Epithelial (E)‑Cadherin (ECAD), also known as cell-CAM120/80 in the human, uvomorulin in the mouse, Arc-1 in the dog, and L-CAM in the chicken, is a member of the cadherin family of cell adhesion molecules. Cadherins are calcium-dependent transmembrane proteins, which bind to one another in a homophilic manner. On their cytoplasmic side, they associate with the three catenins, alpha, beta, and gamma (plakoglobin). This association links the cadherin protein to the cytoskeleton. Without association with the catenins, the cadherins are non-adhesive. Cadherins play a role in development, specifically in tissue formation. They may also help to maintain tissue architecture in the adult. E-Cadherin may also play a role in tumor development, as loss of E-Cadherin has been associated with tumor invasiveness. E-Cadherin is a classical cadherin molecule. Classical cadherins consist of a large extracellular domain which contains DXD and DXNDN repeats responsible for mediating calcium‑dependent adhesion, a single-pass transmembrane domain, and a short carboxy-terminal cytoplasmic domain responsible for interacting with the catenins. E‑Cadherin contains five extracellular calcium-binding domains of approximately 110 amino acids each.
Human/Mouse E‑Cadherin Antibody
R&D Systems | Catalog # AF748

Key Product Details
Validated by
Biological Validation
Species Reactivity
Validated:
Human, Mouse
Cited:
Human, Mouse, Rat, Porcine, Frog - Xenopus (African Clawed Frog), Transgenic Mouse
Applications
Validated:
Immunohistochemistry, Western Blot, Flow Cytometry, Dual RNAscope ISH-IHC Compatible, Immunocytochemistry, Simple Western, CyTOF-ready
Cited:
Immunohistochemistry, Immunohistochemistry-Paraffin, Immunohistochemistry-Frozen, Western Blot, Neutralization, Flow Cytometry, Immunofluorescence, Immunocytochemistry, Confocal Microscopy, IFC, Proximity Ligation Assay (PLA)
Label
Unconjugated
Antibody Source
Polyclonal Goat IgG
Loading...
Product Specifications
Immunogen
Mouse myeloma cell line NS0-derived recombinant mouse E-Cadherin
Asp157-Val709
Accession # P09803
Asp157-Val709
Accession # P09803
Specificity
Detects mouse E-Cadherin in direct ELISAs and Western blots.
Clonality
Polyclonal
Host
Goat
Isotype
IgG
Scientific Data Images for Human/Mouse E‑Cadherin Antibody
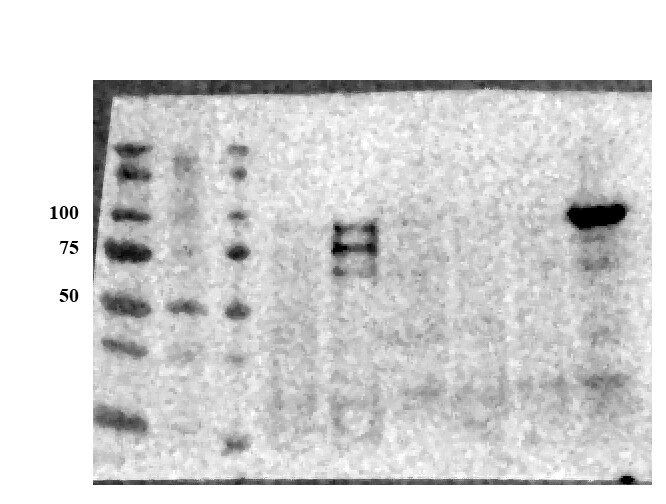
Detection of Human and Mouse E‑Cadherin by Western Blot.
Western blot shows lysates of A431 human epithelial carcinoma cell line, A549 human lung carcinoma cell line, HepG2 human hepatocellular carcinoma cell line, P19 mouse embryonal carcinoma cell line, and 4T1 mouse breast cancer cell line. PVDF membrane was probed with 0.5 µg/mL of Goat Anti-Human/Mouse E-Cadherin Antigen Affinity-purified Polyclonal Antibody (Catalog # AF748) followed by HRP-conjugated Anti-Goat IgG Secondary Antibody (Catalog # HAF017). A specific band was detected for E-Cadherin at approximately 110 kDa (as indicated). This experiment was conducted under reducing conditions and using Immunoblot Buffer Group 1.E‑Cadherin in D3 Mouse Embryonic Stem Cell Line.
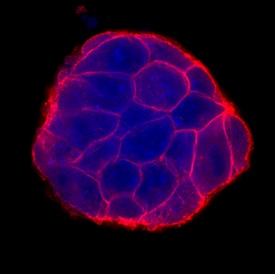
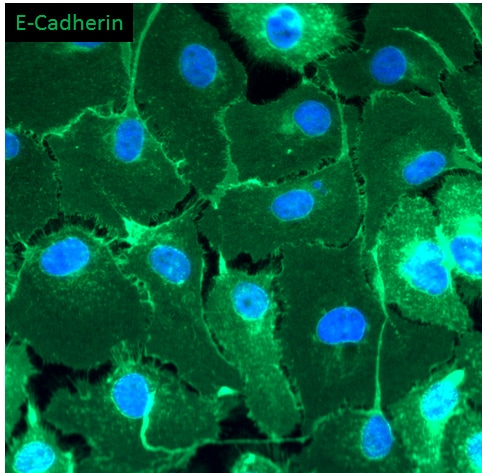
E-Cadherin was detected in immersion fixed D3 mouse embryonic stem cell line using Goat Anti-Human/Mouse E-Cadherin Antigen Affinity-purified Polyclonal Antibody (Catalog # AF748) at 10 µg/mL for 3 hours at room temperature. Cells were stained using the NorthernLights™ 557-conjugated Anti-Goat IgG Secondary Antibody (red; Catalog # NL001) and counterstained with DAPI (blue). Specific staining was localized to cell surfaces. View our protocol for Fluorescent ICC Staining of Stem Cells on Coverslips.E‑Cadherin in Mouse Skin.
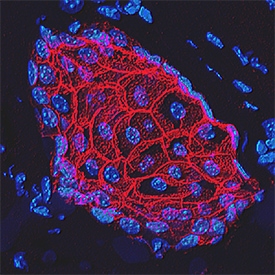
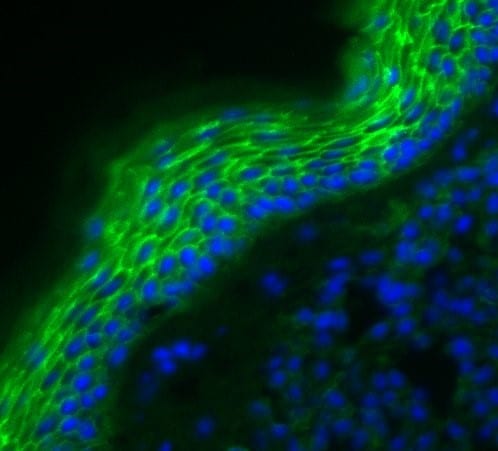
E-Cadherin was detected in perfusion fixed frozen sections of mouse skin using Goat Anti-Human/Mouse E-Cadherin Antigen Affinity-purified Polyclonal Antibody (Catalog # AF748) at 1.7 µg/mL overnight at 4 °C. Tissue was stained using the NorthernLights™ 557-conjugated Anti-Goat IgG Secondary Antibody (red; Catalog # NL001) and counterstained with DAPI (blue). Specific staining was localized to plasma membranes in keratinocytes. View our protocol for Fluorescent IHC Staining of Frozen Tissue Sections.E‑Cadherin in Mouse Intestinal Organoids.
