TLR4 Antibody (76B357.1) - BSA Free
Novus Biologicals | Catalog # NB100-56566

Key Product Details
Validated by
Species Reactivity
Validated:
Cited:
Predicted:
Applications
Validated:
Cited:
Label
Antibody Source
Format
Product Specifications
Immunogen
Clonality
Host
Isotype
Theoretical MW
Disclaimer note: The observed molecular weight of the protein may vary from the listed predicted molecular weight due to post translational modifications, post translation cleavages, relative charges, and other experimental factors.
Scientific Data Images for TLR4 Antibody (76B357.1) - BSA Free
Flow Cytometry: TLR4 Antibody (76B357.1) - BSA Free [NB100-56566]
Flow Cytometry: TLR4 Antibody (76B357.1) [NB100-56566] - Analysis using the Alexa Fluor (R) 647 conjugate of NBP2-27149. TLR4 expression on monocytes from human peripheral blood. PBMC were stained in a 2 color flow test, with CD14 PE version of this antibody and 1 ug of either isotype control (left) or TLR4-Alexa Fluor 647 (right). PPI negative, CD14+ cells were gated for analysis.Immunohistochemistry: TLR4 Antibody (76B357.1) - BSA Free [NB100-56566]
TLR4-Antibody-76B357-1-Immunohistochemistry-NB100-56566-img0040.jpgWestern Blot: TLR4 Antibody (76B357.1)BSA Free [NB100-56566]
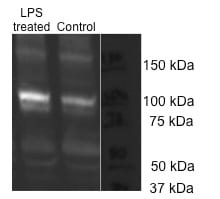
Western Blot: TLR4 Antibody (76B357.1) [NB100-56566] - Analysis using 2 ug/mL on (A) human intestine and 6 ug/mL on (B) mouse intestine and (C) rat intestine lysate.Immunohistochemistry: TLR4 Antibody (76B357.1) - BSA Free [NB100-56566]
TLR4-Antibody-76B357-1-Immunohistochemistry-NB100-56566-img0041.jpgFlow (Intracellular): TLR4 Antibody (76B357.1) - BSA Free [NB100-56566]
Flow (Intracellular): TLR4 Antibody (76B357.1) [NB100-56566] - Analysis using PE conjugate of NBP2-27149. An intracellular stain was performed on Jurkat cells with TLR4 antibody (76B357.1) NBP2-27149PE (blue) and an isotype control MAB004 (orange). Cells were fixed with 4% PFA and then permeablized with 0.1% saponin. Cells were incubated in an antibody dilution of 2.5 ug/mL for 30 minutes at room temperature. Both antibodies were conjugated to phycoerythrin.Immunocytochemistry/ Immunofluorescence: TLR4 Antibody (76B357.1) - BSA Free [NB100-56566]
Immunocytochemistry/Immunofluorescence: TLR4 Antibody (76B357.1) [NB100-56566] - RH-30 cells were fixed in 4% paraformaldehyde for 10 minutes and permeabilized in 0.05% Triton X-100 in PBS for 5 minutes. The cells were incubated with anti-TLR4 Antibody (76B357.1) NB100-56566 at 1 ug/ml overnight at 4C and detected with an anti-mouse Dylight 488 (Green) at a 1:1000 dilution for 60 minutes. Nuclei were counterstained with DAPI (Blue). Cells were imaged using a 100X objective and digitally deconvolved.Immunohistochemistry: TLR4 Antibody (76B357.1) - BSA Free [NB100-56566]
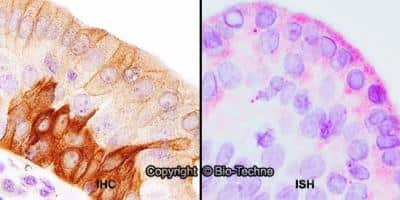
TLR4-Antibody-76B357-1-Immunohistochemistry-NB100-56566-img0037.jpgImmunohistochemistry-Paraffin: TLR4 Antibody (76B357.1) - BSA Free [NB100-56566]
Immunohistochemistry-Paraffin: TLR4 Antibody (76B357.1) [NB100-56566] - Analysis of rat salivary gland tissue section at 1:100 dilution. The antibody generated a membrane-cytoplasmic staining in the tissue with stronger signal in ductal epithelial cells.Immunohistochemistry-Paraffin: TLR4 Antibody (76B357.1) - BSA Free [NB100-56566]
Immunohistochemistry-Paraffin: TLR4 Antibody (76B357.1) [NB100-56566] - Tissue section of normal human skin stained with antibody at 5 ug/mL. Membrane-cytoplasmic immunopositivity of TLR4 was primarily observed in the pigmented basel cells and the adjacent keratinocytes in the epidermal layer.Flow Cytometry: TLR4 Antibody (76B357.1) - BSA Free [NB100-56566]
Flow Cytometry: TLR4 Antibody (76B357.1) [NB100-56566] - An intracellular stain was performed on RH30 cells with TLR4 Antibody (76B357.1) NB100-56566 (blue) and a matched mouse IgG2b Kappa isotype control (orange) MAB004. Cells were fixed with 4% PFA and then permeabilized with 0.1% saponin. Cells were incubated in an antibody dilution of 2.5 ug/mL for 30 minutes at room temperature, followed by Mouse IgG (H+L) Cross-Adsorbed Secondary Antibody, Dylight 550 (35503, Thermo Fisher).Immunohistochemistry-Paraffin: TLR4 Antibody (76B357.1) - BSA Free [NB100-56566]
Immunohistochemistry-Paraffin: TLR4 Antibody (76B357.1) [NB100-56566] - Human skin stained with at 5 ug/mL, peroxidase-conjugate and DAB chromogen. Staining of formalin-fixed tissues is enhanced by boiling tissue sections in 10 mM sodium citrate buffer, pH 6.0 for 10-20 min followed by cooling at RT for 20 min.Immunohistochemistry-Paraffin: TLR4 Antibody (76B357.1) - BSA Free [NB100-56566]
Immunohistochemistry-Paraffin: TLR4 Antibody (76B357.1) [NB100-56566] - Analysis of TLR4 in FFPE human colon tissue using an isotype control (top) and NB100-56566 (bottom) at 5 ug/mL.Immunohistochemistry-Paraffin: TLR4 Antibody (76B357.1) - BSA Free [NB100-56566]
Immunohistochemistry-Paraffin: TLR4 Antibody (76B357.1) [NB100-56566] - Human testis tissue stained with antibody at 5 ug/mL.Immunohistochemistry-Frozen: TLR4 Antibody (76B357.1) - BSA Free [NB100-56566]
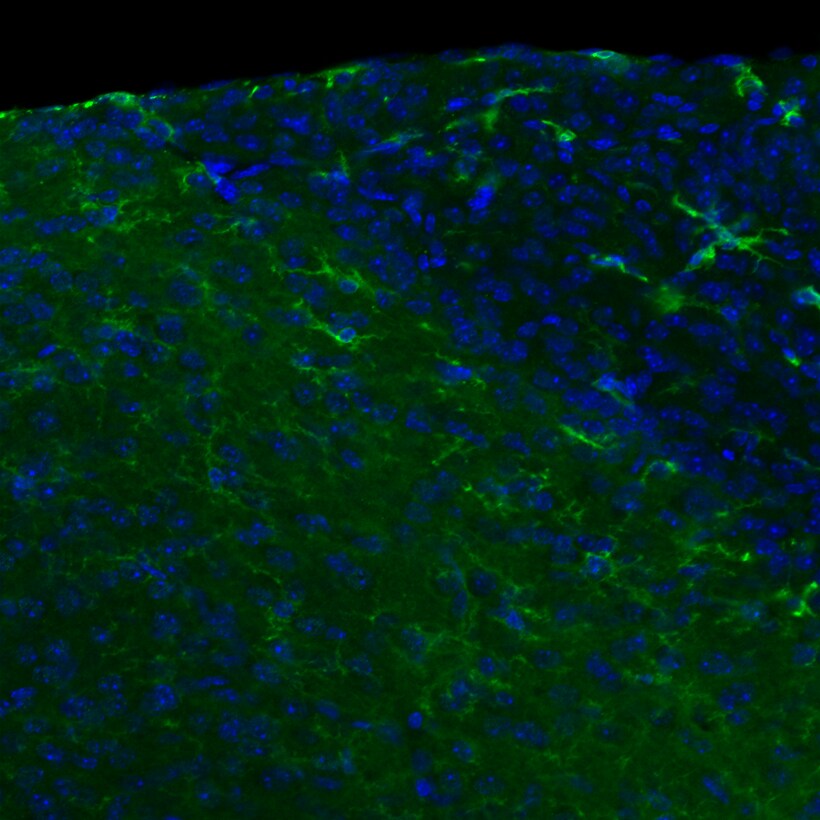
Immunohistochemistry-Frozen: TLR4 Antibody (76B357.1) [NB100-56566] - This image is TLR4(green) and nucleus(blue) at area postrema of the adult male mouse brain, x20 magnification. Primary antibody diluted 1:500. IHC-Fr image submitted by a verified customer review.Flow Cytometry: TLR4 Antibody (76B357.1) - BSA Free [NB100-56566]
Flow Cytometry: TLR4 Antibody (76B357.1) [NB100-56566] - Analysis of formaldehyde fixed THP-1 cells (human monocytic leukemia cells) using 2 ug/10^6 cells TLR4 antibody (clone 76B357.1) with detection employing a donkey anti-mouse IgG (H+L) cross adsorbed secondary antibody, (DyLight 488 conjugated). Isotype control samples incubated with mouse IgG2b isotype control antibody were processed in parallal under the same assay conditions.Flow Cytometry: TLR4 Antibody (76B357.1) - BSA Free [NB100-56566]
Flow Cytometry: TLR4 Antibody (76B357.1) [NB100-56566] - An intracellular stain was performed on Jurkat cells with TLR4 Antibody (76B357.1) NB100-56566 (blue) and a matched mouse IgG2b Kappa isotype control (orange) MAB004. Cells were fixed with 4% PFA and then permeabilized with 0.1% saponin. Cells were incubated in an antibody dilution of 2.5 ug/mL for 30 minutes at room temperature, followed by Mouse IgG (H+L) Cross-Adsorbed Secondary Antibody, Dylight 550 (35503, Thermo Fisher).Flow Cytometry: TLR4 Antibody (76B357.1) - BSA Free [NB100-56566]
Flow Cytometry: TLR4 Antibody (76B357.1) [NB100-56566] - An intracellular stain was performed on Raw264.7 cells with TLR4 Antibody (76B357.1) NB100-56566 (blue) and a matched mouse IgG2b Kappa isotype control (orange) MAB004. Cells were fixed with 4% PFA and then permeabilized with 0.1% saponin. Cells were incubated in an antibody dilution of 2.5 ug/mL for 30 minutes at room temperature, followed by Mouse IgG (H+L) Cross-Adsorbed Secondary Antibody, Dylight 550 (35503, Thermo Fisher).Immunohistochemistry: TLR4 Antibody (76B357.1) - BSA Free [NB100-56566] -
Representative images of immunohistochemical staining of pancreatic adenocarcinoma upregulated factor (PAUF) and toll-like receptor 4 (TLR4) in human ovarian cancer specimens. The top row (A,B) represents normal ovarian tissue, middle row (C,D) shows immunoreactivity of ovarian carcinomas and the bottom row (E,F) indicates immunonegativity of ovarian caricinoma. The stromal cells of normal ovarian tissue show immunonegativity for PAUF and TLR4. The scale bar represents 100 μm.Western Blot: TLR4 Antibody (76B357.1) - BSA Free [NB100-56566] -
Western Blot: TLR4 Antibody (76B357.1) - BSA Free [NB100-56566] - Increased toll‐like receptor 4 (TLR4) expression in the myocardium of chronic heart failure (CHF) rats. (A) TLR4 mRNA levels in infarct & remote myocardium of sham & CHF rats (n = 6/group). (B) Representative Western blot images & (C) quantification of TLR4 proteins in infarct & remote myocardium of sham & CHF rats (n = 4/group). (D) Representative immunohistochemistry images of heart sections stained with TLR4 (green) & CD45 (red). The yellow box indicates the enlarged area shown on the right (data are means ± SD, *P < 0.05, **P < 0.01 versus respective sham). Image collected & cropped by CiteAb from the following publication (https://pubmed.ncbi.nlm.nih.gov/26290459), licensed under a CC-BY license. Not internally tested by Novus Biologicals.Western Blot: TLR4 Antibody (76B357.1) - BSA Free [NB100-56566] -
Western Blot: TLR4 Antibody (76B357.1) - BSA Free [NB100-56566] - Regulation of FOXC1 on TLR3/4 expression in mice subjected to myocardial ischaemia (MI). The siRNA against FOXC1 (si‐FOXC1) or FOXC1 adenovirus (Ad‐FOXC1) was injected into the left ventricle just after LAD ligation to generate FOXC1 knockdown (A) or FOXC1 overexpression (B), & the negative control (NC) siRNA & Ad‐GFP served as control, respectively. The mRNA (upper panel) & protein (middle & lower panel) levels of TLR3/4 were determined after 2 wk. Data are means ± SEM aP < .05, AP < .01 vs respective MI Image collected & cropped by CiteAb from the following publication (https://pubmed.ncbi.nlm.nih.gov/31517441), licensed under a CC-BY license. Not internally tested by Novus Biologicals.Immunohistochemistry: TLR4 Antibody (76B357.1) - BSA Free [NB100-56566] -
