally tested by R&D Systems.Detection of MuSK by Western Blot
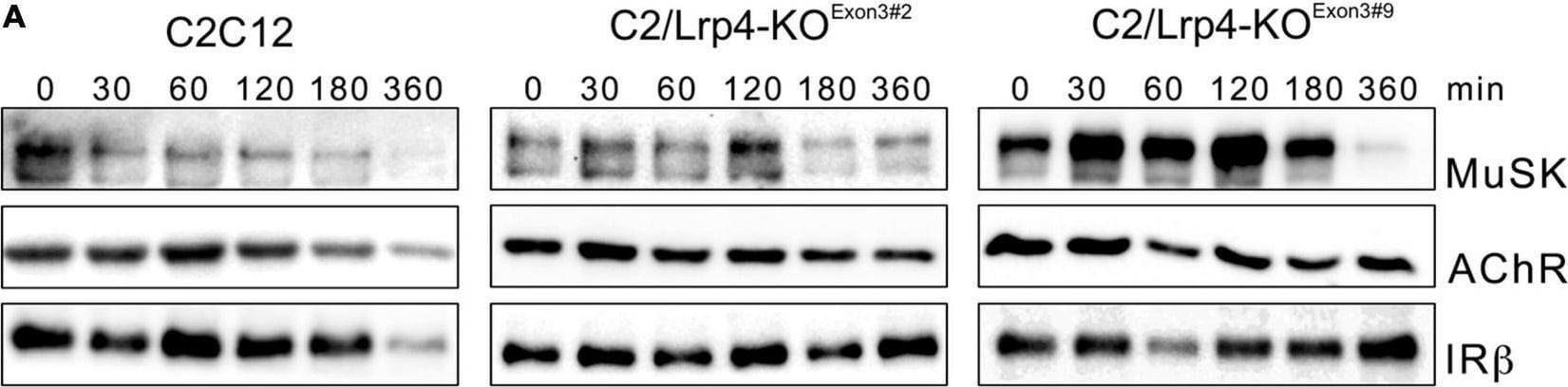
MuSK endocytosis occurs independent of Lrp4. (A) Wild-type C2 cells and C2/Lrp4-KO cells were treated with cycloheximide for the indicated time points (in minutes). Biotinylation was used to isolate surface proteins followed by analysis by immunoblotting using antibodies against MuSK, AChR alpha and IR beta. (B) Quantification of MuSK and AChR surface protein expression in C2 wild-type cells (blue) and C2/Lrp4-KO cells (Exon3#2: orange; Exon3#9: gray) is shown over a time course of 6 h. Isolated surface proteins were normalized against IR beta. Timepoint 0 was set to 1. Data are presented as means ± SEM, n > 4. (C) Quantification of total protein expression as function of time in C2 wild-type cells (blue) and C2/Lrp4-KO cells (Exon3#2: orange; Exon3#9: gray). Total protein samples were normalized against Actin. Timepoint 0 was set to 1. Data are presented as means ± SEM; n > 4. Image collected and cropped by CiteAb from the following open publication (https://pubmed.ncbi.nlm.nih.gov/35370548), licensed under a CC-BY license. Not internally tested by R&D Systems.Applications for Rat MuSK Antibody
Application
Recommended Usage
Western Blot
0.1 µg/mL
Sample: Recombinant Rat MuSK
Sample: Recombinant Rat MuSK
Neutralization
Measured by its ability to neutralize MuSK-dependent acetylcholine receptor clustering on myotubes differentiated from the C2C12 mouse myoblast cell line [Ferns, M.J. et al. (1993) Neuron 11:491]. The Neutralization Dose (ND50) is typically 0.5-2 µg/mL in the presence of 16 ng/mL Recombinant Rat Agrin.
Formulation, Preparation, and Storage
Purification
Antigen Affinity-purified
Reconstitution
Reconstitute at 0.2 mg/mL in sterile PBS. For liquid material, refer to CoA for concentration.
Loading...
Formulation
Lyophilized from a 0.2 μm filtered solution in PBS with Trehalose. *Small pack size (SP) is supplied either lyophilized or as a 0.2 µm filtered solution in PBS.
Shipping
Lyophilized product is shipped at ambient temperature. Liquid small pack size (-SP) is shipped with polar packs. Upon receipt, store immediately at the temperature recommended below.
Stability & Storage
Use a manual defrost freezer and avoid repeated freeze-thaw cycles.
- 12 months from date of receipt, -20 to -70 °C as supplied.
- 1 month, 2 to 8 °C under sterile conditions after reconstitution.
- 6 months, -20 to -70 °C under sterile conditions after reconstitution.
Calculators
Background: MuSK
Long Name
Muscle-specific Receptor Tyrosine Kinase
Alternate Names
EC 2.7.10, EC 2.7.10.1, MGC126323, MGC126324, muscle, skeletal, receptor tyrosine kinase, MuSK, skeletal receptor tyrosine-protein kinase
Gene Symbol
MUSK
Additional MuSK Products
Product Documents for Rat MuSK Antibody
Certificate of Analysis
To download a Certificate of Analysis, please enter a lot or batch number in the search box below.
Note: Certificate of Analysis not available for kit components.
Product Specific Notices for Rat MuSK Antibody
For research use only
Related Research Areas
Citations for Rat MuSK Antibody
Customer Reviews for Rat MuSK Antibody
There are currently no reviews for this product. Be the first to review Rat MuSK Antibody and earn rewards!
Have you used Rat MuSK Antibody?
Submit a review and receive an Amazon gift card!
$25/€18/£15/$25CAN/¥2500 Yen for a review with an image
$10/€7/£6/$10CAN/¥1110 Yen for a review without an image
Submit a review
Protocols
Find general support by application which include: protocols, troubleshooting, illustrated assays, videos and webinars.
- Cellular Response to Hypoxia Protocols
- R&D Systems Quality Control Western Blot Protocol
- Troubleshooting Guide: Western Blot Figures
- Western Blot Conditions
- Western Blot Protocol
- Western Blot Protocol for Cell Lysates
- Western Blot Troubleshooting
- Western Blot Troubleshooting Guide
- View all Protocols, Troubleshooting, Illustrated assays and Webinars
Loading...