Basement membrane extract (BME) is a type of extracellular matrix (ECM) derived from Engelbreth-Holm-Swarm mouse sarcomas. BME is commonly used in cell culture and tissue engineering to provide a 2-D or 3-D scaffold for cells, allowing them to grow, differentiate, and interact in an environment that closely mimics the native ECM. This makes BME invaluable for modeling the behavior of cells in vivo. All Cultrex BME formulations are rigorously tested to ensure performance, quality, and lot-to-lot consistency.
Cultrex Reduced Growth Factor Basement Membrane Extract, Type 2, Pathclear
R&D Systems | Catalog # 3533-010-02

Key Product Details
Features
Key Benefits
- Qualified for use in organoid cell culture
- Commonly used robust and established organoid systems
- Reduced growth factor formulation provides a more defined culture system
- Quality controlled for performance consistency
Species
Product Summary
Cultrex RGF BME, Type 2 provides a proprietary formulation that has a high storage modulus and is designed for use in robust tissue organoid culture as well as other applications requiring an extracellular matrix scaffold.
Video of Cultrex BME Best Practices and Protocols.
Protocol for Mouse Enteric Organoid Culture.
Protocol for Human Gastric Organoid Culture.
Protocol for Human Liver Organoid Culture.
Protocol for Human Lung Organoid Culture.
Protocol for Human Intestinal Organoid Culture.
Protocol for Harvesting Organoids for Biochemical Analysis.
Product Specifications
Source
Protein Concentration
Endotoxin Level
Sterility Testing
Cell Culture Testing
Gelling Assay - Cultrex RGF BME, Type 2 gels in less than 30 minutes at 37 °C, and maintains the gelled form in culture medium for a minimum of 7 days at 37 °C.
Dome Assay Cultrex RGF BME, Type 2 forms and maintains stable 3-D dome structures on cell culture plates.
Viral Testing
Scientific Data Images
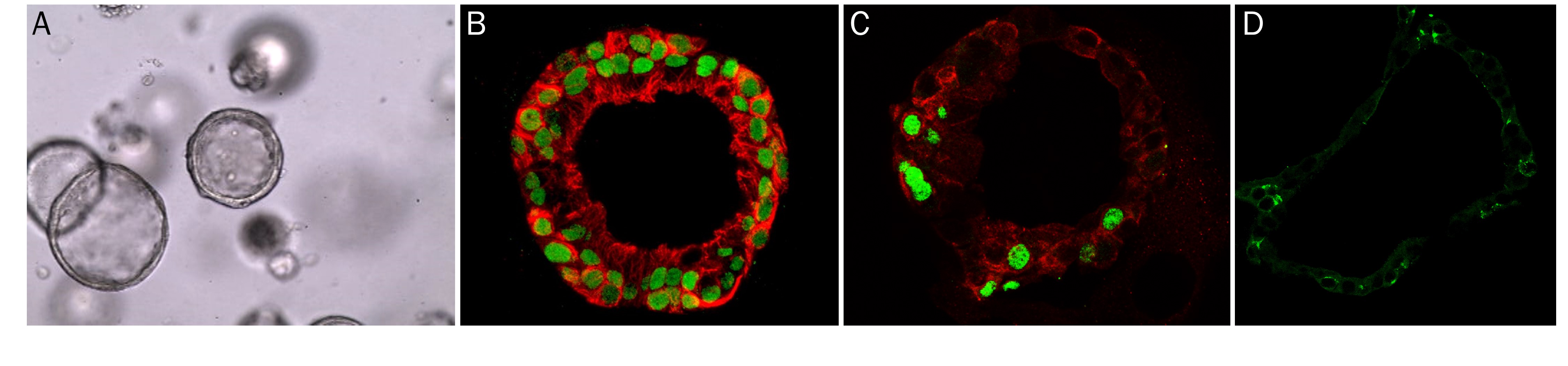
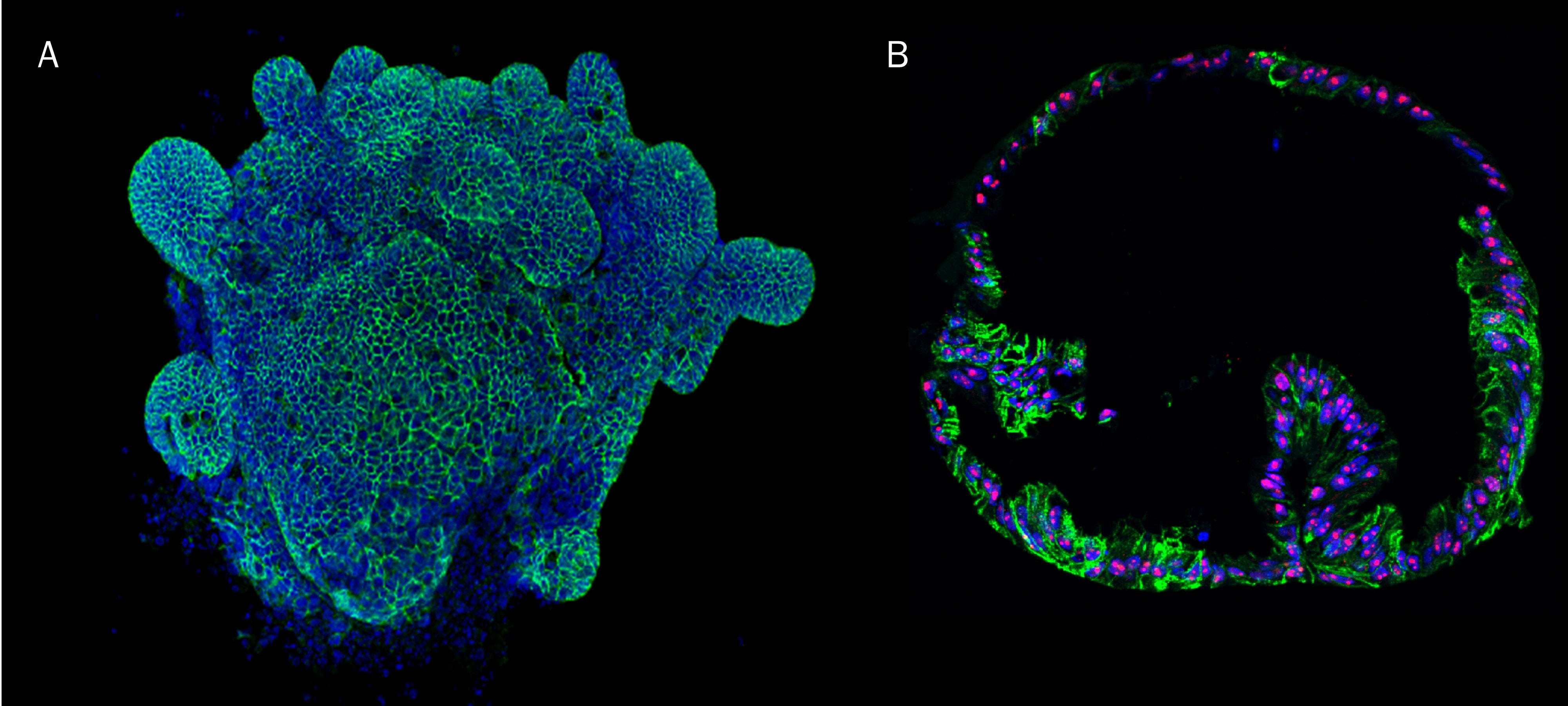
Mouse Intestinal Organoids Cultured in Cultrex RGF BME, Type 2.