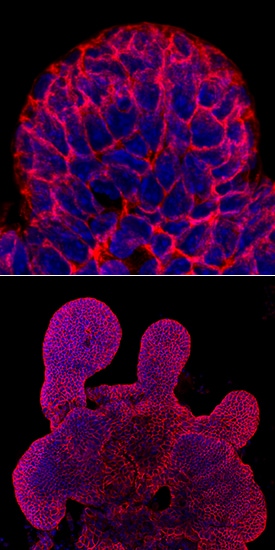
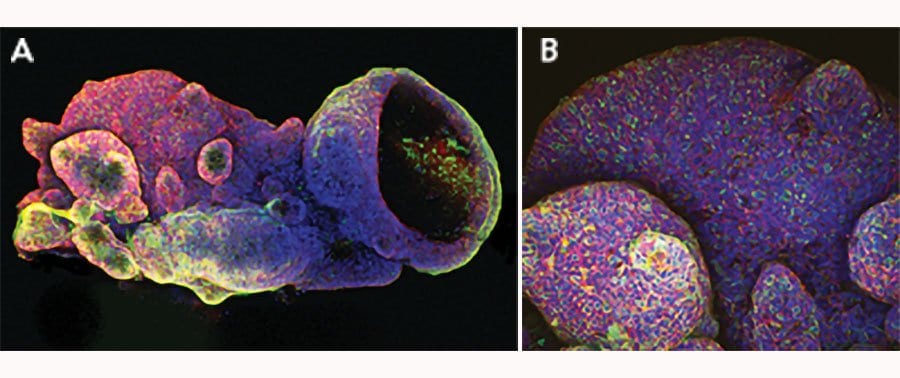
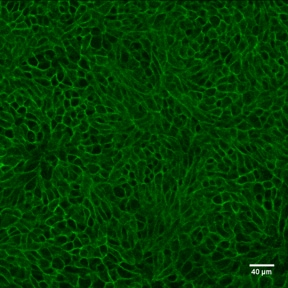
E-Cadherin was detected in immersion fixed mouse intestinal organoids using Goat Anti-Human/Mouse E-Cadherin Antigen Affinity-purified Polyclonal Antibody (Catalog # AF748) at 10 µg/mL for 3 hours at room temperature. Cells were stained using the NorthernLights™ 557-conjugated Anti-Goat IgG Secondary Antibody (green; Catalog # NL001) and counterstained with DAPI (blue). Magnification shown at 100X (upper panel) and 40X (lower panel). Specific staining was localized to cell surfaces. View our protocol for Fluorescent ICC Staining of Cells on Coverslips.E‑Cadherin in Mouse Intestinal Organoids.
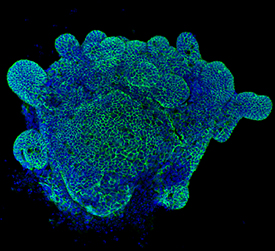
E-Cadherin was detected in immersion fixed mouse intestinal organoids using Goat Anti-Human/Mouse E-Cadherin Antigen Affinity-purified Polyclonal Antibody (Catalog # AF748) at 10 µg/mL for 3 hours at room temperature. Cells were stained using the NorthernLights™ 493-conjugated Anti-Goat IgG Secondary Antibody (green; Catalog # NL003) and counterstained with DAPI (blue). Specific staining was localized to cell surfaces. View our protocol for Fluorescent ICC Staining of Cells on Coverslips.E‑Cadherin in Mouse Spinal Cord.
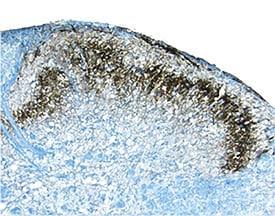
E-Cadherin was detected in perfusion fixed frozen sections of mouse spinal cord using Goat Anti-Human/Mouse E-Cadherin Antigen Affinity-purified Polyclonal Antibody (Catalog # AF748) at 1.7 µg/mL overnight at 4 °C. Tissue was stained using the Anti-Goat HRP-DAB Cell & Tissue Staining Kit (brown; Catalog # CTS008) and counterstained with hematoxylin (blue). Specific staining was localized to dorsal horn. View our protocol for Chromogenic IHC Staining of Frozen Tissue Sections.Detection of Human and Mouse E‑Cadherin by Simple WesternTM.
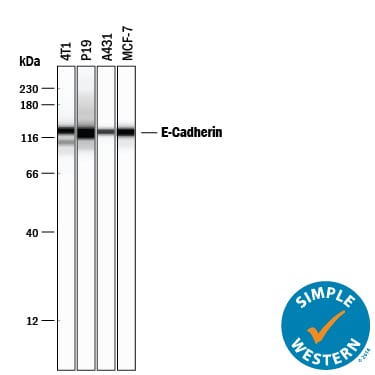
Simple Western lane view shows lysates of 4T1 mouse breast cancer cell line, P19 mouse embryonal carcinoma cell line, A431 human epithelial carcinoma cell line, and MCF-7 human breast cancer cell line, loaded at 0.2 mg/mL. A specific band was detected for E-Cadherin at approximately 128 kDa (as indicated) using 5 µg/mL of Goat Anti-Human/Mouse E-Cadherin Antigen Affinity-purified Polyclonal Antibody (Catalog # AF748) followed by 1:50 dilution of HRP-conjugated Anti-Goat IgG Secondary Antibody (Catalog # HAF109). This experiment was conducted under reducing conditions and using the 12-230 kDa separation system.Detection of Mouse E-Cadherin by Immunocytochemistry/Immunofluorescence
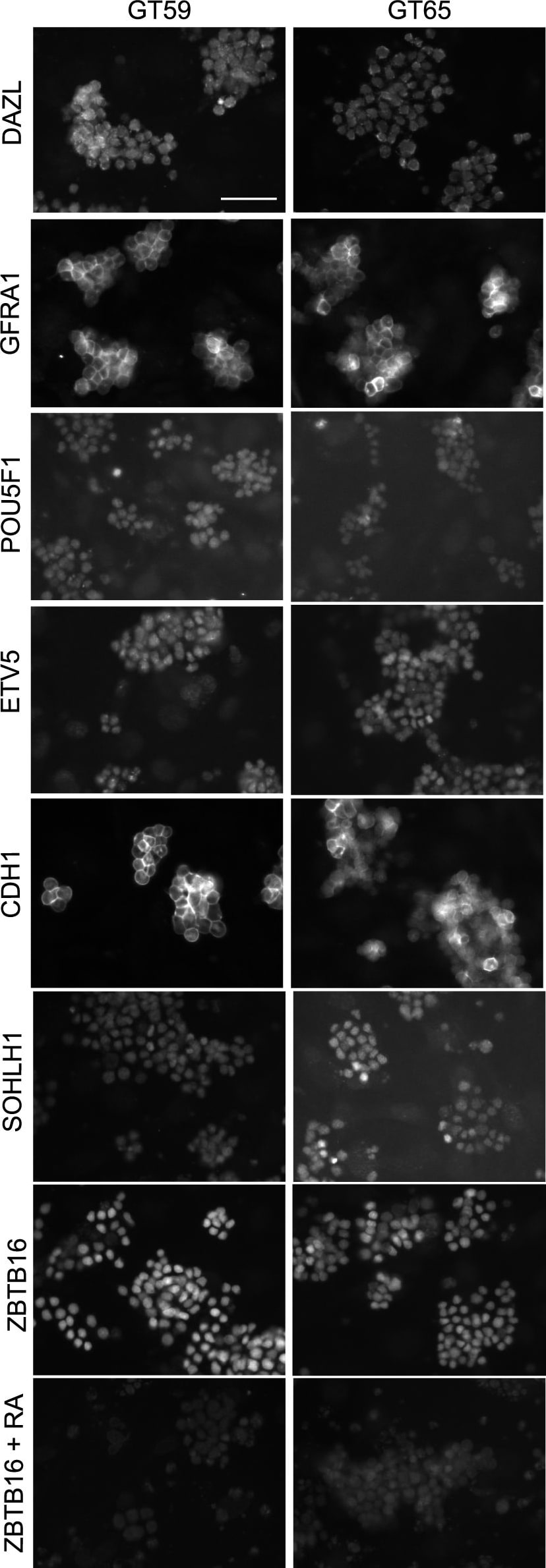
Retention of the spermatogonial phenotype following gene correction.Immunostaining was performed on gene-corrected GT59 (left) and GT65 cells (right): DAZL, a germ cell specific marker; GFRA1, POU5F1, ETV5, CDH1, and SOHLH1, markers of undifferentiated spermatogonia. Additionally, GT59 and GT65 cells were treated with the differentiation factor, retinoic acid (1 µM) or a vehicle control and then immunostained to examine levels of ZBTB16, a marker of undifferentiated spermatogonia. Bar represents 50 microns. Image collected and cropped by CiteAb from the following publication (https://pubmed.ncbi.nlm.nih.gov/25409432), licensed under a CC-BY license. Not internally tested by R&D Systems.Detection of Mouse E-Cadherin by Immunocytochemistry/Immunofluorescence