Immunohistochemistry: TLR4 Antibody (76B357.1) - BSA Free [NB100-56566] - Representative images of immunohistochemical staining of pancreatic adenocarcinoma upregulated factor (PAUF) & toll-like receptor 4 (TLR4) in human ovarian cancer specimens. The top row (A,B) represents normal ovarian tissue, middle row (C,D) shows immunoreactivity of ovarian carcinomas & the bottom row (E,F) indicates immunonegativity of ovarian caricinoma. The stromal cells of normal ovarian tissue show immunonegativity for PAUF & TLR4. The scale bar represents 100 μm. Image collected & cropped by CiteAb from the following publication (https://pubmed.ncbi.nlm.nih.gov/30111860), licensed under a CC-BY license. Not internally tested by Novus Biologicals.Western Blot: TLR4 Antibody (76B357.1) - BSA Free [NB100-56566] -
Western Blot: TLR4 Antibody (76B357.1) - BSA Free [NB100-56566] - Increased toll‐like receptor 4 (TLR4) expression in the surviving cardiomyocytes of chronic heart failure (CHF) rats. (A) Representative immunofluorescent images of TLR4 in cardiomyocytes isolated from sham & CHF rats. (B) TLR4 mRNA levels in cardiomyocytes isolated from sham & CHF rats. (C) Representative Western blot images & (D) quantification of TLR4 proteins in cardiomyocytes isolated from sham & CHF rats (data are means ± SD, n = 6/group, **P < 0.01 versus sham). Image collected & cropped by CiteAb from the following publication (https://pubmed.ncbi.nlm.nih.gov/26290459), licensed under a CC-BY license. Not internally tested by Novus Biologicals.Western Blot: TLR4 Antibody (76B357.1) - BSA Free [NB100-56566] -
Western Blot: TLR4 Antibody (76B357.1) - BSA Free [NB100-56566] - Up‐regulation of FOXC1 & TLR3/4 protein levels under myocardial ischaemia. Representative Western blot images & quantitative data for FOXC1, TLR3 & TLR4 proteins in ischaemia models of mice (A), H9c2 cells (B) & NRVMs (C) are shown herein. Validation data of the antibodies for TLR3 & TLR4 are shown in Figure S1. Data are means ± SEM. n = ~4‐5/group. P values from the one‐way ANOVAs: 0.018 (TLR3 protein in mice),.002 (TLR4 protein in mice) & 0.006 (FOXC1 protein in mice). aP < .05, AP < .01 vs control Image collected & cropped by CiteAb from the following publication (https://pubmed.ncbi.nlm.nih.gov/31517441), licensed under a CC-BY license. Not internally tested by Novus Biologicals.Western Blot: TLR4 Antibody (76B357.1) - BSA Free [NB100-56566] -
Western Blot: TLR4 Antibody (76B357.1) - BSA Free [NB100-56566] - (a) Cell viability of MM cells exposed to increasing doses of LPS for 24 h. (b) Immunoblotting analysis of TLR4 protein expression levels in H929 & L363 cell lines after incubation with various LPS concentrations. Probing with beta -ACTIN was used as total protein loading reference. (c) Flow cytometry analysis for TLR4 expression of MM cell lines before & after exposure to 1 μg/ml LPS for 24 h. (d) Cell viability of MM cell lines pre-treated with 0.5 μg/ml TLR4 inhibitor for 24 h before stimulation with LPS for 24 h. (e) Cell vViability of H929 & JJN3 cell lines exposed to increasing concentrations of TLR4 inhibitor for 48 h. beta -ACTIN probing was used as reference for total protein input. Image collected & cropped by CiteAb from the following publication (https://pubmed.ncbi.nlm.nih.gov/30824741), licensed under a CC-BY license. Not internally tested by Novus Biologicals.Western Blot: TLR4 Antibody (76B357.1) - BSA Free [NB100-56566] -
Western Blot: TLR4 Antibody (76B357.1) - BSA Free [NB100-56566] - Regulation of FOXC1 on TLR3/4 expression in mice subjected to myocardial ischaemia (MI). The siRNA against FOXC1 (si‐FOXC1) or FOXC1 adenovirus (Ad‐FOXC1) was injected into the left ventricle just after LAD ligation to generate FOXC1 knockdown (A) or FOXC1 overexpression (B), & the negative control (NC) siRNA & Ad‐GFP served as control, respectively. The mRNA (upper panel) & protein (middle & lower panel) levels of TLR3/4 were determined after 2 wk. Data are means ± SEM aP < .05, AP < .01 vs respective MI Image collected & cropped by CiteAb from the following publication (https://pubmed.ncbi.nlm.nih.gov/31517441), licensed under a CC-BY license. Not internally tested by Novus Biologicals.Western Blot: TLR4 Antibody (76B357.1) - BSA Free [NB100-56566] -
Western Blot: TLR4 Antibody (76B357.1) - BSA Free [NB100-56566] - Up‐regulation of FOXC1 & TLR3/4 protein levels under myocardial ischaemia. Representative Western blot images & quantitative data for FOXC1, TLR3 & TLR4 proteins in ischaemia models of mice (A), H9c2 cells (B) & NRVMs (C) are shown herein. Validation data of the antibodies for TLR3 & TLR4 are shown in Figure S1. Data are means ± SEM. n = ~4‐5/group. P values from the one‐way ANOVAs: 0.018 (TLR3 protein in mice),.002 (TLR4 protein in mice) & 0.006 (FOXC1 protein in mice). aP < .05, AP < .01 vs control Image collected & cropped by CiteAb from the following publication (https://pubmed.ncbi.nlm.nih.gov/31517441), licensed under a CC-BY license. Not internally tested by Novus Biologicals.Western Blot: TLR4 Antibody (76B357.1) - BSA Free [NB100-56566] -
Western Blot: TLR4 Antibody (76B357.1) - BSA Free [NB100-56566] - Up‐regulation of FOXC1 & TLR3/4 protein levels under myocardial ischaemia. Representative Western blot images & quantitative data for FOXC1, TLR3 & TLR4 proteins in ischaemia models of mice (A), H9c2 cells (B) & NRVMs (C) are shown herein. Validation data of the antibodies for TLR3 & TLR4 are shown in Figure S1. Data are means ± SEM. n = ~4‐5/group. P values from the one‐way ANOVAs: 0.018 (TLR3 protein in mice),.002 (TLR4 protein in mice) & 0.006 (FOXC1 protein in mice). aP < .05, AP < .01 vs control Image collected & cropped by CiteAb from the following publication (https://pubmed.ncbi.nlm.nih.gov/31517441), licensed under a CC-BY license. Not internally tested by Novus Biologicals.Western Blot: TLR4 Antibody (76B357.1) - BSA Free [NB100-56566] -
Western Blot: TLR4 Antibody (76B357.1) - BSA Free [NB100-56566] - TLR4 mRNA (a1) & protein (a2) expression in MM cell lines. (b) TLR4 mRNA expression of 16 CD138+ selected MM patients as determined by PCR & agarose gel electrophoresis. Probing with beta -ACTIN was used as total protein loading reference, whereas beta -ACTIN gene expression was used as reference for RNA input. beta -ACTIN probing & beta -ACTIN mRNA expression were used as reference for total protein & mRNA input, respectively. Image collected & cropped by CiteAb from the following publication (https://pubmed.ncbi.nlm.nih.gov/30824741), licensed under a CC-BY license. Not internally tested by Novus Biologicals.Western Blot: TLR4 Antibody (76B357.1) - BSA Free [NB100-56566] -
Western Blot: TLR4 Antibody (76B357.1) - BSA Free [NB100-56566] - Regulation of FOXC1 on TLR expression. H9c2 cells were transfected with adenovirus or siRNA to overexpress or knock down FoxC1, with GFP adenovirus (Ad‐GFP) & negative control (NC) siRNA serving as control, respectively. A, FoxC1 overexpression increased Tlr3/4 mRNA & protein levels, under both control & ischaemic conditions. P values from the one‐way ANOVAs: <.001 (TLR3 mRNA),.002 (TLR4 mRNA), <.001 (FOXC1 mRNA), <.001 (TLR3 protein), <0.001 (TLR4 protein) & <.001 (FOXC1 protein). B, FoxC1 knockdown decreased Tlr3/4 mRNA & protein levels. P values from the one‐way ANOVAs:.001 (TLR3 mRNA), <.001 (TLR4 mRNA), <.001 (FOXC1 mRNA),.002 (TLR3 protein), <.001 (TLR4 protein) & <.001 (FOXC1 protein). C, FoxC1 overexpression up‐regulated the mRNA expression of multiple Tlr subtypes. P values from the one‐way ANOVAs:.022 (TLR1 mRNA),.047 (TLR2 mRNA),.002 (TLR3 mRNA),.013 (TLR4 mRNA),.039 (TLR5 mRNA),.003 (TLR6 mRNA),.203 (TLR7 mRNA),.078 (TLR8 mRNA) &.009 (TLR9 mRNA). Data are means ± SEM of 4 independent experiments. aP < .05, AP < .01 vs. control; bP < .05, BP < 0.01 vs. ischaemia Image collected & cropped by CiteAb from the following publication (https://pubmed.ncbi.nlm.nih.gov/31517441), licensed under a CC-BY license. Not internally tested by Novus Biologicals.Immunocytochemistry/ Immunofluorescence: TLR4 Antibody (76B357.1) - BSA Free [NB100-56566] -
Immunocytochemistry/ Immunofluorescence: TLR4 Antibody (76B357.1) - BSA Free [NB100-56566] - Immunofluorescent staining of TMAs. A) Low power (10x) view of NCI TMA slide stained for TLR4 (green), intestinal epithelium/pan-cytokeratin (red), & nucleus/DAPI (blue). B) Representative tissue cores from normal (I), adenomatous polyps (II), & CRC (III & IV) are shown. C & D) TLR4 staining score by tissue type & tissue compartment (stroma vs epithelium) are shown. C) TLR4 staining in the tumor stroma had a significantly higher average intensity score for stages 3 & 4 CRC when compared to stage 1. D) TLR4 staining in the tumor epithelium had a significantly higher average intensity score for stages 2 & 3 when compared to stage 1. E) TLR4 staining by compartment broken down by stage (controlling for grade) & grade (controlling for stage). Image collected & cropped by CiteAb from the following publication (https://jeccr.biomedcentral.com/articles/10.1186/1756-9966-33-45), licensed under a CC-BY license. Not internally tested by Novus Biologicals.Immunocytochemistry/ Immunofluorescence: TLR4 Antibody (76B357.1) - BSA Free [NB100-56566] -