Mouse intestinal organoids cultured in Cultrex RGF BME, Type 2 were immersion fixed and processed for whole mount staining or paraffin embedding and sectioning for immunocytochemistry. A) Whole mount mouse intestinal organoids were stained using Goat Anti-Human/Mouse E-Cadherin Antigen Affinity-purified Polyclonal Antibody (green; Catalog # AF748) at 10 µg/mL for 3 hours at room temperature. Cells were counterstained with DAPI (blue). B) Paraffin-embedded mouse intestinal tissue stained for Human Cadherin‑17 Antibody (green; Catalog # MAB1032), Human Ki67/MKI67 Antibody (red; Catalog # AF7617), and counterstained with DAPI (blue).Human Lung Organoids Cultured in Cultrex RGF BME, Type 2.
A) Representative brightfield image of human lung organoids cultured using Cultrex RGF BME, Type 2. B) Expression of Sox2 (green; Catalog # AF2018) and Acetylated Tubulin (red; Novus Biologicals, Catalog # NB600-567). C) Expression of p63/TP73L (green; Catalog # AF1916) and Cytokeratin 10 (red; Novus Biologicals, Catalog # NBP2-61736). D) Expression of Podoplanin (green; Catalog # AF3670) as a marker of type 1 alveolar cells.Liver Organoid Formation and Differentiation in Cultrex RGF BME, Type 2.
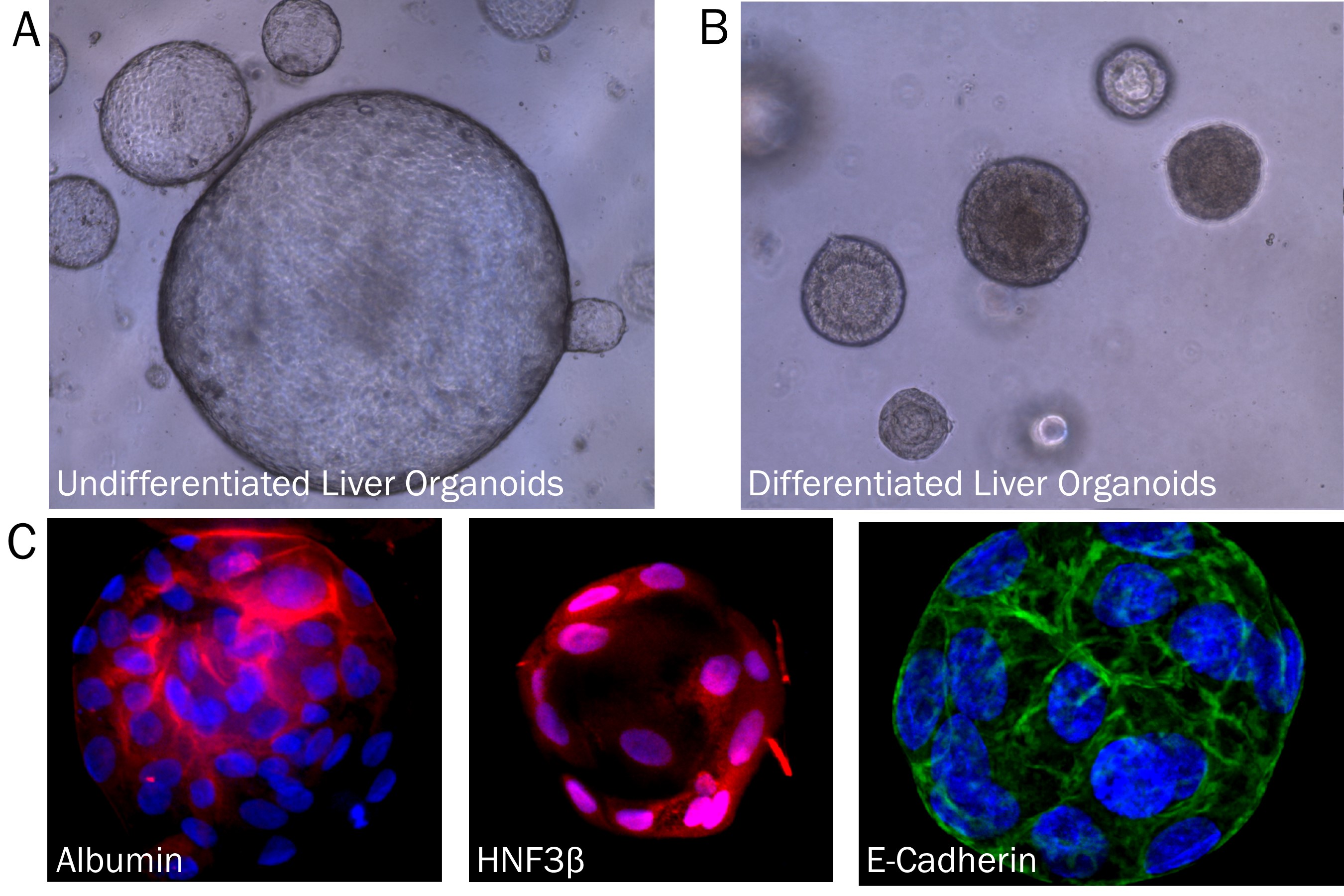
Human Liver organoids were derived from human biopsy tissue. Undifferentiated organoids were formed by embedding dissociated tissue in Cultrex RGF BME, Type 2 and culturing in specialized media. The organoids were differentiated using media containing Recombinant Human FGF-19 (Catalog # 969-FG), DAPT (Catalog # 2634), and Dexamethasone (Catalog # 1126). A) Undifferentiated liver organoids. B) Liver organoids shrink as they differentiate. C) Representative images of differentiated liver organoids expression hepatocyte markers, Albumin and HNF3 beta, as well as E-Cadherin.Human Intestinal Organoids Cultured using Cultrex RGF BME, Type 2.
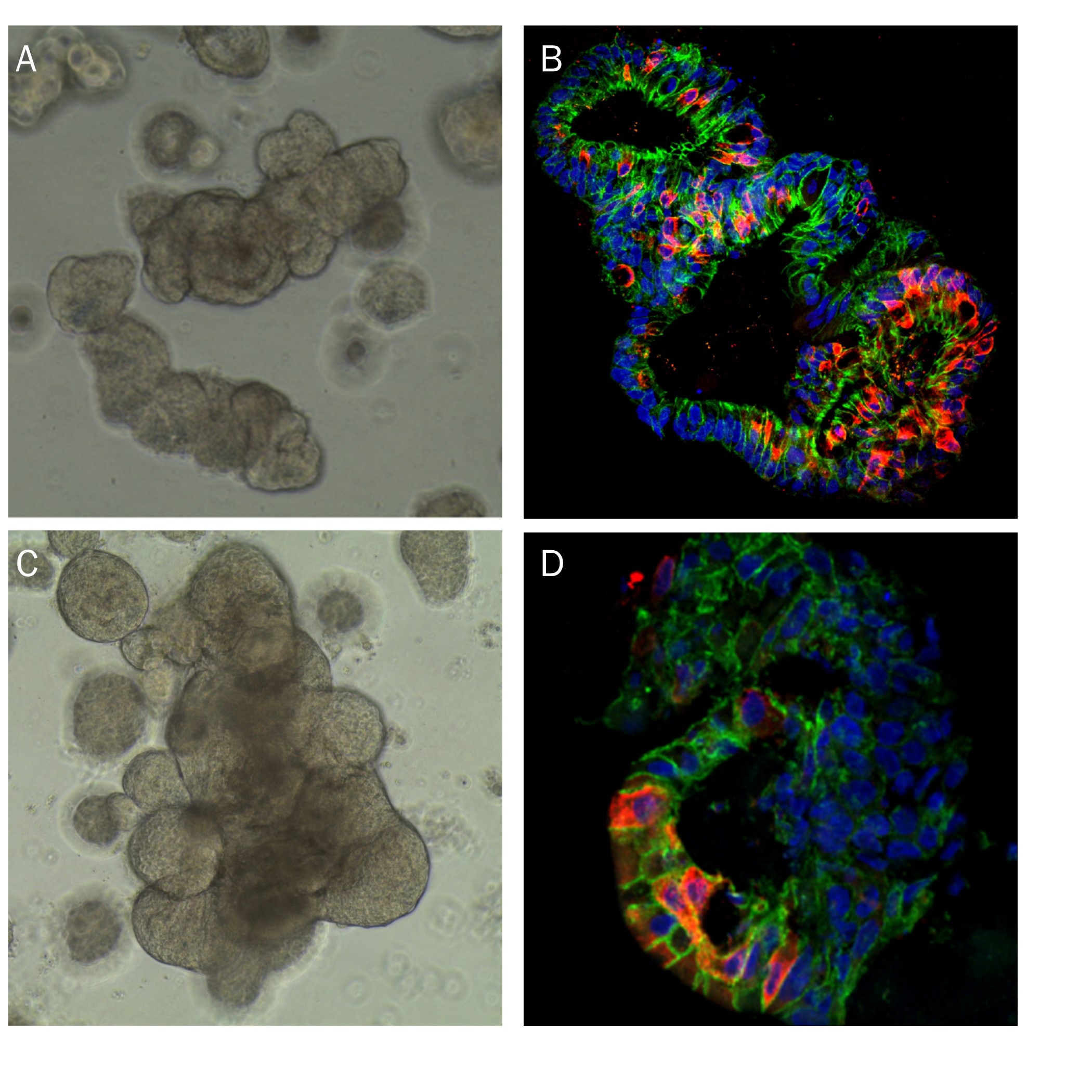
Human transverse colon organoids (A,B) and human ileum organoids (C, D) were grown using cells isolated from transverse colon and ileum biopsy