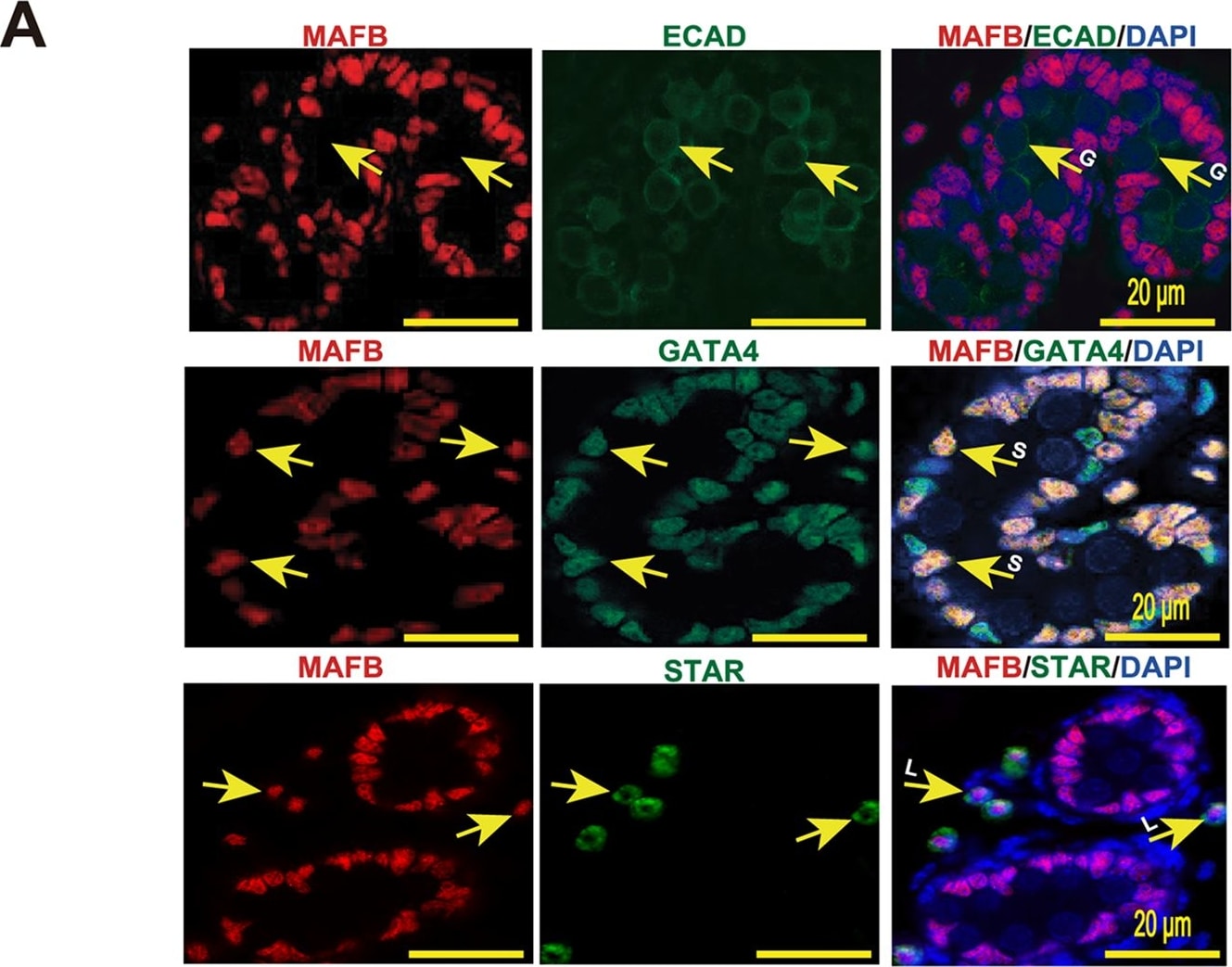
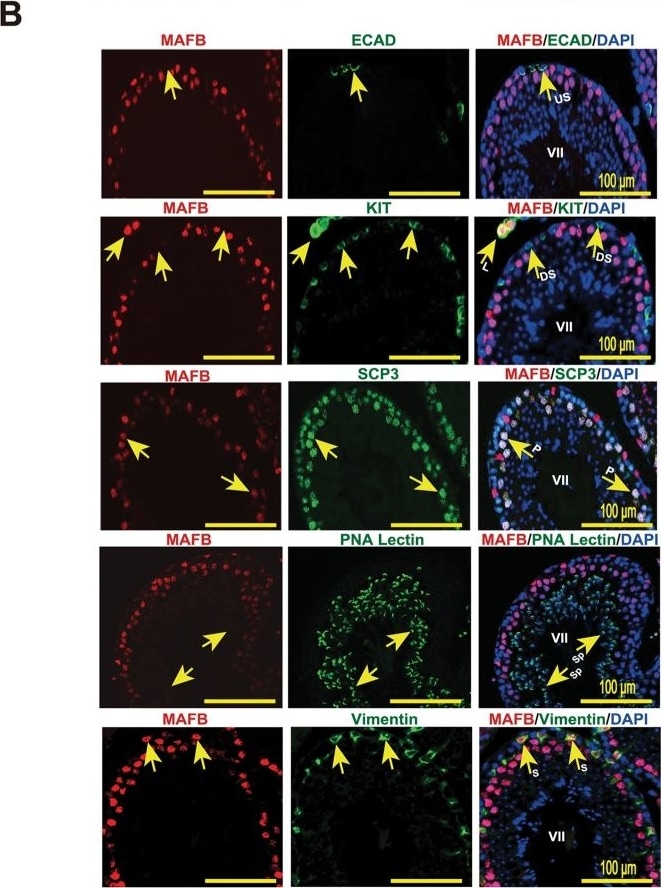
Localization of MAFB in mouse testes.(A) Localization of MAFB in E18.5 mouse testes. Double immunostaining of MAFB with E-cadherin (ECAD), GATA4, or STAR is shown. Nuclei were counterstained with DAPI. The color of each marker is indicated above the images. G; germ cells. S; Sertoli cells. L; Leydig cells. MAFB was specifically detected in Leydig cells and Sertoli cells. (B) Localization of MAFB in adult mouse testes. Double immunostaining of MAFB with E-cadherin (ECAD), KIT, SCP3, PNA Lectin, or vimentin is shown. Nuclei were counterstained with DAPI. The color of each marker is indicated above the images. All seminiferous tubules shown represent stage VII. US; undifferentiated spermatogonia. DS; differentiated spermatogonia. P; pachytene spermatocytes. Sp; spermatids. S; Sertoli cells. L; Leydig cells. MAFB was specifically detected in Leydig cells, Sertoli cells, and pachytene spermatocytes. Image collected and cropped by CiteAb from the following publication (https://dx.plos.org/10.1371/journal.pone.0190800), licensed under a CC-BY license. Not internally tested by R&D Systems.Detection of Mouse E-Cadherin by Immunocytochemistry/Immunofluorescence
Cellular localization of PEDF in testicular spermatogenic cells. Methanol-fixed cells were used to stain premeiotic and meiotic/postmeiotic cells as described in Figure 2. (A) alpha 6-integrin, (B) GFR-alpha, (C) CDH1, (D) and NC—IF staining without the presence of primary antibody. (E) CREM, (F) Boule, (G) Acrosin and (H) NC—IF staining without the presence of primary antibody. Scale bar: 100 μm. Image collected and cropped by CiteAb from the following publication (https://pubmed.ncbi.nlm.nih.gov/33498962), licensed under a CC-BY license. Not internally tested by R&D Systems.Detection of Mouse E-Cadherin by Immunocytochemistry/Immunofluorescence
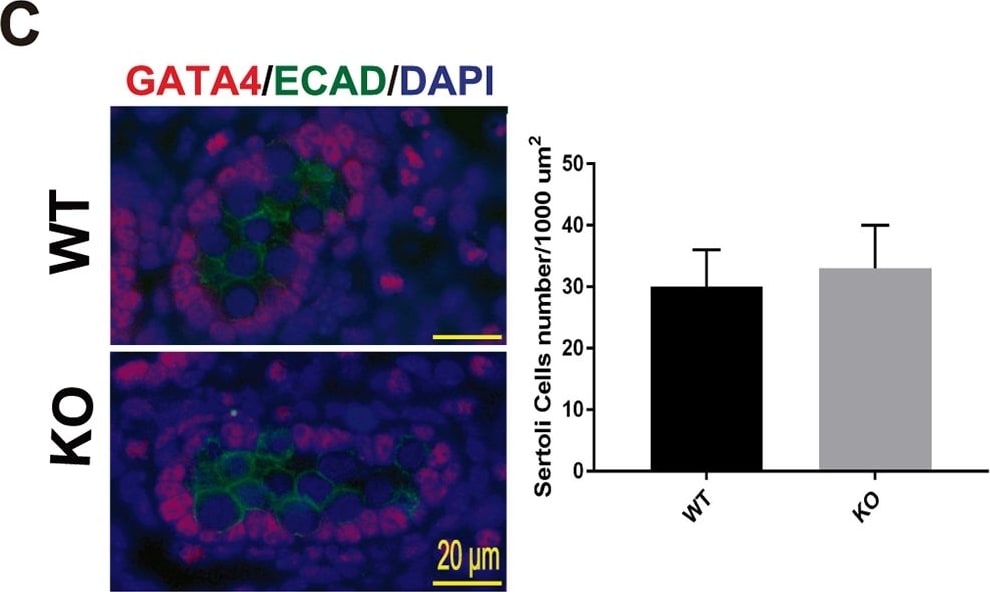
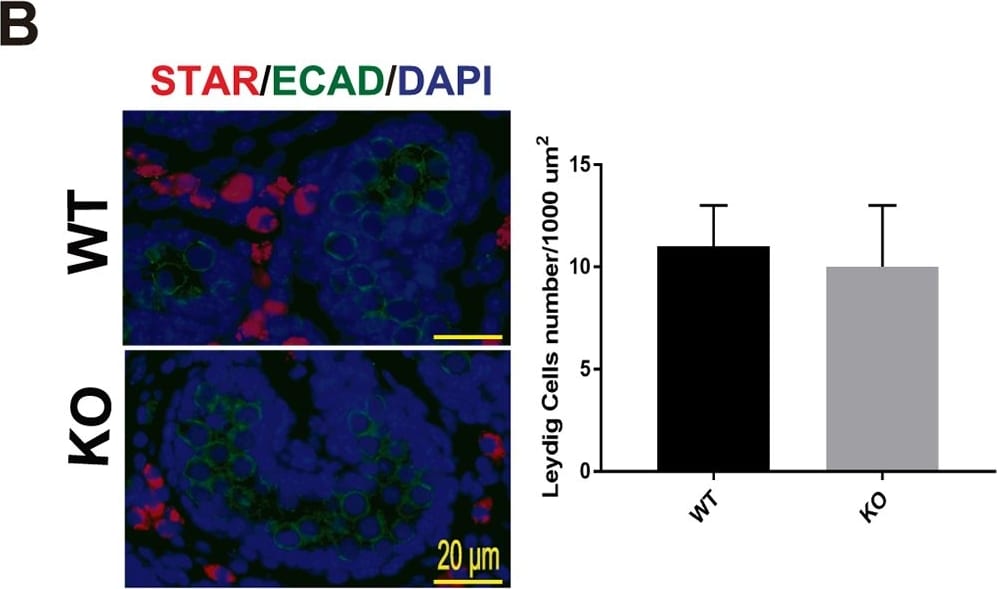
Testis morphogenesis of Mafb KO embryos developed normally.(A) Histological section of WT and KO E18.5 testes stained with HE. No morphological alteration or distribution was detectable. (B and C) Counts of Leydig and Sertoli cells from E18.5 WT and KO testes. Embryonic testes (n = 3 per genotype) were sectioned, and 4 sections for each gonad were randomly selected and stained with the germ cell marker E-cadherin (green) together with either STAR (red) or GATA4 (red). Numbers of STAR-positive cells outside seminiferous tubules (Leydig cells) or GATA4-positive cells inside the tubules (Sertoli cells) were counted per unit area. The values are the mean±S.D. *P<0.05. There was no significant difference between WT and KO cell counts. (D) The expression of genes involved in testes development and function. mRNA expression of