Key Product Details
Validated by
Biological Validation
Species Reactivity
Validated:
Human
Cited:
Human, Mouse, Rat, Rabbit
Applications
Validated:
Immunohistochemistry, Neutralization, Flow Cytometry, CyTOF-ready
Cited:
Immunohistochemistry, Immunohistochemistry-Paraffin, Immunohistochemistry-Frozen, Western Blot, Neutralization, Flow Cytometry, Immunocytochemistry, Immunoprecipitation, Binding Assay, ELISA Detection, Functional Assay
Label
Unconjugated
Antibody Source
Monoclonal Mouse IgG2B Clone # 44716
Loading...
Product Specifications
Immunogen
3T3 cells transfected with human CXCR4
Specificity
Detects human CXCR4. It will also react with cells expressing feline CXCR4 but not rat CXCR4.
Clonality
Monoclonal
Host
Mouse
Isotype
IgG2B
Endotoxin Level
<0.10 EU per 1 μg of the antibody by the LAL method.
Scientific Data Images for Human CXCR4 Antibody
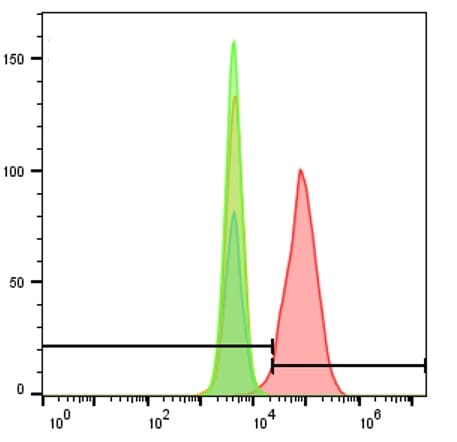
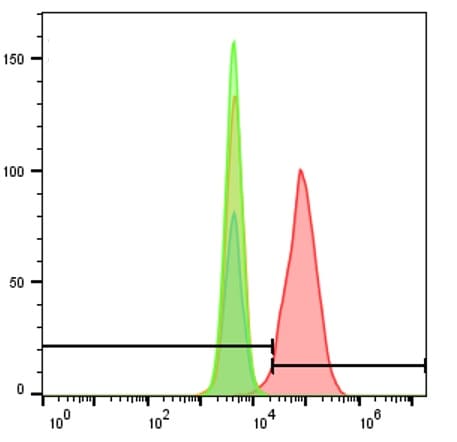
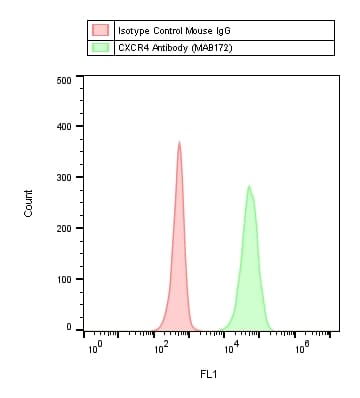
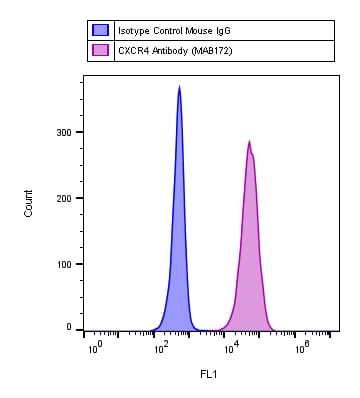
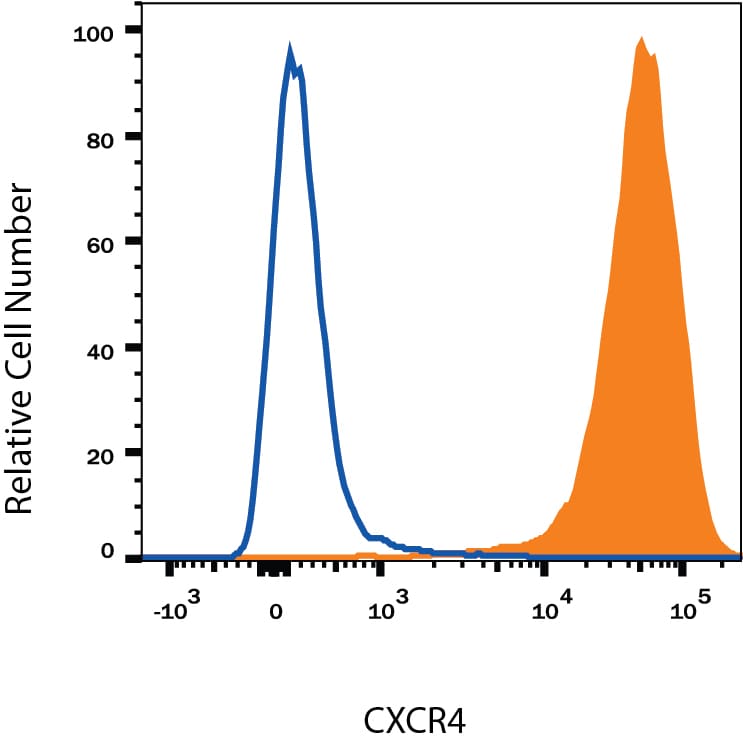
Detection of CXCR4 in Jurkat Human Cell Line by Flow Cytometry.
