CXCL12, also known as SCYB12, PBSF and SDF-1 beta, is an 8.3 kDa, heparin-binding member of the CXC (or alpha-) family of chemokines (1, 2). Feline CXCL12( beta ) is synthesized as a 93 amino acid (aa) precursor that contains a 21 aa signal sequence and a 72 aa mature region (3). The mature molecule exhibits a typical three antiparallel beta -strand chemokine-like fold. There are no potential N-linked glycosylation sites. N-terminal aa’s 1 - 8 form a receptor binding site, while aa’s 1 and 2 (Lys-Pro) are involved in receptor activation (4). The C-terminus is likely associated with heparin binding (5). SDF-1 beta circulates and undergoes proteolytic processing. CD26 will remove the first two N-terminal amino acids, possibly creating a reduced-activity chemokine (5, 6). In addition to the beta -isoform, alternate splicing of the feline SDF-1 gene generates an alpha -isoform. The alpha isoform is identical to SDF-1 beta, but shorter by four aa’s at the C-terminus (3). Although alpha - and beta -isoforms show similar activity, SDF-1 alpha is differentially processed, and different cells secrete the two isoforms (5, 7). Mature feline SDF-1 beta is 96%, 97% and 100% aa identical to rat, mouse and human SDF-1 beta, respectively. Human (and by inference, feline) SDF-1 is active on mouse cells. SDF-1 alpha and beta are reported to be monomers at neutral pH and physiologic ionic strength (4). SDF-1 alpha is also reported to form dimers in the presence of heparansulfate (8). On the cell surface, this may well facilitate SDF-1 interaction with its two receptors, CXCR4 and syndecan-4 (9). Heparin sulfate is known to protect SDF-1 from proteolysis, and CXCR4 exists constitutively as a dimer (9 - 11). Among its many functions, CXCL12 is known to influence lymphopoiesis, regulate patterning and cell number of neural progenitors, and promote angiogenesis (12, 13). It also enhances the survival of myeloid progenitor cells.
Human CXCL12/SDF‑1 beta Antibody
R&D Systems | Catalog # AF-351-NA

Key Product Details
Species Reactivity
Validated:
Human
Cited:
Human
Applications
Validated:
Western Blot, Neutralization
Cited:
Neutralization
Label
Unconjugated
Antibody Source
Polyclonal Goat IgG
Loading...
Product Specifications
Immunogen
E. coli-derived recombinant human CXCL12/SDF‑1 beta
Lys22-Met93
Accession # P48061
Lys22-Met93
Accession # P48061
Specificity
Detects human CXCL12/SDF‑1 beta in direct ELISAs and Western blots. Neutralizes 60‑80% of the biological activity of CXCL12/SDF-1 beta and does not neutralize the biological activity of SDF‑1 alpha. In Western blots, less than 5% cross-reactivity with recombinant human SDF-1 alpha is observed.
Clonality
Polyclonal
Host
Goat
Isotype
IgG
Endotoxin Level
<0.10 EU per 1 μg of the antibody by the LAL method.
Scientific Data Images for Human CXCL12/SDF‑1 beta Antibody
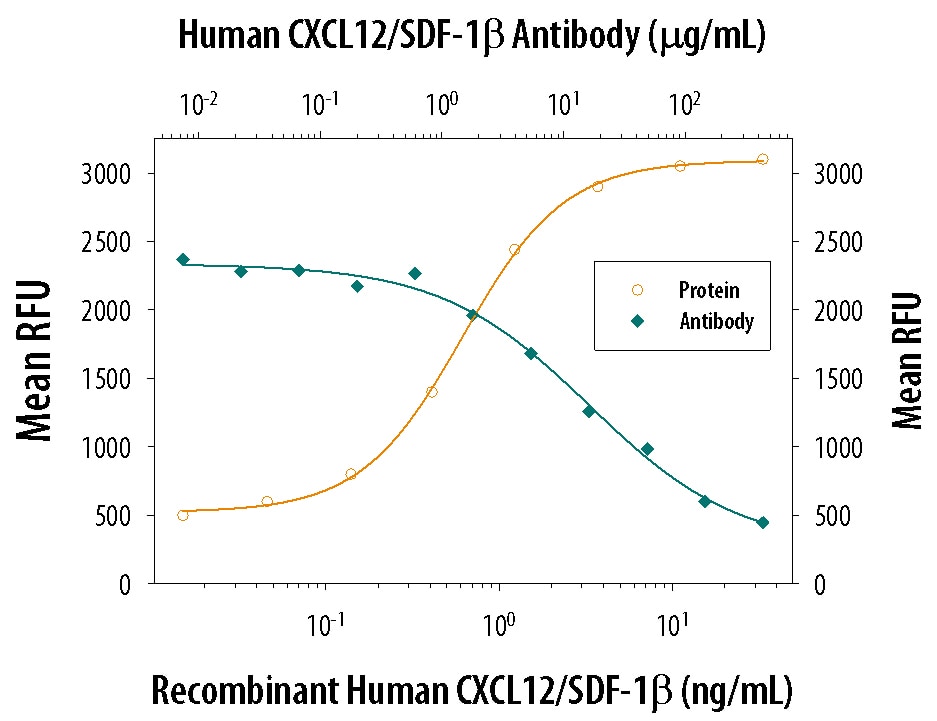
Chemotaxis Induced by CXCL12/SDF‑1 beta and Neutral-ization by Human CXCL12/ SDF‑1 beta Antibody.