Immunocytochemistry/ Immunofluorescence: TLR4 Antibody (76B357.1) - BSA Free [NB100-56566] - Immunofluorescent staining of TMAs. A) Low power (10x) view of NCI TMA slide stained for TLR4 (green), intestinal epithelium/pan-cytokeratin (red), & nucleus/DAPI (blue). B) Representative tissue cores from normal (I), adenomatous polyps (II), & CRC (III & IV) are shown. C & D) TLR4 staining score by tissue type & tissue compartment (stroma vs epithelium) are shown. C) TLR4 staining in the tumor stroma had a significantly higher average intensity score for stages 3 & 4 CRC when compared to stage 1. D) TLR4 staining in the tumor epithelium had a significantly higher average intensity score for stages 2 & 3 when compared to stage 1. E) TLR4 staining by compartment broken down by stage (controlling for grade) & grade (controlling for stage). Image collected & cropped by CiteAb from the following publication (https://jeccr.biomedcentral.com/articles/10.1186/1756-9966-33-45), licensed under a CC-BY license. Not internally tested by Novus Biologicals.Western Blot: TLR4 Antibody (76B357.1) - BSA Free [NB100-56566] -
Western Blot: TLR4 Antibody (76B357.1) - BSA Free [NB100-56566] - Effect of Cucumis sativus L. extract (CSE) on LPS induced TLR4 expression. a Expression of TLR4 mRNA in pAECs treated with LPS (10 μg/ml) for different time (1, 7, 24 h) in the presence or absence of increasing doses of CSE (0.02; 0.2; 2 mg/ml). mRNA expression of TLR4 is determined by quantitative PCR; relative expression was calculated as fold of change in respect to the control cells & error bar represents the range of relative expression. Different letters above the bars indicate significant differences. b Representative Western Blot of TLR4 & relative housekeeping alpha -tubulin were reported. c Expression of TLR4 protein in pAECs treated with LPS (10 μg/ml) in the presence or absence of increasing doses CSE (0.02; 0.2; 2 mg/ml); data shown are representative of at least three independent experiments & represent the mean ± SEM. Different letters above the bars indicate significant differences (p < 0.05 ANOVA post hoc Tukey’s test) Image collected & cropped by CiteAb from the following publication (https://bmccomplementalternmed.biomedcentral.com/articles/10.1186/s1290…), licensed under a CC-BY license. Not internally tested by Novus Biologicals.Western Blot: TLR4 Antibody (76B357.1) - BSA Free [NB100-56566] -
Western Blot: TLR4 Antibody (76B357.1) - BSA Free [NB100-56566] - Activation receptors are shed during intrinsic repair. (A) BMDM were unchallenged (control) or challenged with 500 HU/mL SLO WT, PFO or an equivalent mass of SLO ML for 10 min at 37 °C & centrifuged at 2000 × g for 5 min to yield cell pellet (C). Supernatants were spun at 100,000xg for 40 min at 4 °C to collect the high speed supernatant (S) & microvesicle pellet (M). Samples were solubilized at 95 °C in SDS-sample buffer, resolved by SDS-PAGE & transferred to nitrocellulose. Portions of the blot were probed with 6D11 anti-SLO, anti-IFN gamma R1, 4B4F12 anti-CD14, 76B357.1 anti-TLR4, O91B8 anti-MyD88, 1H4B01 anti-Trif, EPR4477 anti-Alkaline Phosphatase, MANLAC-4A7 anti-Lamin A/C, & AC-15 anti-beta -Actin antibodies followed by relevant secondary antibodies & ECL. Full-length blots are presented in Supplementary Figure S1. (B) BMDM were challenged with 500 HU/mL SLO WT or SLO N402C, SLO ML at equivalent mass to SLO WT, or SLO N402E at equivalent mass to SLO N402C for 10 min. Cell pellets (C), high speed supernatants (S) & microvesicles (M) were isolated as in (A) & probed with 6D11 anti-Streptolysin O, 76B357.1 anti-TLR4, O91B8 anti-MyD88, 1H4B01 anti-Trif, EPR4477 anti-Alkaline Phosphatase, MANLAC-4A7 anti-Lamin A/C, & AC-15 anti-beta -Actin antibodies. Full-length blots are presented in Supplementary Figure S2. The blots are representative of at least 3 independent experiments. Image collected & cropped by CiteAb from the following publication (https://www.nature.com/articles/s41598-018-24955-2), licensed under a CC-BY license. Not internally tested by Novus Biologicals.Immunocytochemistry/ Immunofluorescence: TLR4 Antibody (76B357.1) - BSA Free [NB100-56566] -
Immunocytochemistry/ Immunofluorescence: TLR4 Antibody (76B357.1) - BSA Free [NB100-56566] - Pericryptal Myofibroblasts are Responsible for Increased TLR4 Expression in a Subset of CRCs. A) CRCs were separated into two groups representing low- & high- stromal expression of TLR4 by IHC staining. In normal tissue, stromal TLR4 expression is mainly due to macrophages (Green: TLR4, Red: CD68, Merge: TLR4 + CD68 + DAPI (blue)). Conversely, in CRCs increased vimentin & decreased CD68 staining in the pericryptal space confirm that this signal was due to pericryptal myofibroblasts & not related to tumor-associated macrophages. B) Double-stained immunofluorescence for TLR4 (green) & vimentin (red) in normal (I), adenoma (II), & colon adenocarcinoma (III) (10×). In the stromal compartment of CRCs, immunofluorescent staining for TLR4 localized to the pericryptal myofibroblasts in a subset of samples. C) IHC staining of colon adenocarcinoma for TLR4, vimentin, & alpha -SMA (40×). Staining co-localizes to the pericryptal space, confirming the signal arises from pericryptal myofibroblasts. D, E, & F) An increase in IHC staining for alpha -SMA & vimentin was noted in CRCs when compared to normal or low grade dysplasia. A decrease in staining for CD68 positive macrophages was observed with higher degrees of dysplasia. Image collected & cropped by CiteAb from the following publication (https://jeccr.biomedcentral.com/articles/10.1186/1756-9966-33-45), licensed under a CC-BY license. Not internally tested by Novus Biologicals.Western Blot: TLR4 Antibody (76B357.1) - BSA Free [NB100-56566] -
Western Blot: TLR4 Antibody (76B357.1) - BSA Free [NB100-56566] - (a1) Q-PCR assay of TLR4 mRNA expression levels after transfecting JJN3 cell line with TLR4 RNAi oligonucleotides or a non-targeting pool (siCtrl) for 48 h. (a2) Representative immunoblotting analyses of protein samples probed with an antibody against TLR4 after TLR4 RNAi for 48 h. (b1) % cell viability & (b2) % proliferation of JJN3 cells after TLR4 RNAi for 48 h. (c,d1) Q-PCR expression analyses of TLR4 mRNA expression levels after transfecting H929 (c) & U266 (d1) cell lines with the pCMV6-TLR4 construct or a pCMV6 empty vector for 48 h. (d2) Immunobloting analyses of U266 cells transfected with pCMV6-TLR4 or pCMV6 vector for 48 h; protein samples were probed with an antibody against TLR4. (e1) % cell viability & (e2) % proliferation of H929 & U266 cells after transfection with the pCMV6-TLR4 construct or with the empty pCMV6 vector for 48 h. beta -ACTIN probing & beta -ACTIN mRNA expression were used as reference for total protein & mRNA input, respectively. Cells transfected with the pCMV6-TLR4 construct are labelled as TLR4 OE (TLR4 overexpression), whereas cells transfected with the pCMV6 empty vector are labelled as Con (control). Image collected & cropped by CiteAb from the following publication (https://pubmed.ncbi.nlm.nih.gov/30824741), licensed under a CC-BY license. Not internally tested by Novus Biologicals.Western Blot: TLR4 Antibody (76B357.1) - BSA Free [NB100-56566] -
Western Blot: TLR4 Antibody (76B357.1) - BSA Free [NB100-56566] - Regulation of FOXC1 on TLR expression. H9c2 cells were transfected with adenovirus or siRNA to overexpress or knock down FoxC1, with GFP adenovirus (Ad‐GFP) & negative control (NC) siRNA serving as control, respectively. A, FoxC1 overexpression increased Tlr3/4 mRNA & protein levels, under both control & ischaemic conditions. P values from the one‐way ANOVAs: <.001 (TLR3 mRNA),.002 (TLR4 mRNA), <.001 (FOXC1 mRNA), <.001 (TLR3 protein), <0.001 (TLR4 protein) & <.001 (FOXC1 protein). B, FoxC1 knockdown decreased Tlr3/4 mRNA & protein levels. P values from the one‐way ANOVAs:.001 (TLR3 mRNA), <.001 (TLR4 mRNA), <.001 (FOXC1 mRNA),.002 (TLR3 protein), <.001 (TLR4 protein) & <.001 (FOXC1 protein). C, FoxC1 overexpression up‐regulated the mRNA expression of multiple Tlr subtypes. P values from the one‐way ANOVAs:.022 (TLR1 mRNA),.047 (TLR2 mRNA),.002 (TLR3 mRNA),.013 (TLR4 mRNA),.039 (TLR5 mRNA),.003 (TLR6 mRNA),.203 (TLR7 mRNA),.078 (TLR8 mRNA) &.009 (TLR9 mRNA). Data are means ± SEM of 4 independent experiments. aP < .05, AP < .01 vs. control; bP < .05, BP < 0.01 vs. ischaemia Image collected & cropped by CiteAb from the following publication (https://pubmed.ncbi.nlm.nih.gov/31517441), licensed under a CC-BY license. Not internally tested by Novus Biologicals.Western Blot: TLR4 Antibody (76B357.1) - BSA Free [NB100-56566] -
Western Blot: TLR4 Antibody (76B357.1) - BSA Free [NB100-56566] - Dasatinib affects TLR4 signaling to regulate LPS-induced proinflammatory cytokine levels in BV2 microglial cells. a BV2 microglial cells were pretreated with TAK-242 (a TLR4 receptor inhibitor, 500 nM) or vehicle (1% DMSO) for 30 min, treated with dasatinib (250 nM) or vehicle (1% DMSO) for 30 min, & treated with LPS (1 μg/ml) or PBS for 5 h. The mRNA levels of COX-2 & IL-6 were analyzed by RT-PCR. b, c Quantification of the data in a (COX-2: con, n = 10; LPS, n = 10; dasatinib+LPS, n = 10; TAK-242+LPS, n = 10; TAK-242+dasatinib+LPS, n = 10; & IL-6: con, n = 14; LPS, n = 14; dasatinib+LPS, n = 14; TAK-242+LPS, n = 14; TAK-242+ dasatinib+LPS, n = 14). d BV2 microglial cells were treated with dasatinib (250 nM) or vehicle (1% DMSO) for 30 min followed by LPS (1 μg/ml) or PBS for 5.5 h, & cell surface biotinylation was conducted. e, f Quantification of the data in d (Surface TLR4: con, n = 11; LPS, n = 11; dasatinib+LPS, n = 11; Total TLR4: con, n = 16; LPS, n = 16; dasatinib+LPS, n = 16). One-way ANOVA with Tukey’s post hoc test was used to analyze significant differences. **p < 0.01, ***p < 0.001 Image collected & cropped by CiteAb from the following publication (https://pubmed.ncbi.nlm.nih.gov/31655606), licensed under a CC-BY license. Not internally tested by Novus Biologicals.Western Blot: TLR4 Antibody (76B357.1) - BSA Free [NB100-56566] -