Jurkat human acute T cell leukemia cell line was stained with Mouse Anti-Human CXCR4 Monoclonal Antibody (Catalog # MAB172, filled histogram) or isotype control antibody (Catalog # MAB004, open histogram), followed by Allophycocyanin-conjugated Anti-Mouse IgG Secondary Antibody (Catalog # F0101B). View our protocol for Staining Membrane-associated Proteins.CXCR4 in Human Lymph Node.
CXCR4 was detected in immersion fixed paraffin-embedded sections of human lymph node using 15 µg/mL Human CXCR4 Monoclonal Antibody (Catalog # MAB172) overnight at 4 °C. Tissue was stained with the Anti-Mouse HRP-AEC Cell & Tissue Staining Kit (red; Catalog # CTS003) and counterstained with hematoxylin (blue). View our protocol for Chromogenic IHC Staining of Paraffin-embedded Tissue Sections.CXCR4 in Human Spleen.
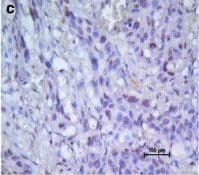
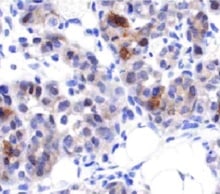
CXCR4 was detected in immersion fixed paraffin-embedded sections of human spleen using Mouse Anti-Human CXCR4 Monoclonal Antibody (Catalog # MAB172) at 15 µg/mL overnight at 4 °C. Before incubation with the primary antibody, tissue was subjected to heat-induced epitope retrieval using Antigen Retrieval Reagent-Basic (Catalog # CTS013). Tissue was stained using the Anti-Mouse HRP-DAB Cell & Tissue Staining Kit (brown; Catalog # CTS002) and counterstained with hematoxylin (blue). Specific staining was localized to cytoplasm and nuclei. View our protocol for Chromogenic IHC Staining of Paraffin-embedded Tissue Sections.Chemotaxis Induced by CXCL12/SDF‑1 alpha and Neutralization by Human CXCR4 Antibody.