Recombinant Human/Feline CXCL12/SDF-1 beta chemo-attracts the BaF3 mouse pro-B cell line transfected with human CXCR4 in a dose-dependent manner (orange line). The amount of cells that migrated through to the lower chemotaxis chamber was measured by Resazurin (Catalog # AR002). Chemotaxis elicited by Recombinant Human/Feline CXCL12/ SDF-1 beta (10 ng/mL) is neutral-ized (green line) by increasing concentrations of Goat Anti-Human CXCL12/SDF-1 beta Anti-gen Affinity-purified Poly-clonal Antibody (Catalog # AF-351-NA). The ND50 is typically 10-30 µg/mL.Applications for Human CXCL12/SDF‑1 beta Antibody
Application
Recommended Usage
Western Blot
0.1 µg/mL
Sample: Recombinant Human/Feline CXCL12/SDF‑1 beta aa 19-93 (Catalog # 2716-SD)
Sample: Recombinant Human/Feline CXCL12/SDF‑1 beta aa 19-93 (Catalog # 2716-SD)
Neutralization
Measured by its ability to neutralize CXCL12/SDF‑1 beta -induced chemotaxis in the BaF3 mouse pro‑B cell line transfected with human CXCR4. The Neutralization Dose (ND50) is typically 10-30 µg/mL in the presence of 10 ng/mL Recombinant Human/Feline CXCL12/SDF‑1 beta.
Formulation, Preparation, and Storage
Purification
Antigen Affinity-purified
Reconstitution
Reconstitute at 0.2 mg/mL in sterile PBS. For liquid material, refer to CoA for concentration.
Loading...
Formulation
Lyophilized from a 0.2 μm filtered solution in PBS with Trehalose. *Small pack size (SP) is supplied either lyophilized or as a 0.2 µm filtered solution in PBS.
Shipping
Lyophilized product is shipped at ambient temperature. Liquid small pack size (-SP) is shipped with polar packs. Upon receipt, store immediately at the temperature recommended below.
Stability & Storage
Use a manual defrost freezer and avoid repeated freeze-thaw cycles.
- 12 months from date of receipt, -20 to -70 °C as supplied.
- 1 month, 2 to 8 °C under sterile conditions after reconstitution.
- 6 months, -20 to -70 °C under sterile conditions after reconstitution.
Calculators
Background: CXCL12/SDF-1 beta
References
- Zlotnik, A. and O. Yoshie (2000) Immunity 12:121.
- Rollins, B.J. (1997) Blood 90:909.
- Nishimura, Y. et al. (1998) Eur. J. Immunogenet. 25:303.
- Crump, M.P. et al. (1997) EMBO J. 16:6996.
- Sierra, M.D. L. et al. (2004) Blood 103:2452.
- Davis, D.A. et al. (2005) Blood 105:4561.
- Stumm, R.K. et al. (2002) J. Neurosci. 22:5865.
- Veldkamp, C.T. et al. (2005) Protein Sci. 14:1071.
- Charnaux, N. et al. (2005) FEBS J. 272:1937.
- Percherancier, Y. et al. (2005) J. Biol. Chem. 280:9895.
- Babcock, G.J. et al. (2003) J. Biol. Chem. 278:3378.
- Klein, R.S. et al. (2004) Trends Immunol. 25:306.
- Salcedo, R. and J.J. Oppenheim (2003) Microcirculation 10:359.
- Broxmeyer, H.E. et al. (2003) J. Leukoc. Biol. 73:630.
Alternate Names
SDF1 beta, SDF1b
Gene Symbol
CXCL12
UniProt
Additional CXCL12/SDF-1 beta Products
Product Documents for Human CXCL12/SDF‑1 beta Antibody
Certificate of Analysis
To download a Certificate of Analysis, please enter a lot or batch number in the search box below.
Note: Certificate of Analysis not available for kit components.
Product Specific Notices for Human CXCL12/SDF‑1 beta Antibody
For research use only
Related Research Areas
Citations for Human CXCL12/SDF‑1 beta Antibody
Customer Reviews for Human CXCL12/SDF‑1 beta Antibody
There are currently no reviews for this product. Be the first to review Human CXCL12/SDF‑1 beta Antibody and earn rewards!
Have you used Human CXCL12/SDF‑1 beta Antibody?
Submit a review and receive an Amazon gift card!
$25/€18/£15/$25CAN/¥2500 Yen for a review with an image
$10/€7/£6/$10CAN/¥1110 Yen for a review without an image
Submit a review
Protocols
Find general support by application which include: protocols, troubleshooting, illustrated assays, videos and webinars.
- Cellular Response to Hypoxia Protocols
- R&D Systems Quality Control Western Blot Protocol
- Troubleshooting Guide: Western Blot Figures
- Western Blot Conditions
- Western Blot Protocol
- Western Blot Protocol for Cell Lysates
- Western Blot Troubleshooting
- Western Blot Troubleshooting Guide
- View all Protocols, Troubleshooting, Illustrated assays and Webinars
Loading...
Associated Pathways