Western Blot: TLR4 Antibody (76B357.1) - BSA Free [NB100-56566] - (a1) Q-PCR assay of TLR4 mRNA expression levels after transfecting JJN3 cell line with TLR4 RNAi oligonucleotides or a non-targeting pool (siCtrl) for 48 h. (a2) Representative immunoblotting analyses of protein samples probed with an antibody against TLR4 after TLR4 RNAi for 48 h. (b1) % cell viability & (b2) % proliferation of JJN3 cells after TLR4 RNAi for 48 h. (c,d1) Q-PCR expression analyses of TLR4 mRNA expression levels after transfecting H929 (c) & U266 (d1) cell lines with the pCMV6-TLR4 construct or a pCMV6 empty vector for 48 h. (d2) Immunobloting analyses of U266 cells transfected with pCMV6-TLR4 or pCMV6 vector for 48 h; protein samples were probed with an antibody against TLR4. (e1) % cell viability & (e2) % proliferation of H929 & U266 cells after transfection with the pCMV6-TLR4 construct or with the empty pCMV6 vector for 48 h. beta -ACTIN probing & beta -ACTIN mRNA expression were used as reference for total protein & mRNA input, respectively. Cells transfected with the pCMV6-TLR4 construct are labelled as TLR4 OE (TLR4 overexpression), whereas cells transfected with the pCMV6 empty vector are labelled as Con (control). Image collected & cropped by CiteAb from the following publication (https://pubmed.ncbi.nlm.nih.gov/30824741), licensed under a CC-BY license. Not internally tested by Novus Biologicals.Western Blot: TLR4 Antibody (76B357.1) - BSA Free [NB100-56566] -
Western Blot: TLR4 Antibody (76B357.1) - BSA Free [NB100-56566] - TLR4 expression in pVW-MSCs cultured with or without LPS (10 μg/ml) for 1 & 4 h & after additional 24 h of recovery after LPS removal (4hR). a: flow cytometry analysis were performed in not fixed & not permeabilized cells for TLR4 surface expression determination (TLR4: Surface) & in fixed & permeabilized cells to measure the overall TLR4 amount (TLR4: Total). Red histograms: stained cells; blue histograms: control cells. b: representative Western Blot of TLR4 & housekeeping beta -tubulin & relative quantification were presented. c: representative images of TLR4 immunostaining of pVW-MSCs cultured with or without LPS (10 μg/ml) for 4 h. pVW-MSCs nuclei were stained with Hoechst 33258 (blue). Scale bar = 10 μm. Data shown represent the mean ± SD of three biological replicates, each experiment is repeated three times. Data were analysed using one-way ANOVA followed by the Tukey’s post hoc comparison test. Different letters above the bars indicate significant differences (p < 0.05). (AU = Arbitrary Units) Image collected & cropped by CiteAb from the following publication (https://pubmed.ncbi.nlm.nih.gov/31029157), licensed under a CC-BY license. Not internally tested by Novus Biologicals.Flow Cytometry: TLR4 Antibody (76B357.1) - BSA Free [NB100-56566] -
Flow Cytometry: TLR4 Antibody (76B357.1) - BSA Free [NB100-56566] - TLR4 expression in pVW-MSCs cultured with or without LPS (10 μg/ml) for 1 & 4 h & after additional 24 h of recovery after LPS removal (4hR). a: flow cytometry analysis were performed in not fixed & not permeabilized cells for TLR4 surface expression determination (TLR4: Surface) & in fixed & permeabilized cells to measure the overall TLR4 amount (TLR4: Total). Red histograms: stained cells; blue histograms: control cells. b: representative Western Blot of TLR4 & housekeeping beta -tubulin & relative quantification were presented. c: representative images of TLR4 immunostaining of pVW-MSCs cultured with or without LPS (10 μg/ml) for 4 h. pVW-MSCs nuclei were stained with Hoechst 33258 (blue). Scale bar = 10 μm. Data shown represent the mean ± SD of three biological replicates, each experiment is repeated three times. Data were analysed using one-way ANOVA followed by the Tukey’s post hoc comparison test. Different letters above the bars indicate significant differences (p < 0.05). (AU = Arbitrary Units) Image collected & cropped by CiteAb from the following publication (https://pubmed.ncbi.nlm.nih.gov/31029157), licensed under a CC-BY license. Not internally tested by Novus Biologicals.Western Blot: TLR4 Antibody (76B357.1) - BSA Free [NB100-56566] -
Western Blot: TLR4 Antibody (76B357.1) - BSA Free [NB100-56566] - Toll‐like receptor 4 (TLR4)‐shRNA lentivirus reduced myocardial inflammation & improved heart function after myocardial infarction (MI). The rats received intra‐myocardial injection of normal saline (NS), control‐shRNA lentivirus or TLR4‐shRNA lentivirus (1 × 109 TU/ml, 100 μl/heart) just after left anterior descending coronary artery (LAD) ligation or sham operation. All examinations were performed after 4 weeks of MI. (A) Expression of green fluorescent protein (GFP; green), the marker gene carried by TLR4‐shRNA lentivirus, in the myocardium. The nuclei were counter‐stained with Hoechst 33258 (blue). (B) Representative Western blot images & quantification of TLR4 proteins in sham & chronic heart failure (CHF) myocardium. (C) tumour necrosis factor (TNF)‐ alpha & interleukin (IL)‐6 protein contents in infarct & remote myocardium. (D) Representative images of Masson's trichrome staining (upper panel) & quantification (lower panel) of post‐infarct failing hearts, showing that TLR4‐shRNA lentivirus reduced cardiac fibrosis. Cross‐sections were cut at the midhorizontal plane of the fixed paraffin‐embedded heart, & stained with Masson's trichrome reagents. (E) Infarct size of post‐infarct failing hearts. (F) Fractional shortening (%) of the left ventricle (data are means ± SD, n = 4/group, aP < 0.05, AP < 0.01 versus respective sham‐NS; BP < 0.01 versus respective CHF‐NS). Image collected & cropped by CiteAb from the following publication (https://pubmed.ncbi.nlm.nih.gov/26290459), licensed under a CC-BY license. Not internally tested by Novus Biologicals.Western Blot: TLR4 Antibody (76B357.1) - BSA Free [NB100-56566] -
Western Blot: TLR4 Antibody (76B357.1) - BSA Free [NB100-56566] - ALWPs significantly reduced LPS-induced cell-surface levels of TLR4. (A) BV2 microglial cells were pretreated with TAK-242 (a TLR4 inhibitor, 500 nM) for 30 min, followed by treatment with ALWPs (500 μg/ml) or PBS for 30 min & finally LPS (1 μg/ml) or PBS for 5 h. Total RNA was isolated, & IL-1 beta mRNA levels were measured by RT-PCR. (B) Quantification of the data from (A) (con, n = 20; LPS, n = 20; ALWPs + LPS, n = 20; TAK-242 + LPS, n = 20; TAK-242 + ALWPs + LPS, n = 20). (C) BV2 microglial cells were pretreated with ALWPs (500 μg/ml) or PBS for 30 min, followed by treatment with LPS (1 μg/ml) or PBS for 5.5 h. Cell-surface biotinylation was then conducted with a TLR4 antibody recognizing the N-terminal region of TLR4. (D) Quantification of the data from (C) (Surface TLR4: con, n = 10; LPS, n = 10; ALWPs + LPS, n = 10; Total TLR4: con, n = 4; LPS, n = 4; ALWPs + LPS, n = 4). ∗p < 0.05, ∗∗p < 0.01, ∗∗∗p < 0.001. Image collected & cropped by CiteAb from the following publication (https://pubmed.ncbi.nlm.nih.gov/30319390), licensed under a CC-BY license. Not internally tested by Novus Biologicals.Immunocytochemistry/ Immunofluorescence: TLR4 Antibody (76B357.1) - BSA Free [NB100-56566] -
Immunocytochemistry/ Immunofluorescence: TLR4 Antibody (76B357.1) - BSA Free [NB100-56566] - Pericryptal Myofibroblasts are Responsible for Increased TLR4 Expression in a Subset of CRCs. A) CRCs were separated into two groups representing low- & high- stromal expression of TLR4 by IHC staining. In normal tissue, stromal TLR4 expression is mainly due to macrophages (Green: TLR4, Red: CD68, Merge: TLR4 + CD68 + DAPI (blue)). Conversely, in CRCs increased vimentin & decreased CD68 staining in the pericryptal space confirm that this signal was due to pericryptal myofibroblasts & not related to tumor-associated macrophages. B) Double-stained immunofluorescence for TLR4 (green) & vimentin (red) in normal (I), adenoma (II), & colon adenocarcinoma (III) (10×). In the stromal compartment of CRCs, immunofluorescent staining for TLR4 localized to the pericryptal myofibroblasts in a subset of samples. C) IHC staining of colon adenocarcinoma for TLR4, vimentin, & alpha -SMA (40×). Staining co-localizes to the pericryptal space, confirming the signal arises from pericryptal myofibroblasts. D, E, & F) An increase in IHC staining for alpha -SMA & vimentin was noted in CRCs when compared to normal or low grade dysplasia. A decrease in staining for CD68 positive macrophages was observed with higher degrees of dysplasia. Image collected & cropped by CiteAb from the following publication (https://jeccr.biomedcentral.com/articles/10.1186/1756-9966-33-45), licensed under a CC-BY license. Not internally tested by Novus Biologicals.Applications for TLR4 Antibody (76B357.1) - BSA Free
Chromatin Immunoprecipitation (ChIP)
ELISA Capture (Matched Antibody Pair)
Immunocytochemistry/ Immunofluorescence
Immunohistochemistry
Immunohistochemistry-Frozen
Immunohistochemistry-Paraffin
SDS-Page
Western Blot
Reviewed Applications
Read 5 reviews rated 4.2 using NB100-56566 in the following applications:
Flow Cytometry Panel Builder
Bio-Techne Knows Flow Cytometry
Save time and reduce costly mistakes by quickly finding compatible reagents using the Panel Builder Tool.