Recombinant Human/Feline/Rhesus Macaque CXCL12/SDF-1a (Catalog # 350-NS) chemoattracts the BaF3 mouse pro-B cell line transfected with human CXCR4 in a dose-dependent manner (orange line). The amount of cells that migrated through to the lower chemotaxis chamber was measured by Resazurin (Catalog # AR002). Chemotaxis elicited by Recombinant Human/Feline/Rhesus Macaque CXCL12/SDF-1a (1 ng/mL) is neutralized (green line) by increasing concentrations of Mouse Anti-Human CXCR4 Monoclonal Antibody (Catalog # MAB172). The ND50 is typically 2.5-12 µg/mL.Detection of Human CXCR4 by Flow Cytometry
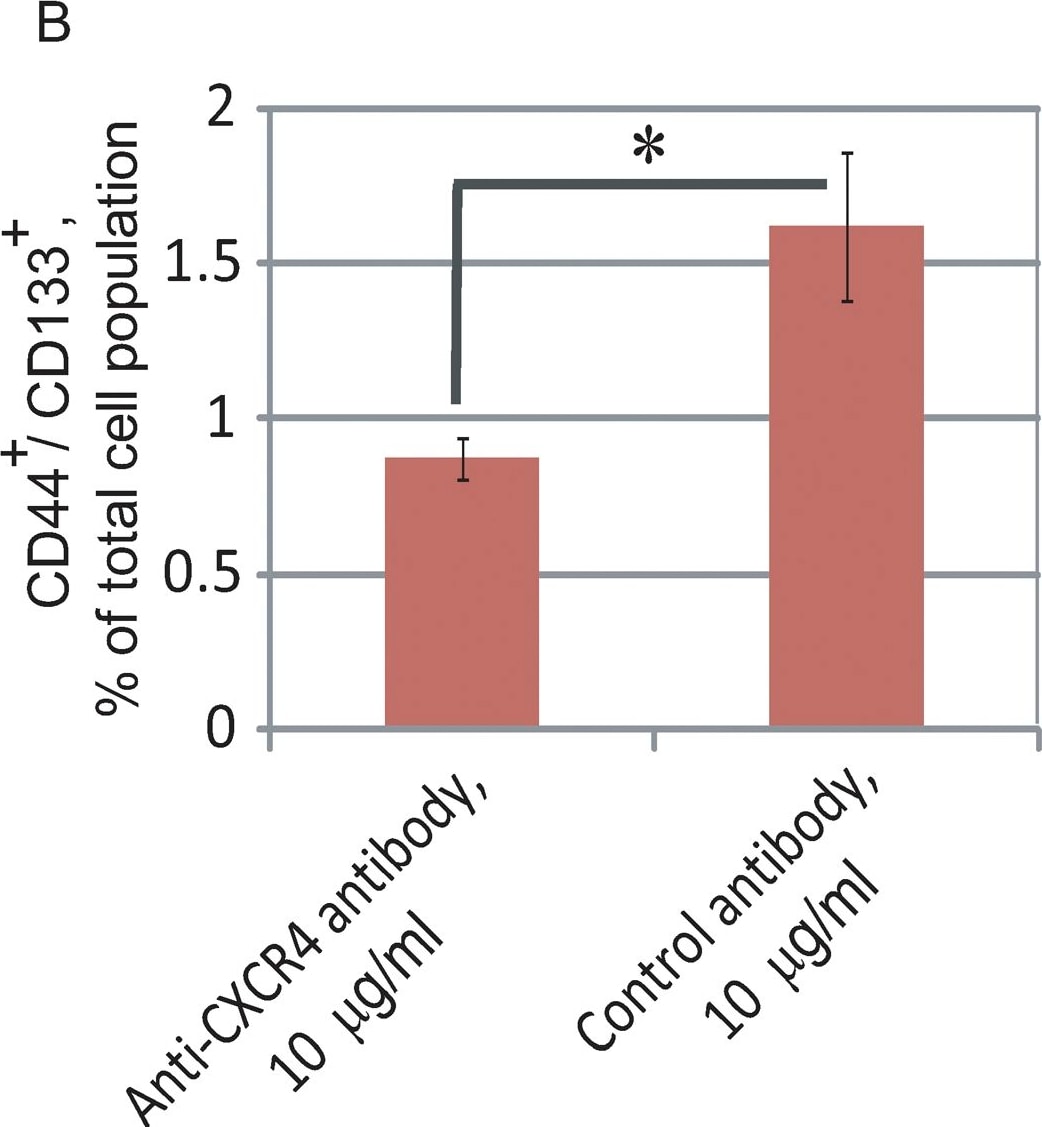
CXCR4 neutralization leads to attenuation of the CD44+/CD133+ prostate progenitor population.(A) DU145 cells were plated in 96-well low-attachment plates at 100 cells per well (5 replicates) and the spheres were grown in serum-free, EBM medium with supplements. The antibodies were replenished daily. Cells were imaged with an Acumen eX3 microplate cytometer and spheres were detected using image analysis software. The sphere size was measured by GFP intensity. The spheres were discriminated from cell debris using a Gaussian filter. The spheres included in the analysis are outlined in red and indicated by arrows. Representative data from one of two independent experiments is shown; *- p value<0.05. (B) Flow cytometry analysis revealed attenuation of CD44+/CD133+ population in DU145 cells treated with 10 µg/ml neutralizing anti-CXCR4 (mouse monoclonal IgG, clone 44716, R&D Systems) or 10 µg/ml control antibody (mouse IgG isotype control, Lifespan Bioscience Inc.) for 5 days. The cell were grown in medium supplemented with 