the gene markers encoding for PGCs (Oct4), Leydig cells (Cyp17a1, StAR, Insl3, Hsd3b1, and Cyp11a1), and Sertoli cells (Amh, Sox9, and WT1) was determined by qRT-PCR in E18.5 WT and Mafb KO testes. Gene expression levels were normalized to Hprt. The bars represent the mean±SEM of five individuals. *P<0.05. Image collected and cropped by CiteAb from the following publication (https://dx.plos.org/10.1371/journal.pone.0190800), licensed under a CC-BY license. Not internally tested by R&D Systems.Detection of Mouse E-Cadherin by Immunocytochemistry/Immunofluorescence
Localization of MAFB in mouse testes.(A) Localization of MAFB in E18.5 mouse testes. Double immunostaining of MAFB with E-cadherin (ECAD), GATA4, or STAR is shown. Nuclei were counterstained with DAPI. The color of each marker is indicated above the images. G; germ cells. S; Sertoli cells. L; Leydig cells. MAFB was specifically detected in Leydig cells and Sertoli cells. (B) Localization of MAFB in adult mouse testes. Double immunostaining of MAFB with E-cadherin (ECAD), KIT, SCP3, PNA Lectin, or vimentin is shown. Nuclei were counterstained with DAPI. The color of each marker is indicated above the images. All seminiferous tubules shown represent stage VII. US; undifferentiated spermatogonia. DS; differentiated spermatogonia. P; pachytene spermatocytes. Sp; spermatids. S; Sertoli cells. L; Leydig cells. MAFB was specifically detected in Leydig cells, Sertoli cells, and pachytene spermatocytes. Image collected and cropped by CiteAb from the following publication (https://dx.plos.org/10.1371/journal.pone.0190800), licensed under a CC-BY license. Not internally tested by R&D Systems.Detection of Mouse E-Cadherin by Immunocytochemistry/Immunofluorescence
Testis morphogenesis of Mafb KO embryos developed normally.(A) Histological section of WT and KO E18.5 testes stained with HE. No morphological alteration or distribution was detectable. (B and C) Counts of Leydig and Sertoli cells from E18.5 WT and KO testes. Embryonic testes (n = 3 per genotype) were sectioned, and 4 sections for each gonad were randomly selected and stained with the germ cell marker E-cadherin (green) together with either STAR (red) or GATA4 (red). Numbers of STAR-positive cells outside seminiferous tubules (Leydig cells) or GATA4-positive cells inside the tubules (Sertoli cells) were counted per unit area. The values are the mean±S.D. *P<0.05. There was no significant difference between WT and KO cell counts. (D) The expression of genes involved in testes development and function. mRNA expression of the gene markers encoding for PGCs (Oct4), Leydig cells (Cyp17a1, StAR, Insl3, Hsd3b1, and Cyp11a1), and Sertoli cells (Amh, Sox9, and WT1) was determined by qRT-PCR in E18.5 WT and Mafb KO testes. Gene expression levels were normalized to Hprt. The bars represent the mean±SEM of five individuals. *P<0.05. Image collected and cropped by CiteAb from the following publication (https://dx.plos.org/10.1371/journal.pone.0190800), licensed under a CC-BY license. Not internally tested by R&D Systems.Detection of E-Cadherin in mouse intestine.