tissue, respectively. Organoids were embedded in Cultrex RGF BME, Type 2 as a scaffold matrix. A) Brightfield image of human transverse organoids. B) Human transverse organoid stained using Goat Anti-Human/Mouse E-Cadherin Antigen Affinity-purified Polyclonal Antibody (green; Catalog # AF748), a MUC2 Antibody (red; Catalog # NBP2-44431), and DAPI (blue). C) Brightfield image of human ileum organoids. D) Human ileum organoid stained using a Aldolase B Antibody (red; Catalog # NBP2-15345), a Human Cadherin-17 Antibody (green; Catalog # MAB1032, and DAPI (blue).Formation of HepG2 Spheroids in Cultrex RGF BME, Type 2.
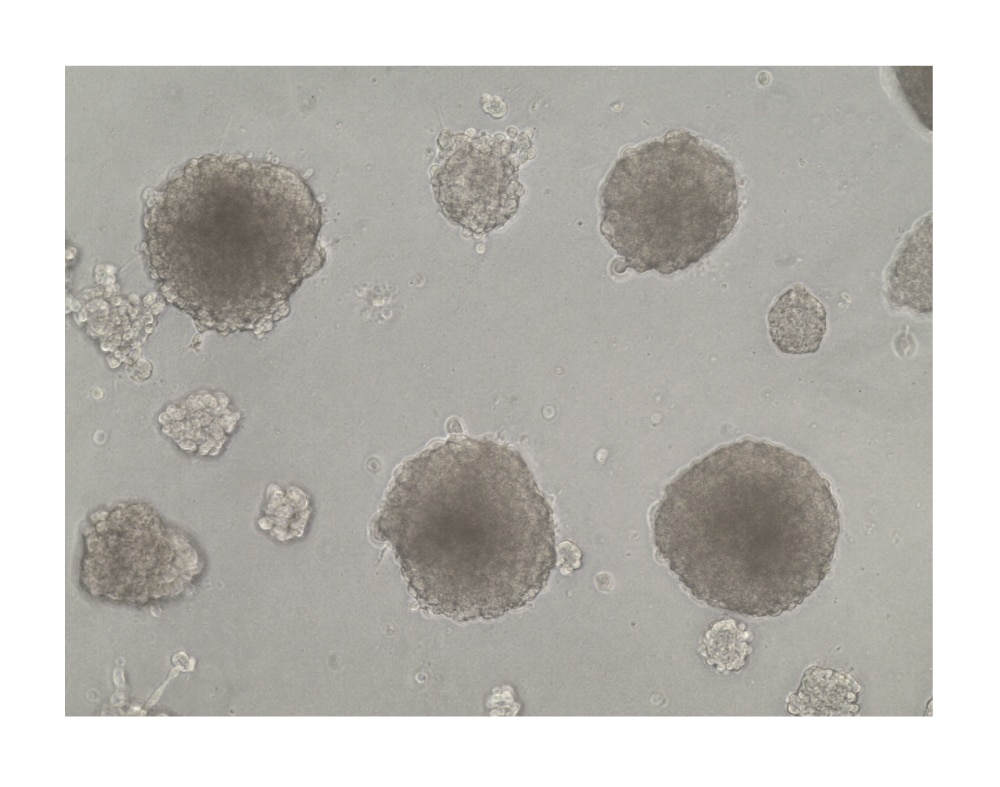
Hepatocyte spheroids were formed by plating HepG2 liver hepatocellular carcinoma cells (10,000 cells per well) in a 24-well plate coated with 5 mg/mL of Cultrex RGF BME, Type 2. Spheroids were cultured for 21 days prior imaging by brightfield microscopy.Formulation, Preparation, and Storage
Shipping
Storage
Stability
Background: Basement Membrane Extracts
Additional Basement Membrane Extracts Products