2% FBS. Culture medium was refreshed every second day; *- p value<0.05. (C) Western blot analysis of DU145 cells treated with 10 µg/ml neutralizing anti-CXCR4 antibody for 5 days demonstrated downregulation of the PI3K/AKT pathway activation compared to the cells treated with 10 µg/ml control antibody. The cell were grown in medium supplemented with 2% FBS. Culture medium was refreshed every second day. (D) Preincubation of prostate cancer cells with neutralizing anti-CXCR4 antibody significantly delays tumor growth. 5×105 DU145 cells pretreated with neutralizing anti-CXCR4 or control antibody for 5 days were embedded in BD matrigel and injected s.c. into NOD/SCID mice.*- p value<0.01. Image collected and cropped by CiteAb from the following publication (https://dx.plos.org/10.1371/journal.pone.0031226), licensed under a CC-BY license. Not internally tested by R&D Systems.Detection of Human CXCR4 by Immunohistochemistry
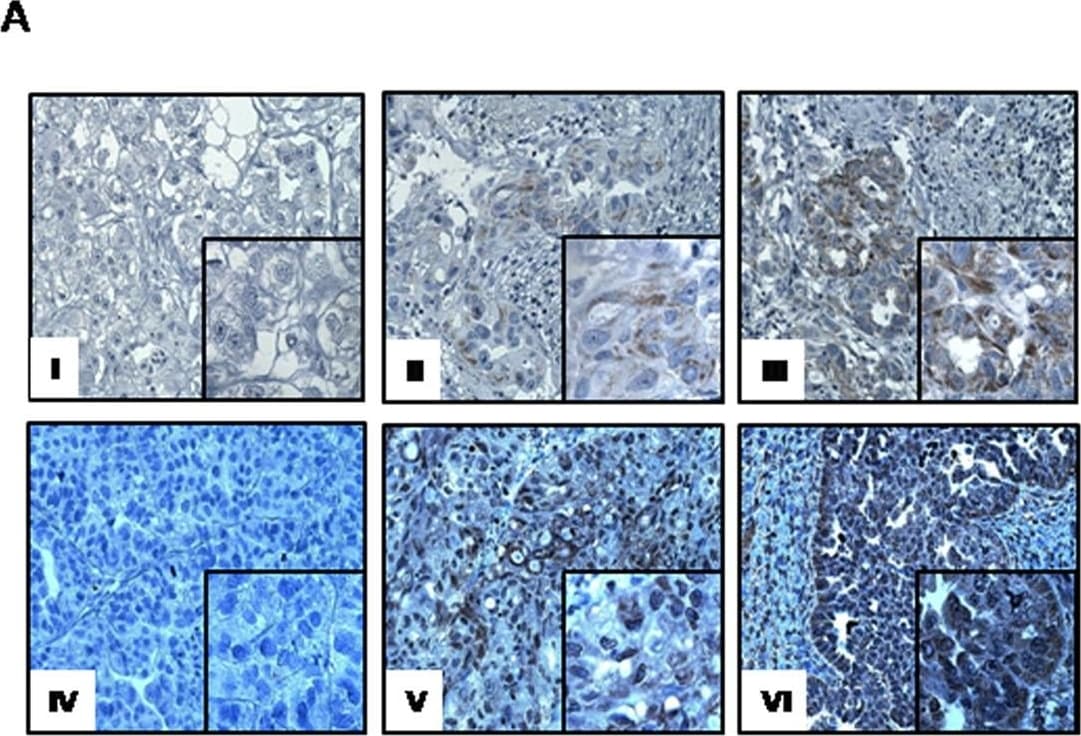
CXCR4 and CD133 are highly expressed in ovarian cancer patients. (A) Representative panel of immunohistochemistry for CD133 and CXCR4 expression (Magnification 200X; insert 400X). Non-homogeneous and focal cytoplasmic and membranous CD133 expression rated as negative (I), focally low (1–10% cancer cells) (II), focally high CD133 expression (>10% stained cancer cells) (III). Extensive homogeneous cytoplasmic and membranous, CXCR4 staining was reported rated as negative (IV) moderate (<50% of cancer cells) (V) and high expression (>50% of cancer cells) (VI). (B) QPCR analysis of CXCR4 and CD133 in freshly resected ovarian tumors and corresponding normal