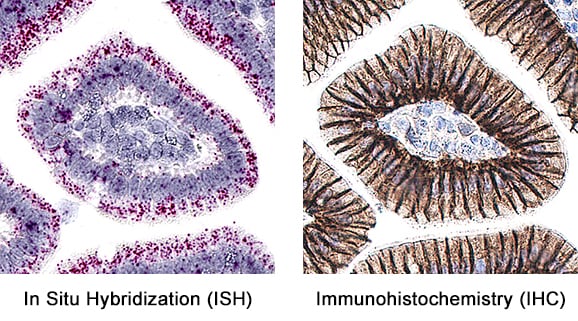
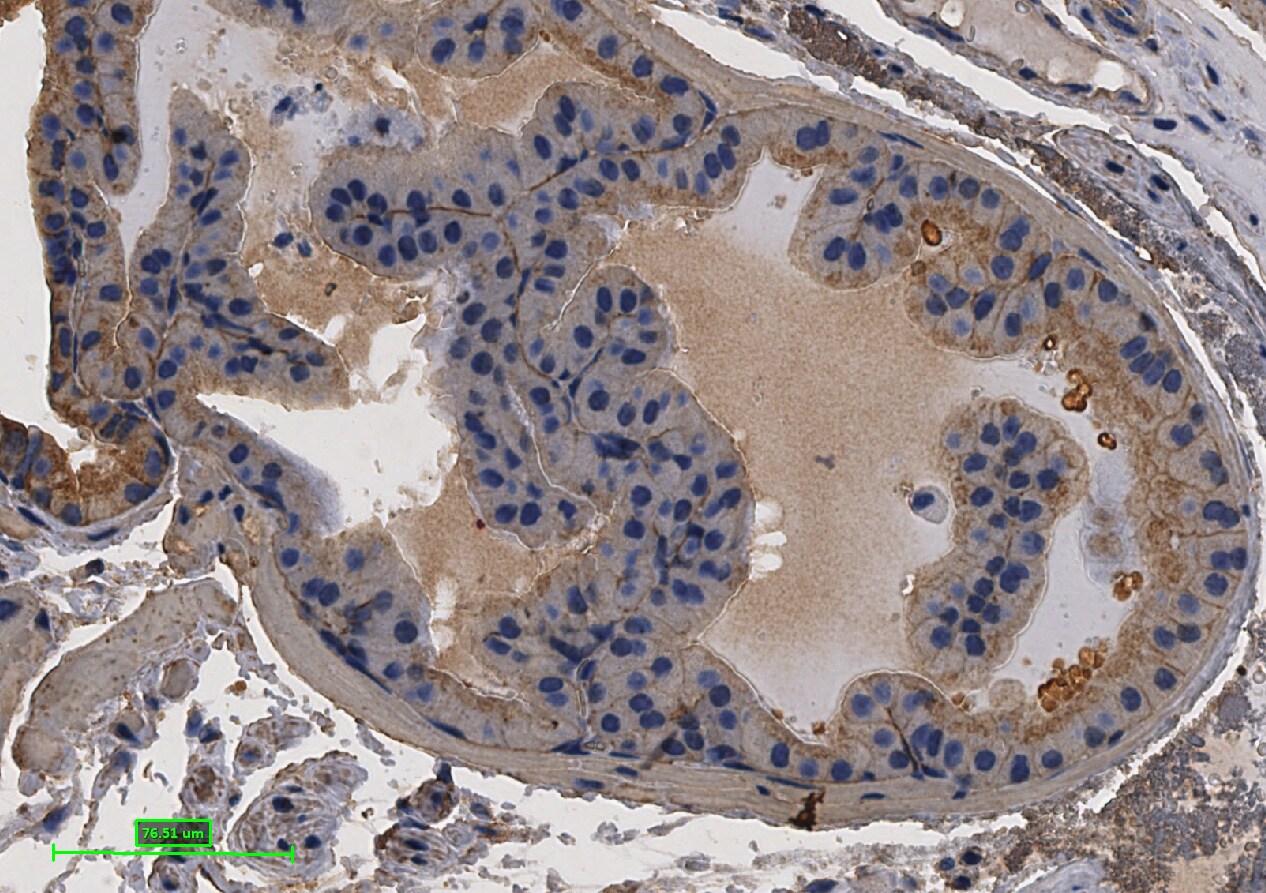
Formalin-fixed paraffin-embedded tissue sections of mouse intestine were probed for E-Cadherin mRNA (ACD RNAScope Probe, catalog # 408651; Fast Red chromogen, ACD catalog # 322360). Adjacent tissue section was processed for immunohistochemistry using goat anti-mouse E-Cadherin polyclonal antibody (R&D Systems catalog # AF748) at 0.5ug/mL with overnight incubation at 4 degrees Celsius followed by incubation with anti-goat IgG VisUCyte HRP Polymer Antibody (Catalog # VC004) and DAB chromogen (yellow-brown). Tissue was counterstained with hematoxylin (blue). Specific staining was localized to glandular cells.Detection of Mouse E-Cadherin by Immunohistochemistry-Paraffin
SARS-CoV-2 infection of the OE and brain in hACE2fl mice.(A, B) Immunohistochemistry of SARS-CoV-2 nucleocapsid and epithelial cell E-cadherin in the RE, OE, and OB 2 and 5–6 days after infection of hACE2fl/y and ShhCre/+; hACE2fl/y mice. Arrowheads indicate sites of viral nucleocapsid detection. Representative of N = 4 animals per genotype and time point. Scale bars 100 μm. (C, D) Immunohistochemistry of SARS-CoV-2 nucleocapsid, neuronal NeuN, and glial cell GFAP in the cerebral cortex (Co) 2 and 5–6 days after infection. Arrowheads indicate sites of GFAP+ reactive gliosis. Arrows indicate nucleocapsid colocalization with NeuN staining. Representative of N = 4 animals per genotype and time point. Scale bars 100 μm top, 50 μm bottom. (E) Diagram of the mouse nasal cavity and cranial anatomy. (F) In situ hybridization detection of SARS-CoV-2 mRNA 5 days postinfection reveals virus in the OB and cerebral cortex of the brain, but not the OE of the nose. Scale bar 250 μm. Note: Images in each panel were taken at lower or higher magnification from the same tissue section respective to genotype and highlight different anatomical regions. OB, olfactory bulb of the brain; OE, olfactory epithelium; RE, respiratory epithelium; SARS-CoV-2, Severe Acute Respiratory Syndrome Coronavirus 2. Image collected and cropped by CiteAb from the following open publication (https://pubmed.ncbi.nlm.nih.gov/36745682), licensed under a CC-BY license. Not internally tested by R&D Systems.Immunofluorescent Staining of Adult Stem Cell-derived Human Descending Colon Organoids.