Advanced Features
- Spectra Viewer - Custom analysis of spectra from multiple fluorochromes
- Spillover Popups - Visualize the spectra of individual fluorochromes
- Antigen Density Selector - Match fluorochrome brightness with antigen density
Formulation, Preparation, and Storage
Purification
Formulation
Format
Preservative
Concentration
Shipping
Stability & Storage
Background: TLR4
TLR4 signaling occurs through two distinct pathways: The MyD88 (myeloid differentiation primary response gene 88)-dependent pathway and the MyD88-independent (TRIF-dependent, TIR domain-containing adaptor inducing IFN-beta) pathway (3, 5-7). The MyD88-dependent pathway occurs mainly at the plasma membrane and involves the binding of MyD88-adaptor-like (MAL) protein followed by a signaling cascade that results in the activation of transcription factors including nuclear factor-kappaB (NF-kappaB) that promote the secretion of inflammatory molecules and increased phagocytosis (5-7). Conversely, the MyD88-independent pathway occurs after TLR4-MD2 complex internalization in the endosomal compartment. This pathway involves the binding of adapter proteins TRIF and TRIF-related adaptor molecule (TRAM), a signaling activation cascade resulting in IFN regulatory factor 3 (IRF3) translocation into the nucleus, and secretion of interferon-beta (INF-beta) genes and increased phagocytosis (5-7).
Given its expression on immune-related cells and its role in inflammation, TLR4 activation can contribute to various diseases (6-8). For instance, several studies have found that TLR4 activation is associated with neurodegeneration and several central nervous system (CNS) pathologies, including Alzheimer's disease, Parkinson's disease, and Huntington's disease (6, 7). Furthermore, TLR4 mutations have been shown to lead to higher rates of infections and increased susceptibility to sepsis (7-8). One potential therapeutic approach aimed at targeting TLR4 and neuroinflammation is polyphenolic compounds which include flavonoids and phenolic acids and alcohols (8).
Alternative names for TLR4 includes 76B357.1, ARMD10, CD284 antigen, CD284, EC 3.2.2.6, homolog of Drosophila toll, hToll, toll like receptor 4 protein, TOLL, toll-like receptor 4.
References
1. Vaure, C., & Liu, Y. (2014). A comparative review of toll-like receptor 4 expression and functionality in different animal species. Frontiers in immunology. https://doi.org/10.3389/fimmu.2014.00316
2. Park, B. S., & Lee, J. O. (2013). Recognition of lipopolysaccharide pattern by TLR4 complexes. Experimental & molecular medicine. https://doi.org/10.1038/emm.2013.97
3. Krishnan, J., Anwar, M.A., & Choi, S. (2016) TLR4 (Toll-Like Receptor 4). In: Choi S. (eds) Encyclopedia of Signaling Molecules. Springer, New York, NY. https://doi.org/10.1007/978-1-4614-6438-9_592-1
4. Botos, I., Segal, D. M., & Davies, D. R. (2011). The structural biology of Toll-like receptors. Structure. https://doi.org/10.1016/j.str.2011.02.004
5. Lu, Y. C., Yeh, W. C., & Ohashi, P. S. (2008). LPS/TLR4 signal transduction pathway. Cytokine. https://doi.org/10.1016/j.cyto.2008.01.006
6. Leitner, G. R., Wenzel, T. J., Marshall, N., Gates, E. J., & Klegeris, A. (2019). Targeting toll-like receptor 4 to modulate neuroinflammation in central nervous system disorders. Expert opinion on therapeutic targets. https://doi.org/10.1080/14728222.2019.1676416
7. Molteni, M., Gemma, S., & Rossetti, C. (2016). The Role of Toll-Like Receptor 4 in Infectious and Noninfectious Inflammation. Mediators of inflammation. https://doi.org/10.1155/2016/6978936
8. Rahimifard, M., Maqbool, F., Moeini-Nodeh, S., Niaz, K., Abdollahi, M., Braidy, N., Nabavi, S. M., & Nabavi, S. F. (2017). Targeting the TLR4 signaling pathway by polyphenols: A novel therapeutic strategy for neuroinflammation. Ageing research reviews. https://doi.org/10.1016/j.arr.2017.02.004
Long Name
Alternate Names
Gene Symbol
UniProt
Additional TLR4 Products
Product Documents for TLR4 Antibody (76B357.1) - BSA Free
Certificate of Analysis
To download a Certificate of Analysis, please enter a lot or batch number in the search box below.
Product Specific Notices for TLR4 Antibody (76B357.1) - BSA Free
This product is for research use only and is not approved for use in humans or in clinical diagnosis. Primary Antibodies are guaranteed for 1 year from date of receipt.
Citations for TLR4 Antibody (76B357.1) - BSA Free
Customer Reviews for TLR4 Antibody (76B357.1) - BSA Free (5)
Have you used TLR4 Antibody (76B357.1) - BSA Free?
Submit a review and receive an Amazon gift card!
$25/€18/£15/$25CAN/¥2500 Yen for a review with an image
$10/€7/£6/$10CAN/¥1110 Yen for a review without an image
Submit a review
Customer Images
-
Application: IHC -FrozenSample Tested: brain tissue slice(30umSpecies: MouseVerified Customer | Posted 05/25/2017This image is TLR4(green) and nucleus(blue) at area postrema of the adult male mouse brai n, x20 magnification.IHC-frozen, 1:500
-
Application: ImmunohistochemistrySample Tested: Mouse brain, Adult mouse brain tissue and Mouse cortexSpecies: MouseVerified Customer | Posted 05/16/2017IHC image showing TLR4 expression in mice cerebral cortex. Dilution 1:4000.
-
Application: Western BlotSample Tested: Pig whole ovarian lysateSpecies: OtherVerified Customer | Posted 05/12/2016TLR4 detection on porcine whole ovarian homogenate
-
Application: Western BlotSample Tested:Species: MouseVerified Customer | Posted 08/07/2014A review for TLR4 antibody (NB100-56566)
-
Application: Western BlotSample Tested: small intestinal lysate, Sample Amount: 50 ugSpecies: OtherVerified Customer | Posted 08/11/2011
There are no reviews that match your criteria.
Protocols
View specific protocols for TLR4 Antibody (76B357.1) - BSA Free (NB100-56566):
Protocol for Flow Cytometry Intracellular Staining
Sample Preparation.
1. Grow cells to 60-85% confluency. Flow cytometry requires between 2 x 105 and 1 x 106 cells for optimal performance.
2. If cells are adherent, harvest gently by washing once with staining buffer and then scraping. Avoid using trypsin as this can disrupt certain epitopes of interest. If enzymatic harvest is required, use Accutase, Collagenase, or TrypLE Express for a less damaging option.
3. Reserve 100 uL for counting, then transfer cell volume into a 50 mL conical tube and centrifuge for 8 minutes at 400 RCF.
a. Count cells using a hemocytometer and a 1:1 trypan blue exclusion stain to determine cell viability before starting the flow protocol. If cells appear blue, do not proceed.
4. Re-suspend cells to a concentration of 1 x 106 cells/mL in staining buffer (NBP2-26247).
5. Aliquot out 100 uL samples in accordance with your experimental samples.
Tip: When cell surface and intracellular staining are required in the same sample, it is advisable that the cell surface staining be performed first since the fixation and permeablization steps might reduce the availability of surface antigens.
Intracellular Staining.
Tip: When performing intracellular staining, it is important to use appropriate fixation and permeabilization reagents based upon the target and its subcellular location. Generally, our Intracellular Flow Assay Kit (NBP2-29450) is a good place to start as it contains an optimized combination of reagents for intracellular staining as well as an inhibitor of intracellular protein transport (necessary if staining secreted proteins). Certain targets may require more gentle or transient permeabilization protocols such as the commonly employed methanol or saponin-based methods.
Protocol for Cytoplasmic Targets:
1. Fix the cells by adding 100 uL fixation solution (such as 4% PFA) to each sample for 10-15 minutes.
2. Permeabilize cells by adding 100 uL of a permeabization buffer to every 1 x 106 cells present in the sample. Mix well and incubate at room temperature for 15 minutes.
a. For cytoplasmic targets, use a gentle permeabilization solution such as 1X PBS + 0.5% Saponin or 1X PBS + 0.5% Tween-20.
b. To maintain the permeabilized state throughout your experiment, use staining buffer + 0.1% of the permeabilization reagent (i.e. 0.1% Tween-20 or 0.1% Saponin).
3. Following the 15 minute incubation, add 2 mL of the staining buffer + 0.1% permeabilizer to each sample.
4. Centrifuge for 1 minute at 400 RCF.
5. Discard supernatant and re-suspend in 100 uL of staining buffer + 0.1% permeabilizer.
6. Add appropriate amount of each antibody (eg. 1 test or 1 ug per sample, as experimentally determined).
7. Mix well and incubate at room temperature for 30 minutes- 1 hour. Gently mix samples every 10-15 minutes.
8. Following the primary/conjugate incubation, add 1-2 mL/sample of staining buffer +0.1% permeabilizer and centrifuge for 1 minute at 400 RCF.
