Recombinant Mouse Angiopoietin-like 3 (aa 17-220) Biotin, CF
R&D Systems | Catalog # BT8344
N-Terminal Fragment

Loading...
Key Product Details
- R&D Systems CHO-derived Recombinant Mouse Angiopoietin-like 3 (aa 17-220) Biotin (BT8344)
- Quality control testing to verify active proteins with lot specific assays by in-house scientists
- All R&D Systems proteins are covered with a 100% guarantee
Source
CHO
Accession Number
Structure / Form
Biotinylated protein via sugars
Conjugate
Biotin
Applications
Bioactivity
Loading...
Product Specifications
Source
Chinese Hamster Ovary cell line, CHO-derived mouse Angiopoietin-like Protein 3/ANGPTL3 protein
Ser17-Thr206, with a C-terminal 6-His tag
Ser17-Thr206, with a C-terminal 6-His tag
Purity
>95%, by SDS-PAGE visualized with Silver Staining and quantitative densitometry by Coomassie® Blue Staining.
Endotoxin Level
<0.10 EU per 1 μg of the protein by the LAL method.
N-terminal Sequence Analysis
Ser17
Predicted Molecular Mass
23 kDa (unlabeled)
Activity
Measured by its ability to promote the expansion of E16 rat liver mononuclear cells in vitro, in the presence of Recombinant Mouse SCF/c‑kit Ligand (Catalog # 455-MC), Recombinant Mouse Thrombopoietin/Tpo (Catalog # 488-TO), and Recombinant Mouse Flt‑3 Ligand (Catalog # 427-FL).
The ED50 for this effect is 20-120 ng/mL in the presence of a cross‑linking antibody, Mouse Anti-polyHistidine Monoclonal Antibody (Catalog # MAB050).
The ED50 for this effect is 20-120 ng/mL in the presence of a cross‑linking antibody, Mouse Anti-polyHistidine Monoclonal Antibody (Catalog # MAB050).
Scientific Data Images for Recombinant Mouse Angiopoietin-like 3 (aa 17-220) Biotin, CF
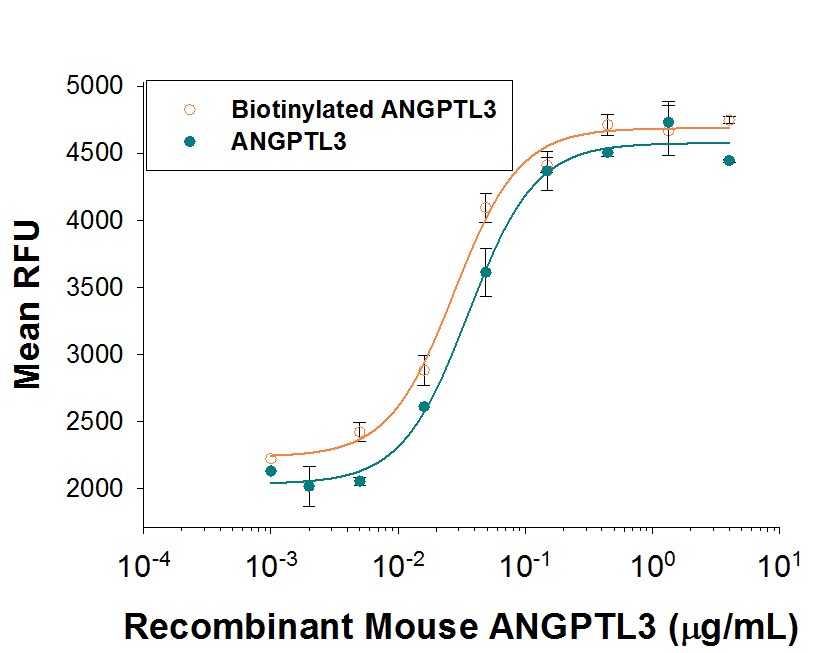
Recombinant Mouse Angiopoietin-like 3 (aa 17-220) Biotin Bioactivity
Both Biotinylated rmANGPTL3 (Catalog # BT8344) and unlabeled rmANGPTL3 (Catalog # 8344-AN) stimulate the expansion of E16-E18 rat liver mononuclear cells link, in the presence of rmSCF (Catalog # 455-MC), rmTpo (Catalog # 488-TO), and rmFlt-3L (Catalog # 427-FL). The ED50 for this effect is 20-120 ng/mL in the presence of a cross-linking antibody, His Tag MAb (Catalog # MAB050). The similarity in activity highlights that the biotinylated protein is fully functional.Formulation, Preparation, and Storage
BT8344
| Formulation | Lyophilized from a 0.2 μm filtered solution in MOPS, NaCl and CHAPS. |
| Reconstitution | Reconstitute at 100 μg/mL in PBS.