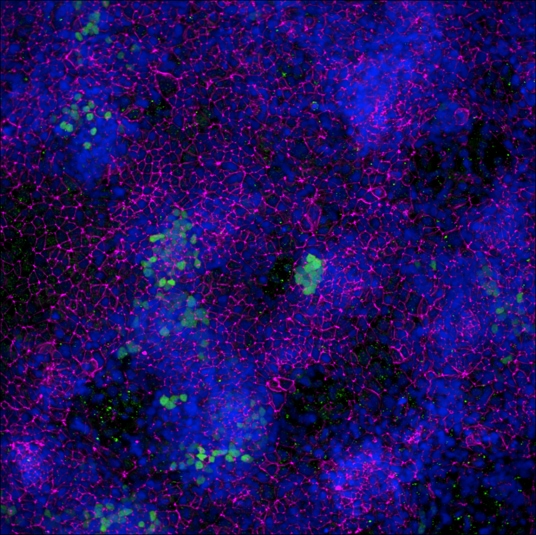
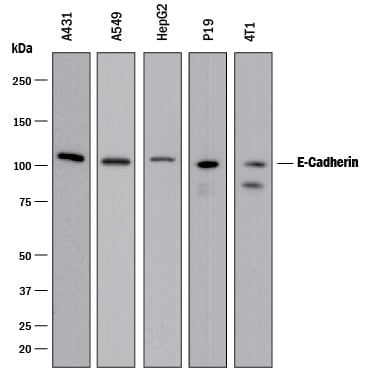
Adult stem cells isolated from human descending colon were cultured following the steps detailed in the human intestinal organoid culture protocol. (A) The organoids were fixed and stained with a Mouse Anti-Human MUC2 Monoclonal Antibody (Novus Biologicals; Catalog # NBP2-44431; green) to visualize intestinal goblet cells and counterstained with a Goat Anti-Human/Mouse E-Cadherin Antigen Affinity-purified Polyclonal Antibody (Catalog # AF748; red) and DAPI (Catalog # 5748; blue). The image shown was taken at 10x magnification. (B) The organoids were fixed and stained with a Mouse Anti-Human Chromogranin A Monoclonal Antibody (Catalog # MAB90981; green) to visualize enteroendocrine cells and counterstained with a Goat Anti-Human/Mouse E-Cadherin Antigen Affinity-purified Polyclonal Antibody (Catalog # AF748; red) and DAPI (Catalog # 5748; blue). The image shown was taken at 20x magnification.Detection of E-Cadherin by Western Blot
Characterisation of mammary organoids. (A) Endoxifen (endox) mediated knockout of E-cadherin and/or Tp53 in mammary organoids was detected utilizing western blotting. (B) Relative expression of E-cadherin and Tp53 protein in WT, Cdh1−/− and Cdh1−/−Tp53−/− mammary organoids. (C) 20× Brightfield and RFP channel images of mammary organoids induced with endoxifen. Image collected and cropped by CiteAb from the following open publication (https://pubmed.ncbi.nlm.nih.gov/35406381), licensed under a CC-BY license. Not internally tested by R&D Systems.Applications for Human/Mouse E‑Cadherin Antibody
Application
Recommended Usage
CyTOF-ready
Ready to be labeled using established conjugation methods. No BSA or other carrier proteins that could interfere with conjugation.
Dual RNAscope ISH-IHC Compatible
5-15 µg/mL
Sample: Immersion fixed paraffin-embedded sections of mouse intestine
Sample: Immersion fixed paraffin-embedded sections of mouse intestine
Flow Cytometry
0.25 µg/106 cells
Sample: D3 mouse embryonic stem cell line
Sample: D3 mouse embryonic stem cell line
Immunocytochemistry
5-15 µg/mL
Sample: Immersion fixed D3 mouse embryonic stem cell line and immersion fixed mouse intestinal organoids
Sample: Immersion fixed D3 mouse embryonic stem cell line and immersion fixed mouse intestinal organoids
Immunohistochemistry
5-15 µg/mL
Sample: Perfusion fixed frozen sections of mouse spinal cord and mouse skin
Sample: Perfusion fixed frozen sections of mouse spinal cord and mouse skin
Simple Western
5 µg/mL
Sample: 4T1 mouse breast cancer cell line, P19 mouse embryonal carcinoma cell line, A431 human epithelial carcinoma cell line, and MCF‑7 human breast cancer cell line
Sample: 4T1 mouse breast cancer cell line, P19 mouse embryonal carcinoma cell line, A431 human epithelial carcinoma cell line, and MCF‑7 human breast cancer cell line
Western Blot
0.5 µg/mL
Sample: A431 human epithelial carcinoma cell line, A549 human lung carcinoma cell line, HepG2 human hepatocellular carcinoma cell line, P19 mouse embryonal carcinoma cell line, and 4T1 mouse breast cancer cell line
Sample: A431 human epithelial carcinoma cell line, A549 human lung carcinoma cell line, HepG2 human hepatocellular carcinoma cell line, P19 mouse embryonal carcinoma cell line, and 4T1 mouse breast cancer cell line
Reviewed Applications
Read 12 reviews rated 4.6 using AF748 in the following applications:
Flow Cytometry Panel Builder
Bio-Techne Knows Flow Cytometry
Save time and reduce costly mistakes by quickly finding compatible reagents using the Panel Builder Tool.
Advanced Features
- Spectra Viewer - Custom analysis of spectra from multiple fluorochromes
- Spillover Popups - Visualize the spectra of individual fluorochromes
- Antigen Density Selector - Match fluorochrome brightness with antigen density
Formulation, Preparation, and Storage
Purification
Antigen Affinity-purified
Reconstitution
Reconstitute at 0.2 mg/mL in sterile PBS. For liquid material, refer to CoA for concentration.
Loading...
Formulation
Lyophilized from a 0.2 μm filtered solution in PBS with Trehalose. See Certificate of Analysis for details.
*Small pack size (-SP) is supplied either lyophilized or as a 0.2 µm filtered solution in PBS.
*Small pack size (-SP) is supplied either lyophilized or as a 0.2 µm filtered solution in PBS.
Shipping
Lyophilized product is shipped at ambient temperature. Liquid small pack size (-SP) is shipped with polar packs. Upon receipt, store immediately at the temperature recommended below.