9. Wash twice by re-suspending cells in staining buffer (2 mL for tubes or 200 uL for wells) and centrifuging at 400 RCF for 5 minutes. Discard supernatant.
10. Add appropriate amount of secondary antibody (as experimentally determined) to each sample.
11. Incubate at room temperature in dark for 20 minutes.
12. Add 1-2 mL of staining buffer and centrifuge at 400 RCF for 1 minute and discard supernatant.
13. Wash twice by re-suspending cells in staining buffer (2 mL for tubes or 200 uL for wells) and centrifuging at 400 RCF for 5 minutes. Discard supernatant.
14. Resuspend in an appropriate volume of staining buffer (usually 500 uL per sample) and proceed with analysis on your flow cytometer.
Culture cells to appropriate density in 35 mm culture dishes or 6-well plates.
1. Remove culture medium and wash the cells briefly in PBS. Add 10% formalin to the dish and fix at room temperature for 10 minutes.
2. Remove the formalin and wash the cells in PBS.
3. Permeablize the cells with 0.1% Triton X100 or other suitable detergent for 10 min.
4. Remove the permeablization buffer and wash three times for 10 minutes each in PBS. Be sure to not let the specimen dry out.
5. To block nonspecific antibody binding, incubate in 10% normal goat serum from 1 hour to overnight at room temperature.
6. Add primary antibody at appropriate dilution and incubate overnight at 4C.
7. Remove primary antibody and replace with PBS. Wash three times for 10 minutes each.
8. Add secondary antibody at appropriate dilution. Incubate for 1 hour at room temperature.
9. Remove secondary antibody and replace with PBS. Wash three times for 10 minutes each.
10. Counter stain DNA with DAPi if required.
Antigen Unmasking:
Bring slides to a boil in 10 mM sodium citrate buffer (pH 6.0) then maintain at a sub-boiling temperature for 10 minutes. Cool slides on bench-top for 30 minutes (keep slides in the sodium citrate buffer all the time).
Staining:
1. Wash sections in deionized water three times for 5 minutes each.
2. Wash sections in PBS for 5 minutes.
3. Block each section with 100-400 ul blocking solution (1% BSA in PBS) for 1 hour at room temperature.
4. Remove blocking solution and add 100-400 ul diluted primary antibody. Incubate overnight at 4 C.
5. Remove antibody solution and wash sections in wash buffer three times for 5 minutes each.
6. Add 100-400 ul HRP polymer conjugated secondary antibody. Incubate 30 minutes at room temperature.
7. Wash sections three times in wash buffer for 5 minutes each.
8. Add 100-400 ul DAB substrate to each section and monitor staining closely.
9. As soon as the sections develop, immerse slides in deionized water.
10. Counterstain sections in hematoxylin.
11. Wash sections in deionized water two times for 5 minutes each.
12. Dehydrate sections.
13. Mount coverslips.
Western Blot Protocol
1. Perform SDS-PAGE on samples to be analyzed, loading 10-25 ug of total protein per lane.
2. Transfer proteins to PVDF membrane according to the instructions provided by the manufacturer of the membrane and transfer apparatus.
3. Stain the membrane with Ponceau S (or similar product) to assess transfer success, and mark molecular weight standards where appropriate.
4. Rinse the blot TBS -0.05% Tween 20 (TBST).
5. Block the membrane in 5% Non-fat milk in TBST (blocking buffer) for at least 1 hour.
6. Wash the membrane in TBST three times for 10 minutes each.
7. Dilute anti-TLR4 primary antibody in blocking buffer and incubate overnight at 4C with gentle rocking.
8. Wash the membrane in TBST three times for 10 minutes each.
9. Incubate the membrane in diluted HRP conjugated secondary antibody in blocking buffer (as per manufacturer's instructions) for 1 hour at room temperature.
10. Wash the blot in TBST three times for 10 minutes each (this step can be repeated as required to reduce background).
11. Apply the detection reagent of choice in accordance with the manufacturers instructions.
Find general support by application which include: protocols, troubleshooting, illustrated assays, videos and webinars.
- 7-Amino Actinomycin D (7-AAD) Cell Viability Flow Cytometry Protocol
- Antigen Retrieval Protocol (PIER)
- Antigen Retrieval for Frozen Sections Protocol
- Appropriate Fixation of IHC/ICC Samples
- Cellular Response to Hypoxia Protocols
- ChIP Protocol Video
- Chromatin Immunoprecipitation (ChIP) Protocol
- Chromatin Immunoprecipitation Protocol
- Chromogenic IHC Staining of Formalin-Fixed Paraffin-Embedded (FFPE) Tissue Protocol
- Chromogenic Immunohistochemistry Staining of Frozen Tissue
- ClariTSA™ Fluorophore Kits
- Detection & Visualization of Antibody Binding
- ELISA Sample Preparation & Collection Guide
- ELISA Troubleshooting Guide
- Extracellular Membrane Flow Cytometry Protocol
- Flow Cytometry Protocol for Cell Surface Markers
- Flow Cytometry Protocol for Staining Membrane Associated Proteins
- Flow Cytometry Staining Protocols
- Flow Cytometry Troubleshooting Guide
- Fluorescent IHC Staining of Frozen Tissue Protocol
- Graphic Protocol for Heat-induced Epitope Retrieval
- Graphic Protocol for the Preparation and Fluorescent IHC Staining of Frozen Tissue Sections
- Graphic Protocol for the Preparation and Fluorescent IHC Staining of Paraffin-embedded Tissue Sections
- Graphic Protocol for the Preparation of Gelatin-coated Slides for Histological Tissue Sections
- How to Run an R&D Systems DuoSet ELISA
- How to Run an R&D Systems Quantikine ELISA
- How to Run an R&D Systems Quantikine™ QuicKit™ ELISA
- ICC Cell Smear Protocol for Suspension Cells
- ICC Immunocytochemistry Protocol Videos
- ICC for Adherent Cells
- IHC Sample Preparation (Frozen sections vs Paraffin)
- ISH-IHC Protocol for Chromogenic Detection on Formalin Fixed Paraffin Embedded (FFPE) Tissue
- Immunocytochemistry (ICC) Protocol
- Immunocytochemistry Troubleshooting
- Immunofluorescence of Organoids Embedded in Cultrex Basement Membrane Extract
- Immunofluorescent IHC Staining of Formalin-Fixed Paraffin-Embedded (FFPE) Tissue Protocol
- Immunohistochemistry (IHC) and Immunocytochemistry (ICC) Protocols
- Immunohistochemistry Frozen Troubleshooting
- Immunohistochemistry Paraffin Troubleshooting
- Intracellular Flow Cytometry Protocol Using Alcohol (Methanol)
- Intracellular Flow Cytometry Protocol Using Detergents
- Intracellular Nuclear Staining Flow Cytometry Protocol Using Detergents
- Intracellular Staining Flow Cytometry Protocol Using Alcohol Permeabilization
- Intracellular Staining Flow Cytometry Protocol Using Detergents to Permeabilize Cells
- Preparing Samples for IHC/ICC Experiments
- Preventing Non-Specific Staining (Non-Specific Binding)
- Primary Antibody Selection & Optimization
- Propidium Iodide Cell Viability Flow Cytometry Protocol
- Protocol for Heat-Induced Epitope Retrieval (HIER)
- Protocol for Liperfluo
- Protocol for Making a 4% Formaldehyde Solution in PBS
- Protocol for VisUCyte™ HRP Polymer Detection Reagent
- Protocol for the Characterization of Human Th22 Cells
- Protocol for the Characterization of Human Th9 Cells
- Protocol for the Fluorescent ICC Staining of Cell Smears - Graphic
- Protocol for the Fluorescent ICC Staining of Cultured Cells on Coverslips - Graphic
- Protocol for the Preparation & Fixation of Cells on Coverslips
- Protocol for the Preparation and Chromogenic IHC Staining of Frozen Tissue Sections
- Protocol for the Preparation and Chromogenic IHC Staining of Frozen Tissue Sections - Graphic
- Protocol for the Preparation and Chromogenic IHC Staining of Paraffin-embedded Tissue Sections
- Protocol for the Preparation and Chromogenic IHC Staining of Paraffin-embedded Tissue Sections - Graphic
- Protocol for the Preparation and Fluorescent ICC Staining of Cells on Coverslips
- Protocol for the Preparation and Fluorescent ICC Staining of Non-adherent Cells
- Protocol for the Preparation and Fluorescent ICC Staining of Stem Cells on Coverslips
- Protocol for the Preparation and Fluorescent IHC Staining of Frozen Tissue Sections
- Protocol for the Preparation and Fluorescent IHC Staining of Paraffin-embedded Tissue Sections
- Protocol for the Preparation of Gelatin-coated Slides for Histological Tissue Sections
- Protocol for the Preparation of a Cell Smear for Non-adherent Cell ICC - Graphic
- Protocol: Annexin V and PI Staining by Flow Cytometry
- Protocol: Annexin V and PI Staining for Apoptosis by Flow Cytometry
- Quantikine HS ELISA Kit Assay Principle, Alkaline Phosphatase
- Quantikine HS ELISA Kit Principle, Streptavidin-HRP Polymer
- R&D Systems Quality Control Western Blot Protocol
- Sandwich ELISA (Colorimetric) – Biotin/Streptavidin Detection Protocol
- Sandwich ELISA (Colorimetric) – Direct Detection Protocol
- TUNEL and Active Caspase-3 Detection by IHC/ICC Protocol
- The Importance of IHC/ICC Controls
- Troubleshooting Guide: ELISA
- Troubleshooting Guide: Fluorokine Flow Cytometry Kits
- Troubleshooting Guide: Immunohistochemistry
- Troubleshooting Guide: Western Blot Figures
- Western Blot Conditions
- Western Blot Protocol
- Western Blot Protocol for Cell Lysates
- Western Blot Troubleshooting
- Western Blot Troubleshooting Guide
- View all Protocols, Troubleshooting, Illustrated assays and Webinars
FAQs for TLR4 Antibody (76B357.1) - BSA Free
-
Q: Do you know the amino acid sequence against which this antibody is directed to? Also, what isoform of the protein would it be recognizing, or from what specific TLR4 transcript?
A:
This antibody binds within amino acids 100-200 of TLR4. This antibody will recognize isoforms 1 and 2 of TLR4 as this region is present in both of these isoforms, but not isoform 3, according to UniProt O00206.
-
Q: How do I choose secondary antibodies to label the same cells when I have two primary antibodies from the same host?
A: Use isotype-specific secondary antibodies if the primary antibodies are of different isotypes. You can also make direct conjugates of the primary antibodies by use of antibody labeling kits, dyes, or custom conjugations (please contact Technical Support for custom orders).
-
Q: I want to check the TLR4 expression on Swine embryonic fibroblast cells. Is it applicable to use this human TLR4 antibody? Have you obtained any results on swine cells using this TLR4 antibody.