Product Documents
Certificate of Analysis
To download a Certificate of Analysis, please enter a lot or batch number in the search box below.
Note: Certificate of Analysis not available for kit components.
Citations for Cultrex Reduced Growth Factor Basement Membrane Extract, Type 2, Pathclear
Customer Reviews (5)
Have you used Cultrex Reduced Growth Factor Basement Membrane Extract, Type 2, Pathclear?
Submit a review and receive an Amazon gift card!
$25/€18/£15/$25CAN/¥2500 Yen for a review with an image
$10/€7/£6/$10CAN/¥1110 Yen for a review without an image
Submit a review
Customer Images
-
Verified Customer | Posted 06/18/2022dissolve it on ice, and don't put it outside for too not time
-
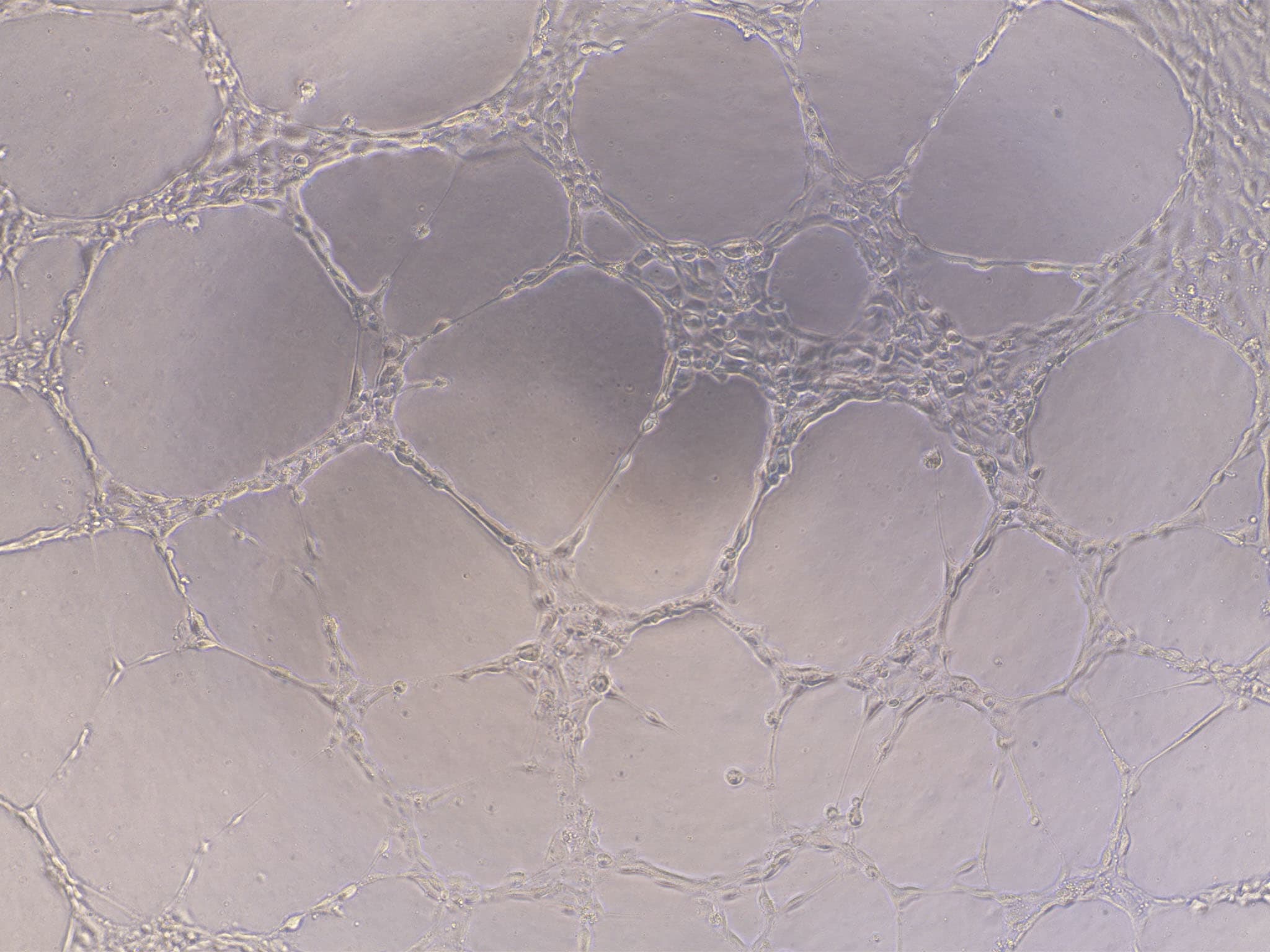
Verified Customer | Posted 02/21/2022Aortic sprouting assay: mouse aortic ring cultured in a 48-well plate and imaged in an inverted microscope after 5 days. Results are very consistent with this BME (used one layer under and another on top of the aortic ring).
-
Verified Customer | Posted 04/13/2021
-
Verified Customer | Posted 12/09/2020We are using the BME at a final dilution of 1/30 for the maintenance of iPSC and differentiation into endoderm as well as mesoderm lineages. Attachment, growth, and differentiation of our iPSC is consistent, and we observed only minor variations between different lots.
-
Application: Cell migration/motilityVerified Customer | Posted 06/01/2018
There are no reviews that match your criteria.
FAQs
-
Q: How does Cultrex® Basement Membrane Extract (BME) promote cell differentiation?
A: All epithelial and endothelial cells are in contact with a basement membrane matrix on at least one of their surfaces. By providing them with their natural matrix in vitro as a substrate for the cells that provides biological cues, the cells can assume a more physiological morphology (i.e. correct shape) and begin expression of cell-lineage specific proteins. Two-dimensional plastic surfaces, in combination with serum-containing media, cause cells to flatten, proliferate and de-differentiate.