Stability & Storage
Use a manual defrost freezer and avoid repeated freeze-thaw cycles.
- 12 months from date of receipt, -20 to -70 °C as supplied.
- 1 month, 2 to 8 °C under sterile conditions after reconstitution.
- 6 months, -20 to -70 °C under sterile conditions after reconstitution.
Calculators
Background: E-Cadherin
References
- Bussemakers, M.J.G. et al. (1993) Mol. Biol. Reports 17:123.
- Overduin, M. et al. (1995) Science 267:386.
- Takeichi, M. (1991) 251:1451.
Alternate Names
Arc-1, CAD1, Cadherin-1, CD324, CDH1, Cell-CAM120/80, ECAD, ECadherin, L-CAM, Uvomorulin
Gene Symbol
CDH1
UniProt
Additional E-Cadherin Products
Product Documents for Human/Mouse E‑Cadherin Antibody
Certificate of Analysis
To download a Certificate of Analysis, please enter a lot or batch number in the search box below.
Note: Certificate of Analysis not available for kit components.
Product Specific Notices for Human/Mouse E‑Cadherin Antibody
For research use only
Citations for Human/Mouse E‑Cadherin Antibody
Customer Reviews for Human/Mouse E‑Cadherin Antibody (12)
4.6 out of 5
12 Customer Ratings
Have you used Human/Mouse E‑Cadherin Antibody?
Submit a review and receive an Amazon gift card!
$25/€18/£15/$25CAN/¥2500 Yen for a review with an image
$10/€7/£6/$10CAN/¥1110 Yen for a review without an image
Submit a review
Customer Images
Showing
1
-
5 of
12 reviews
Showing All
Filter By:
-
Application: Immunocytochemistry/ImmunofluorescenceSample Tested: Human Lung Progenitor Cells Derived from Embryonic Stem CellSpecies: HumanVerified Customer | Posted 07/10/20251:200 dilution for 1 hour at RT
-
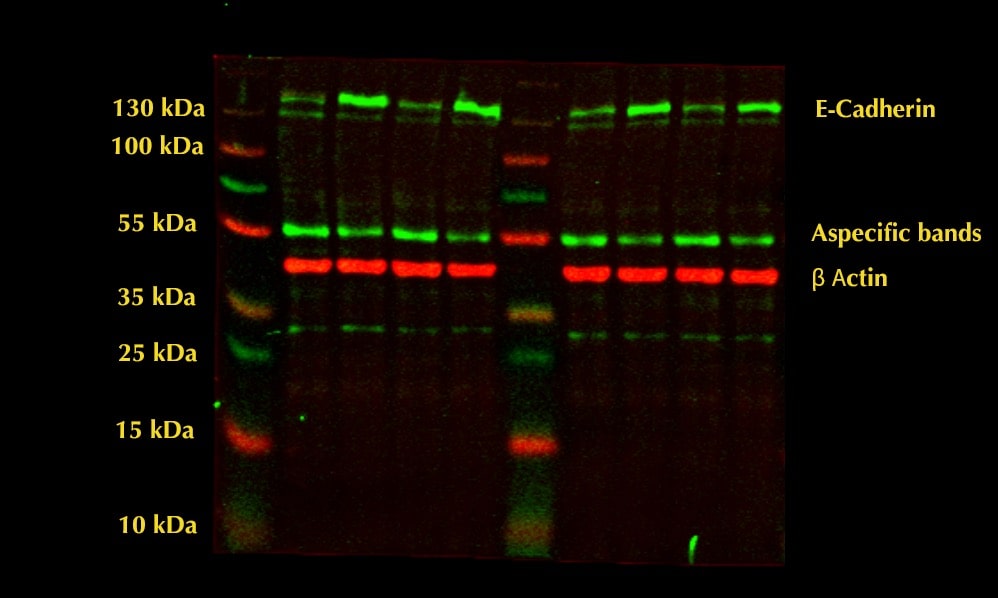
Application: Western BlotSample Tested: HepG2 human hepatocellular carcinoma cell lineSpecies: HumanVerified Customer | Posted 11/29/2022HepG2 40 ug cell lysate loaded. Aspecific bands appeared at 55 kDa
-
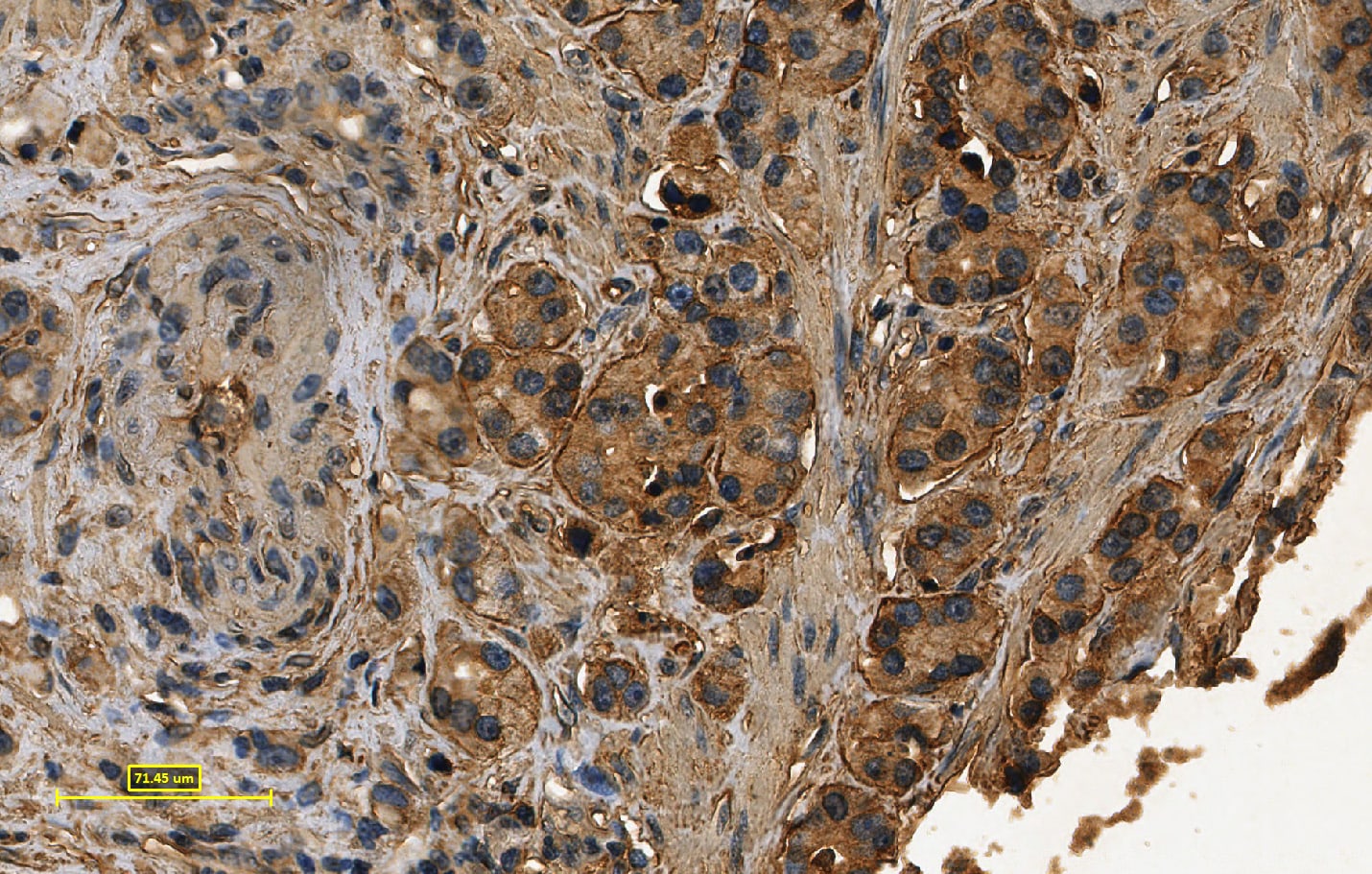
Application: ImmunohistochemistrySample Tested: Prostate tissueSpecies: HumanVerified Customer | Posted 04/01/2022
-
Application: ImmunohistochemistrySample Tested: Mice prostate tissueSpecies: Mice prostate tissue and MouseVerified Customer | Posted 04/01/2022
-
Application: Immunocytochemistry/ImmunofluorescenceSample Tested: Skin tissueSpecies: HumanVerified Customer | Posted 09/22/2021
-
Application: Immunocytochemistry/ImmunofluorescenceSample Tested: Embryonic lungSpecies: MouseVerified Customer | Posted 03/21/2019
-
Application: Immunocytochemistry/ImmunofluorescenceSample Tested: RT4-D6P2T rat schwannoma cell lineSpecies: HumanVerified Customer | Posted 01/14/2019
-
Application: ImmunohistochemistrySample Tested: Tumor cell lyastesSpecies: MouseVerified Customer | Posted 04/13/2018
-
Application: Immunocytochemistry/ImmunofluorescenceSample Tested: Bladder cancer tissueSpecies: HumanVerified Customer | Posted 12/19/2017
-
Application: Immunocytochemistry/ImmunofluorescenceSample Tested: iPSC derived epithelial cellsSpecies: HumanVerified Customer | Posted 09/27/2017
-
Application: Western BlotSample Tested: 293T human embryonic kidney cell lineSpecies: HumanVerified Customer | Posted 07/31/2017
-
Application: Western BlotSample Tested: mouse primary brain cell culturesSpecies: MouseVerified Customer | Posted 04/21/2016Ab concentration: 0.1 ug/ml
There are no reviews that match your criteria.