A:
This antibody has not been used on swine however I have carried out a blast and the % homology is 73% between the human protein and the pig protein. If you would be interested in testing this novel species, please take a look at our Innovators Reward Program.
-
Q: I would like to use this antibody but it has not been validated in my species of interest. Is there any way I can find out if it will work?
A: We offer risk-free testing of all of our primary antibodies. Please check out our Innovator's Reward Program and test this TLR4 antibody in any unvalidated species or application, without the financial risk of failure.
-
Q:
Would you be able to share the sequence homology/alignment of the immunogen sequence of NB100-56566 to the Zebrafish protein (Uniprot S5Q1H4)?
A: We ran the sequence you provided against the immunogen, and unfortunately, there is only a 28% homology.
-
Q: Do you know the amino acid sequence against which this antibody is directed to? Also, what isoform of the protein would it be recognizing, or from what specific TLR4 transcript?
A:
This antibody binds within amino acids 100-200 of TLR4. This antibody will recognize isoforms 1 and 2 of TLR4 as this region is present in both of these isoforms, but not isoform 3, according to UniProt O00206.
-
Q: How do I choose secondary antibodies to label the same cells when I have two primary antibodies from the same host?
A: Use isotype-specific secondary antibodies if the primary antibodies are of different isotypes. You can also make direct conjugates of the primary antibodies by use of antibody labeling kits, dyes, or custom conjugations (please contact Technical Support for custom orders).
-
Q: I want to check the TLR4 expression on Swine embryonic fibroblast cells. Is it applicable to use this human TLR4 antibody? Have you obtained any results on swine cells using this TLR4 antibody.
A:
This antibody has not been used on swine however I have carried out a blast and the % homology is 73% between the human protein and the pig protein. If you would be interested in testing this novel species, please take a look at our Innovators Reward Program.
-
Q: I would like to use this antibody but it has not been validated in my species of interest. Is there any way I can find out if it will work?
A: We offer risk-free testing of all of our primary antibodies. Please check out our Innovator's Reward Program and test this TLR4 antibody in any unvalidated species or application, without the financial risk of failure.
-
Q:
Would you be able to share the sequence homology/alignment of the immunogen sequence of NB100-56566 to the Zebrafish protein (Uniprot S5Q1H4)?
A: We ran the sequence you provided against the immunogen, and unfortunately, there is only a 28% homology.
-
Q: Do you know the amino acid sequence against which this antibody is directed to? Also, what isoform of the protein would it be recognizing, or from what specific TLR4 transcript?
A:
This antibody binds within amino acids 100-200 of TLR4. This antibody will recognize isoforms 1 and 2 of TLR4 as this region is present in both of these isoforms, but not isoform 3, according to UniProt O00206.
-
Q: How do I choose secondary antibodies to label the same cells when I have two primary antibodies from the same host?
A: Use isotype-specific secondary antibodies if the primary antibodies are of different isotypes. You can also make direct conjugates of the primary antibodies by use of antibody labeling kits, dyes, or custom conjugations (please contact Technical Support for custom orders).
-
Q: I want to check the TLR4 expression on Swine embryonic fibroblast cells. Is it applicable to use this human TLR4 antibody? Have you obtained any results on swine cells using this TLR4 antibody.
A:
This antibody has not been used on swine however I have carried out a blast and the % homology is 73% between the human protein and the pig protein. If you would be interested in testing this novel species, please take a look at our Innovators Reward Program.
-
Q: I would like to use this antibody but it has not been validated in my species of interest. Is there any way I can find out if it will work?
A: We offer risk-free testing of all of our primary antibodies. Please check out our Innovator's Reward Program and test this TLR4 antibody in any unvalidated species or application, without the financial risk of failure.
-
Q:
Would you be able to share the sequence homology/alignment of the immunogen sequence of NB100-56566 to the Zebrafish protein (Uniprot S5Q1H4)?
A: We ran the sequence you provided against the immunogen, and unfortunately, there is only a 28% homology.
-
Q: Do you know the amino acid sequence against which this antibody is directed to? Also, what isoform of the protein would it be recognizing, or from what specific TLR4 transcript?
A:
This antibody binds within amino acids 100-200 of TLR4. This antibody will recognize isoforms 1 and 2 of TLR4 as this region is present in both of these isoforms, but not isoform 3, according to UniProt O00206.
-
Q: How do I choose secondary antibodies to label the same cells when I have two primary antibodies from the same host?
A: Use isotype-specific secondary antibodies if the primary antibodies are of different isotypes. You can also make direct conjugates of the primary antibodies by use of antibody labeling kits, dyes, or custom conjugations (please contact Technical Support for custom orders).
-
Q: I want to check the TLR4 expression on Swine embryonic fibroblast cells. Is it applicable to use this human TLR4 antibody? Have you obtained any results on swine cells using this TLR4 antibody.
A:
This antibody has not been used on swine however I have carried out a blast and the % homology is 73% between the human protein and the pig protein. If you would be interested in testing this novel species, please take a look at our Innovators Reward Program.
-
Q: I would like to use this antibody but it has not been validated in my species of interest. Is there any way I can find out if it will work?
A: We offer risk-free testing of all of our primary antibodies. Please check out our Innovator's Reward Program and test this TLR4 antibody in any unvalidated species or application, without the financial risk of failure.
-
Q:
Would you be able to share the sequence homology/alignment of the immunogen sequence of NB100-56566 to the Zebrafish protein (Uniprot S5Q1H4)?
A: We ran the sequence you provided against the immunogen, and unfortunately, there is only a 28% homology.
-
Q: Do you know the amino acid sequence against which this antibody is directed to? Also, what isoform of the protein would it be recognizing, or from what specific TLR4 transcript?
A:
This antibody binds within amino acids 100-200 of TLR4. This antibody will recognize isoforms 1 and 2 of TLR4 as this region is present in both of these isoforms, but not isoform 3, according to UniProt O00206.
-
Q: How do I choose secondary antibodies to label the same cells when I have two primary antibodies from the same host?
A: Use isotype-specific secondary antibodies if the primary antibodies are of different isotypes. You can also make direct conjugates of the primary antibodies by use of antibody labeling kits, dyes, or custom conjugations (please contact Technical Support for custom orders).
-
Q: I want to check the TLR4 expression on Swine embryonic fibroblast cells. Is it applicable to use this human TLR4 antibody? Have you obtained any results on swine cells using this TLR4 antibody.
A:
This antibody has not been used on swine however I have carried out a blast and the % homology is 73% between the human protein and the pig protein. If you would be interested in testing this novel species, please take a look at our Innovators Reward Program.
-
Q: I would like to use this antibody but it has not been validated in my species of interest. Is there any way I can find out if it will work?
A: We offer risk-free testing of all of our primary antibodies. Please check out our Innovator's Reward Program and test this TLR4 antibody in any unvalidated species or application, without the financial risk of failure.
-
Q:
Would you be able to share the sequence homology/alignment of the immunogen sequence of NB100-56566 to the Zebrafish protein (Uniprot S5Q1H4)?
A: We ran the sequence you provided against the immunogen, and unfortunately, there is only a 28% homology.

![Flow Cytometry: TLR4 Antibody (76B357.1) - BSA Free [NB100-56566] Flow Cytometry: TLR4 Antibody (76B357.1) - BSA Free [NB100-56566]](https://resources.rndsystems.com/images/products/TLR4-Antibody-76B357-1-Flow-Cytometry-NB100-56566-img0024.jpg)
![Immunohistochemistry: TLR4 Antibody (76B357.1) - BSA Free [NB100-56566] Immunohistochemistry: TLR4 Antibody (76B357.1) - BSA Free [NB100-56566]](https://resources.rndsystems.com/images/products/TLR4-Antibody-76B357-1-Immunohistochemistry-NB100-56566-img0040.jpg)
![Western Blot: TLR4 Antibody (76B357.1)BSA Free [NB100-56566] Western Blot: TLR4 Antibody (76B357.1)BSA Free [NB100-56566]](https://resources.rndsystems.com/images/products/TLR4-Antibody-76B357-1-Western-Blot-NB100-56566-img0018.jpg)
![Immunohistochemistry: TLR4 Antibody (76B357.1) - BSA Free [NB100-56566] Immunohistochemistry: TLR4 Antibody (76B357.1) - BSA Free [NB100-56566]](https://resources.rndsystems.com/images/products/TLR4-Antibody-76B357-1-Immunohistochemistry-NB100-56566-img0041.jpg)
![Flow (Intracellular): TLR4 Antibody (76B357.1) - BSA Free [NB100-56566] Flow (Intracellular): TLR4 Antibody (76B357.1) - BSA Free [NB100-56566]](https://resources.rndsystems.com/images/products/TLR4-Antibody-76B357-1-Flow-Intracellular-NB100-56566-img0025.jpg)