Loading...
|
| Shipping | The product is shipped at ambient temperature. Upon receipt, store it immediately at the temperature recommended below. |
| Stability & Storage | Use a manual defrost freezer and avoid repeated freeze-thaw cycles.
|
Calculators
Background: Angiopoietin-like Protein 3/ANGPTL3
N- and C-terminal fragments containing the coiled coil domain and fibrinogen-like domains, respectively (4, 5). The cleavage of ANGPTL3 by Furin and Proprotein Convertase 5/6 is enhanced by its interaction with the related ANGPTL8 (6, 7). ANGPTL3 is found as 70 kDa, 50 kDa, and 32 kDa species and can form weakly associated noncovalent multimers in vitro (3, 4). ANGPTL3 directly inhibits lipoprotein lipase (LPL) and endothelial lipase (EL), enzymes responsible for hydrolyzing circulating triglycerides and HDL phospholipids (8, 9). This activity requires a putative heparin-binding motif which is N-terminal to the coiled coil domain (4). Proteolytic removal of the fibrinogen-like domain from the N-terminal fragment serves to activate ANGPTL3 and increase its ability to inhibit LPL in vitro and function in vivo [Ono 41804]. ANGPTL3 promotes an increase in circulating triglyceride levels without altering VLDL or HDL secretion or uptake (4, 5, 8). ANGPTL3 knockout mice are hypolipidemic and have elevated LPL activity (10). ANGPTL3 expression in vivo is upregulated by LXR agonists and downregulated by insulin, leptin, and agonists of TR beta or PPAR beta (11-14). Dysregulated ANGPTL3 expression and elevated plasma triglyceride levels are characteristic of some strains of obese and diabetic mice (5, 8, 12). ANGPTL3 does not bind Tie1 or Tie2, but its fibrinogen-like domain interacts with Integrin alpha V beta 3 to induce endothelial cell adhesion, migration, and neovascularization (15). ANGPTL3, secreted by fetal liver cells, also promotes the expansion of hematopoietic stem cells (16).
References
- Santulli, G. (2014) Front. Endocrinol. (Lausanne) 5:4.
- Conklin, D. et al. (1999) Genomics 62:477.
- Ge, H. et al. (2005) J. Lipid Res. 46:1484.
- Ono, M. et al. (2003) J. Biol. Chem. 278:41804.
- Koishi, R. et al. (2002) Nat. Genet. 30:151.
- Essalmani, R. et al. (2013) J. Biol. Chem. 288:26410.
- Quagliarini, F. et al. (2012) Proc. Natl. Acad. Sci. USA 109:19751.
- Shimizugawa, T. et al. (2002) J. Biol. Chem. 277:33742.
- Shimamura, M. et al. (2007) Arterioscler. Thromb. Vasc. Biol. 27:366.
- Koster, A. et al. (2005) Endocrinology 146:4943.
- Inaba, T. et al. (2003) J. Biol. Chem. 278:21344.
- Shimamura, M. et al. (2004) Biochem. Biophys. Res. Commun. 322:1080.
- Fugier, C. et al. (2006) J. Biol. Chem. 281:11553.
- Matsusue, K. et al. (2006) Mol. Cell Endocrinol. 256:23.
- Camenisch, G. et al. (2002) J. Biol. Chem. 277:17281.
- Zhang, C.C. et al. (2006) Nat. Med. 12:240.
Alternate Names
AGNPT5, Ang-5, Angiopoietin-5, ANGPTL3
Gene Symbol
ANGPTL3
UniProt
Additional Angiopoietin-like Protein 3/ANGPTL3 Products
- All Products for Angiopoietin-like Protein 3/ANGPTL3
- Angiopoietin-like Protein 3/ANGPTL3 cDNA Clones
- Angiopoietin-like Protein 3/ANGPTL3 ELISA Kits
- Angiopoietin-like Protein 3/ANGPTL3 Lysates
- Angiopoietin-like Protein 3/ANGPTL3 Primary Antibodies
- Angiopoietin-like Protein 3/ANGPTL3 Proteins and Enzymes
- Angiopoietin-like Protein 3/ANGPTL3 Simple Plex
Product Documents for Recombinant Mouse Angiopoietin-like 3 (aa 17-220) Biotin, CF
Certificate of Analysis
To download a Certificate of Analysis, please enter a lot or batch number in the search box below.
Note: Certificate of Analysis not available for kit components.
Product Specific Notices for Recombinant Mouse Angiopoietin-like 3 (aa 17-220) Biotin, CF
For research use only
Related Research Areas
Customer Reviews for Recombinant Mouse Angiopoietin-like 3 (aa 17-220) Biotin, CF
There are currently no reviews for this product. Be the first to review Recombinant Mouse Angiopoietin-like 3 (aa 17-220) Biotin, CF and earn rewards!
Have you used Recombinant Mouse Angiopoietin-like 3 (aa 17-220) Biotin, CF?
Submit a review and receive an Amazon gift card!
$25/€18/£15/$25CAN/¥2500 Yen for a review with an image
$10/€7/£6/$10CAN/¥1110 Yen for a review without an image
Submit a review
Loading...