Protocols
Find general support by application which include: protocols, troubleshooting, illustrated assays, videos and webinars.
- 7-Amino Actinomycin D (7-AAD) Cell Viability Flow Cytometry Protocol
- Antigen Retrieval Protocol (PIER)
- Antigen Retrieval for Frozen Sections Protocol
- Appropriate Fixation of IHC/ICC Samples
- Cellular Response to Hypoxia Protocols
- Chromogenic IHC Staining of Formalin-Fixed Paraffin-Embedded (FFPE) Tissue Protocol
- Chromogenic Immunohistochemistry Staining of Frozen Tissue
- ClariTSA™ Fluorophore Kits
- Detection & Visualization of Antibody Binding
- Extracellular Membrane Flow Cytometry Protocol
- Flow Cytometry Protocol for Cell Surface Markers
- Flow Cytometry Protocol for Staining Membrane Associated Proteins
- Flow Cytometry Staining Protocols
- Flow Cytometry Troubleshooting Guide
- Fluorescent IHC Staining of Frozen Tissue Protocol
- Graphic Protocol for Heat-induced Epitope Retrieval
- Graphic Protocol for the Preparation and Fluorescent IHC Staining of Frozen Tissue Sections
- Graphic Protocol for the Preparation and Fluorescent IHC Staining of Paraffin-embedded Tissue Sections
- Graphic Protocol for the Preparation of Gelatin-coated Slides for Histological Tissue Sections
- ICC Cell Smear Protocol for Suspension Cells
- ICC Immunocytochemistry Protocol Videos
- ICC for Adherent Cells
- IHC Sample Preparation (Frozen sections vs Paraffin)
- ISH-IHC Protocol for Chromogenic Detection on Formalin Fixed Paraffin Embedded (FFPE) Tissue
- Immunocytochemistry (ICC) Protocol
- Immunocytochemistry Troubleshooting
- Immunofluorescence of Organoids Embedded in Cultrex Basement Membrane Extract
- Immunofluorescent IHC Staining of Formalin-Fixed Paraffin-Embedded (FFPE) Tissue Protocol
- Immunohistochemistry (IHC) and Immunocytochemistry (ICC) Protocols
- Immunohistochemistry Frozen Troubleshooting
- Immunohistochemistry Paraffin Troubleshooting
- Intracellular Flow Cytometry Protocol Using Alcohol (Methanol)
- Intracellular Flow Cytometry Protocol Using Detergents
- Intracellular Nuclear Staining Flow Cytometry Protocol Using Detergents
- Intracellular Staining Flow Cytometry Protocol Using Alcohol Permeabilization
- Intracellular Staining Flow Cytometry Protocol Using Detergents to Permeabilize Cells
- Preparing Samples for IHC/ICC Experiments
- Preventing Non-Specific Staining (Non-Specific Binding)
- Primary Antibody Selection & Optimization
- Propidium Iodide Cell Viability Flow Cytometry Protocol
- Protocol for Heat-Induced Epitope Retrieval (HIER)
- Protocol for Liperfluo
- Protocol for Making a 4% Formaldehyde Solution in PBS
- Protocol for VisUCyte™ HRP Polymer Detection Reagent
- Protocol for the Characterization of Human Th22 Cells
- Protocol for the Characterization of Human Th9 Cells
- Protocol for the Fluorescent ICC Staining of Cell Smears - Graphic
- Protocol for the Fluorescent ICC Staining of Cultured Cells on Coverslips - Graphic
- Protocol for the Preparation & Fixation of Cells on Coverslips
- Protocol for the Preparation and Chromogenic IHC Staining of Frozen Tissue Sections
- Protocol for the Preparation and Chromogenic IHC Staining of Frozen Tissue Sections - Graphic
- Protocol for the Preparation and Chromogenic IHC Staining of Paraffin-embedded Tissue Sections
- Protocol for the Preparation and Chromogenic IHC Staining of Paraffin-embedded Tissue Sections - Graphic
- Protocol for the Preparation and Fluorescent ICC Staining of Cells on Coverslips
- Protocol for the Preparation and Fluorescent ICC Staining of Non-adherent Cells
- Protocol for the Preparation and Fluorescent ICC Staining of Stem Cells on Coverslips
- Protocol for the Preparation and Fluorescent IHC Staining of Frozen Tissue Sections
- Protocol for the Preparation and Fluorescent IHC Staining of Paraffin-embedded Tissue Sections
- Protocol for the Preparation of Gelatin-coated Slides for Histological Tissue Sections
- Protocol for the Preparation of a Cell Smear for Non-adherent Cell ICC - Graphic
- Protocol: Annexin V and PI Staining by Flow Cytometry
- Protocol: Annexin V and PI Staining for Apoptosis by Flow Cytometry
- R&D Systems Quality Control Western Blot Protocol
- TUNEL and Active Caspase-3 Detection by IHC/ICC Protocol
- The Importance of IHC/ICC Controls
- Troubleshooting Guide: Fluorokine Flow Cytometry Kits
- Troubleshooting Guide: Immunohistochemistry
- Troubleshooting Guide: Western Blot Figures
- Western Blot Conditions
- Western Blot Protocol
- Western Blot Protocol for Cell Lysates
- Western Blot Troubleshooting
- Western Blot Troubleshooting Guide
- View all Protocols, Troubleshooting, Illustrated assays and Webinars
Loading...
Associated Pathways