![Immunocytochemistry/ Immunofluorescence: TLR4 Antibody (76B357.1) - BSA Free [NB100-56566] Immunocytochemistry/ Immunofluorescence: TLR4 Antibody (76B357.1) - BSA Free [NB100-56566]](https://resources.rndsystems.com/images/products/TLR4-Antibody-76B357-1-Immunocytochemistry-Immunofluorescence-NB100-56566-img0042.jpg)
![Immunohistochemistry: TLR4 Antibody (76B357.1) - BSA Free [NB100-56566] Immunohistochemistry: TLR4 Antibody (76B357.1) - BSA Free [NB100-56566]](https://resources.rndsystems.com/images/products/TLR4-Antibody-76B357-1-Immunohistochemistry-NB100-56566-img0037.jpg)
![Immunohistochemistry-Paraffin: TLR4 Antibody (76B357.1) - BSA Free [NB100-56566] Immunohistochemistry-Paraffin: TLR4 Antibody (76B357.1) - BSA Free [NB100-56566]](https://resources.rndsystems.com/images/products/TLR4-Antibody-76B357-1-Immunohistochemistry-Paraffin-NB100-56566-img0015.jpg)
![Immunohistochemistry-Paraffin: TLR4 Antibody (76B357.1) - BSA Free [NB100-56566] Immunohistochemistry-Paraffin: TLR4 Antibody (76B357.1) - BSA Free [NB100-56566]](https://resources.rndsystems.com/images/products/TLR4-Antibody-76B357-1-Immunohistochemistry-Paraffin-NB100-56566-img0013.jpg)
![Flow Cytometry: TLR4 Antibody (76B357.1) - BSA Free [NB100-56566] Flow Cytometry: TLR4 Antibody (76B357.1) - BSA Free [NB100-56566]](https://resources.rndsystems.com/images/products/TLR4-Antibody-76B357-1-Flow-Cytometry-NB100-56566-img0045.jpg)
![Immunohistochemistry-Paraffin: TLR4 Antibody (76B357.1) - BSA Free [NB100-56566] Immunohistochemistry-Paraffin: TLR4 Antibody (76B357.1) - BSA Free [NB100-56566]](https://resources.rndsystems.com/images/products/TLR4-Antibody-76B357-1-Immunohistochemistry-Paraffin-NB100-56566-img0007.jpg)
![Immunohistochemistry-Paraffin: TLR4 Antibody (76B357.1) - BSA Free [NB100-56566] Immunohistochemistry-Paraffin: TLR4 Antibody (76B357.1) - BSA Free [NB100-56566]](https://resources.rndsystems.com/images/products/TLR4-Antibody-76B357-1-Immunohistochemistry-Paraffin-NB100-56566-img0016.jpg)
![Immunohistochemistry-Paraffin: TLR4 Antibody (76B357.1) - BSA Free [NB100-56566] Immunohistochemistry-Paraffin: TLR4 Antibody (76B357.1) - BSA Free [NB100-56566]](https://resources.rndsystems.com/images/products/TLR4-Antibody-76B357-1-Immunohistochemistry-Paraffin-NB100-56566-img0017.jpg)
![Immunohistochemistry-Frozen: TLR4 Antibody (76B357.1) - BSA Free [NB100-56566] Immunohistochemistry-Frozen: TLR4 Antibody (76B357.1) - BSA Free [NB100-56566]](https://resources.rndsystems.com/images/products/TLR4-Antibody-76B357-1-Immunohistochemistry-Frozen-NB100-56566-img0032.jpg)
![Flow Cytometry: TLR4 Antibody (76B357.1) - BSA Free [NB100-56566] Flow Cytometry: TLR4 Antibody (76B357.1) - BSA Free [NB100-56566]](https://resources.rndsystems.com/images/products/TLR4-Antibody-76B357-1-Flow-Cytometry-NB100-56566-img0031.jpg)
![Flow Cytometry: TLR4 Antibody (76B357.1) - BSA Free [NB100-56566] Flow Cytometry: TLR4 Antibody (76B357.1) - BSA Free [NB100-56566]](https://resources.rndsystems.com/images/products/TLR4-Antibody-76B357-1-Flow-Cytometry-NB100-56566-img0043.jpg)
![Flow Cytometry: TLR4 Antibody (76B357.1) - BSA Free [NB100-56566] Flow Cytometry: TLR4 Antibody (76B357.1) - BSA Free [NB100-56566]](https://resources.rndsystems.com/images/products/TLR4-Antibody-76B357-1-Flow-Cytometry-NB100-56566-img0044.jpg)
![Immunohistochemistry: TLR4 Antibody (76B357.1) - BSA Free [NB100-56566] - TLR4 Antibody (76B357.1) - BSA Free](https://resources.rndsystems.com/images/products/nb100-56566_mouse-monoclonal-tlr4-antibody-76b357-1-imgenex-img-5031a-271220231253369.jpg)
![Western Blot: TLR4 Antibody (76B357.1) - BSA Free [NB100-56566] - TLR4 Antibody (76B357.1) - BSA Free](https://resources.rndsystems.com/images/products/nb100-56566_mouse-monoclonal-tlr4-antibody-76b357-1-imgenex-img-5031a-31020241539724.jpg)
![Western Blot: TLR4 Antibody (76B357.1) - BSA Free [NB100-56566] - TLR4 Antibody (76B357.1) - BSA Free](https://resources.rndsystems.com/images/products/nb100-56566_mouse-monoclonal-tlr4-antibody-76b357-1-imgenex-img-5031a-310202415345314.jpg)
![Immunohistochemistry: TLR4 Antibody (76B357.1) - BSA Free [NB100-56566] - TLR4 Antibody (76B357.1) - BSA Free](https://resources.rndsystems.com/images/products/nb100-56566_mouse-monoclonal-tlr4-antibody-76b357-1-imgenex-img-5031a-310202415293340.jpg)
![Western Blot: TLR4 Antibody (76B357.1) - BSA Free [NB100-56566] - TLR4 Antibody (76B357.1) - BSA Free](https://resources.rndsystems.com/images/products/nb100-56566_mouse-monoclonal-tlr4-antibody-76b357-1-imgenex-img-5031a-310202415175298.jpg)
![Western Blot: TLR4 Antibody (76B357.1) - BSA Free [NB100-56566] - TLR4 Antibody (76B357.1) - BSA Free](https://resources.rndsystems.com/images/products/nb100-56566_mouse-monoclonal-tlr4-antibody-76b357-1-imgenex-img-5031a-31020241534393.jpg)
![Western Blot: TLR4 Antibody (76B357.1) - BSA Free [NB100-56566] - TLR4 Antibody (76B357.1) - BSA Free](https://resources.rndsystems.com/images/products/nb100-56566_mouse-monoclonal-tlr4-antibody-76b357-1-imgenex-img-5031a-310202415304291.jpg)
![Western Blot: TLR4 Antibody (76B357.1) - BSA Free [NB100-56566] - TLR4 Antibody (76B357.1) - BSA Free](https://resources.rndsystems.com/images/products/nb100-56566_mouse-monoclonal-tlr4-antibody-76b357-1-imgenex-img-5031a-310202415371989.jpg)
![Western Blot: TLR4 Antibody (76B357.1) - BSA Free [NB100-56566] - TLR4 Antibody (76B357.1) - BSA Free](https://resources.rndsystems.com/images/products/nb100-56566_mouse-monoclonal-tlr4-antibody-76b357-1-imgenex-img-5031a-310202415291964.jpg)
![Western Blot: TLR4 Antibody (76B357.1) - BSA Free [NB100-56566] - TLR4 Antibody (76B357.1) - BSA Free](https://resources.rndsystems.com/images/products/nb100-56566_mouse-monoclonal-tlr4-antibody-76b357-1-imgenex-img-5031a-31020241530423.jpg)
![Western Blot: TLR4 Antibody (76B357.1) - BSA Free [NB100-56566] - TLR4 Antibody (76B357.1) - BSA Free](https://resources.rndsystems.com/images/products/nb100-56566_mouse-monoclonal-tlr4-antibody-76b357-1-imgenex-img-5031a-31020241534332.jpg)
![Western Blot: TLR4 Antibody (76B357.1) - BSA Free [NB100-56566] - TLR4 Antibody (76B357.1) - BSA Free](https://resources.rndsystems.com/images/products/nb100-56566_mouse-monoclonal-tlr4-antibody-76b357-1-imgenex-img-5031a-31020241683915.jpg)
![Immunocytochemistry/ Immunofluorescence: TLR4 Antibody (76B357.1) - BSA Free [NB100-56566] - TLR4 Antibody (76B357.1) - BSA Free](https://resources.rndsystems.com/images/products/nb100-56566_mouse-monoclonal-tlr4-antibody-76b357-1-imgenex-img-5031a-310202415525244.jpg)
![Immunocytochemistry/ Immunofluorescence: TLR4 Antibody (76B357.1) - BSA Free [NB100-56566] - TLR4 Antibody (76B357.1) - BSA Free](https://resources.rndsystems.com/images/products/nb100-56566_mouse-monoclonal-tlr4-antibody-76b357-1-imgenex-img-5031a-31020241553739.jpg)
![Western Blot: TLR4 Antibody (76B357.1) - BSA Free [NB100-56566] - TLR4 Antibody (76B357.1) - BSA Free](https://resources.rndsystems.com/images/products/nb100-56566_mouse-monoclonal-tlr4-antibody-76b357-1-imgenex-img-5031a-31020241553730.jpg)
![Western Blot: TLR4 Antibody (76B357.1) - BSA Free [NB100-56566] - TLR4 Antibody (76B357.1) - BSA Free](https://resources.rndsystems.com/images/products/nb100-56566_mouse-monoclonal-tlr4-antibody-76b357-1-imgenex-img-5031a-310202415535180.jpg)
![Immunocytochemistry/ Immunofluorescence: TLR4 Antibody (76B357.1) - BSA Free [NB100-56566] - TLR4 Antibody (76B357.1) - BSA Free](https://resources.rndsystems.com/images/products/nb100-56566_mouse-monoclonal-tlr4-antibody-76b357-1-imgenex-img-5031a-31020241553724.jpg)
![Western Blot: TLR4 Antibody (76B357.1) - BSA Free [NB100-56566] - TLR4 Antibody (76B357.1) - BSA Free](https://resources.rndsystems.com/images/products/nb100-56566_mouse-monoclonal-tlr4-antibody-76b357-1-imgenex-img-5031a-31020241682147.jpg)
![Western Blot: TLR4 Antibody (76B357.1) - BSA Free [NB100-56566] - TLR4 Antibody (76B357.1) - BSA Free](https://resources.rndsystems.com/images/products/nb100-56566_mouse-monoclonal-tlr4-antibody-76b357-1-imgenex-img-5031a-31020241552396.jpg)
![Western Blot: TLR4 Antibody (76B357.1) - BSA Free [NB100-56566] - TLR4 Antibody (76B357.1) - BSA Free](https://resources.rndsystems.com/images/products/nb100-56566_mouse-monoclonal-tlr4-antibody-76b357-1-imgenex-img-5031a-3102024168390.jpg)
![Western Blot: TLR4 Antibody (76B357.1) - BSA Free [NB100-56566] - TLR4 Antibody (76B357.1) - BSA Free](https://resources.rndsystems.com/images/products/nb100-56566_mouse-monoclonal-tlr4-antibody-76b357-1-imgenex-img-5031a-310202415532051.jpg)
![Western Blot: TLR4 Antibody (76B357.1) - BSA Free [NB100-56566] - TLR4 Antibody (76B357.1) - BSA Free](https://resources.rndsystems.com/images/products/nb100-56566_mouse-monoclonal-tlr4-antibody-76b357-1-imgenex-img-5031a-3102024155457.jpg)
![Flow Cytometry: TLR4 Antibody (76B357.1) - BSA Free [NB100-56566] - TLR4 Antibody (76B357.1) - BSA Free](https://resources.rndsystems.com/images/products/nb100-56566_mouse-monoclonal-tlr4-antibody-76b357-1-imgenex-img-5031a-31020241552524.jpg)
![Western Blot: TLR4 Antibody (76B357.1) - BSA Free [NB100-56566] - TLR4 Antibody (76B357.1) - BSA Free](https://resources.rndsystems.com/images/products/nb100-56566_mouse-monoclonal-tlr4-antibody-76b357-1-imgenex-img-5031a-310202415523925.jpg)
![Western Blot: TLR4 Antibody (76B357.1) - BSA Free [NB100-56566] - TLR4 Antibody (76B357.1) - BSA Free](https://resources.rndsystems.com/images/products/nb100-56566_mouse-monoclonal-tlr4-antibody-76b357-1-imgenex-img-5031a-310202415535173.jpg)
![Immunocytochemistry/ Immunofluorescence: TLR4 Antibody (76B357.1) - BSA Free [NB100-56566] - TLR4 Antibody (76B357.1) - BSA Free](https://resources.rndsystems.com/images/products/nb100-56566_mouse-monoclonal-tlr4-antibody-76b357-1-imgenex-img-5031a-31020241612688.jpg)

-(01-mg)_NB100-56566_9196.jpg)