ovarian tissue. Image collected and cropped by CiteAb from the following publication (https://www.nature.com/articles/srep10357), licensed under a CC-BY license. Not internally tested by R&D Systems.Detection of Human CXCR4 by Immunohistochemistry
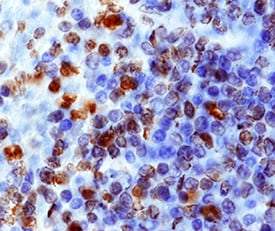
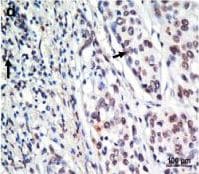
Immunohistochemical staining for MIF and CXCR4 in human esophageal carcinoma. Our data showed low expression levels of MIF (a) and CXCR4 (c) (X 400) and high expression levels of MIF (b) and CXCR4 (d), compared with the negative control (e) (X 400), in tumor tissues from patients with ESCC. The arrows point to the positive staining of tumor cells or TILs. Image collected and cropped by CiteAb from the following publication (https://translational-medicine.biomedcentral.com/articles/10.1186/1479-…), licensed under a CC-BY license. Not internally tested by R&D Systems.Detection of Human CXCR4 by Immunohistochemistry
Immunohistochemical staining for MIF and CXCR4 in human esophageal carcinoma. Our data showed low expression levels of MIF (a) and CXCR4 (c) (X 400) and high expression levels of MIF (b) and CXCR4 (d), compared with the negative control (e) (X 400), in tumor tissues from patients with ESCC. The arrows point to the positive staining of tumor cells or TILs. Image collected and cropped by CiteAb from the following publication (https://translational-medicine.biomedcentral.com/articles/10.1186/1479-…), licensed under a CC-BY license. Not internally tested by R&D Systems.Detection of Sheep CXCR4 by Immunohistochemistry
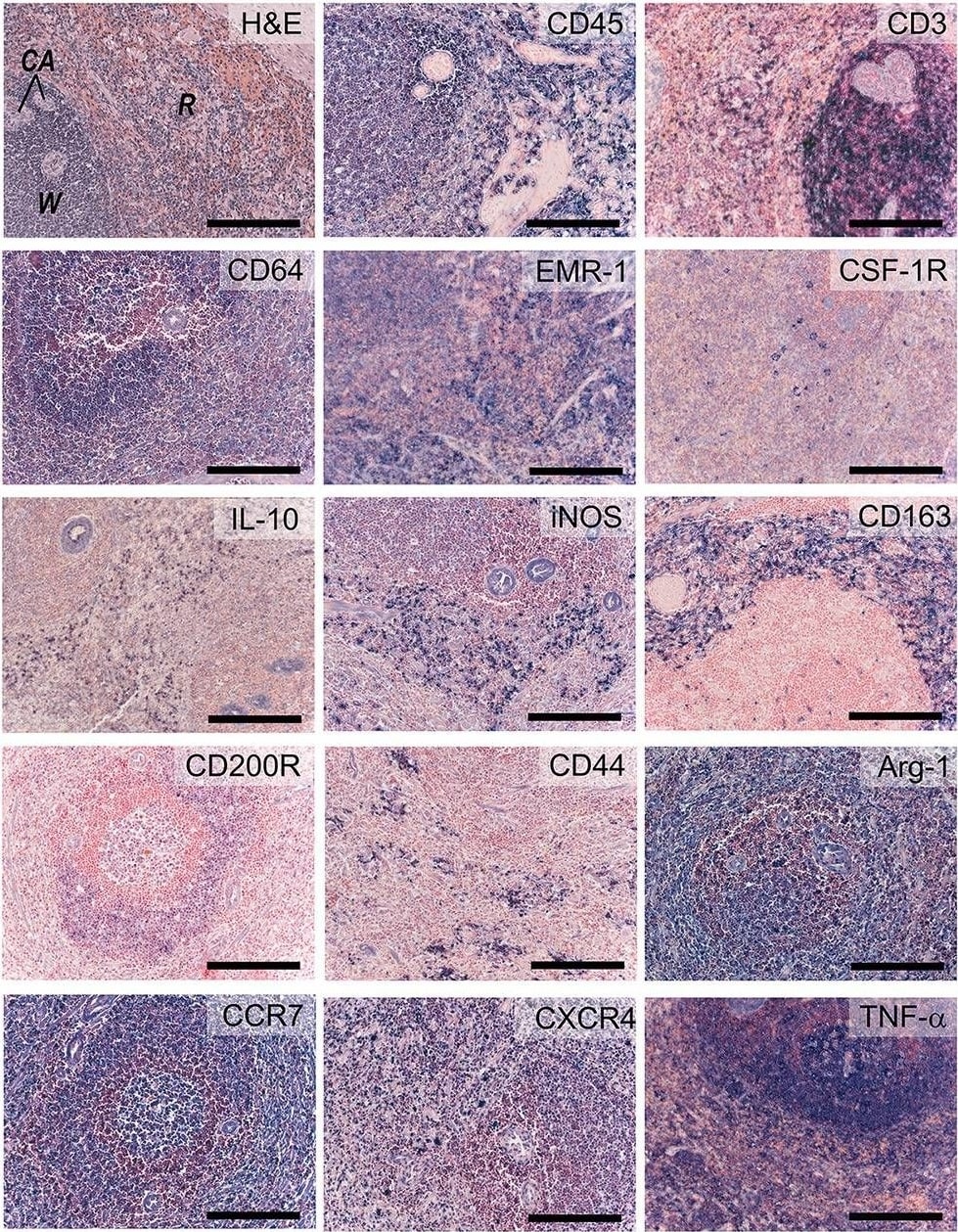
Inflammatory markers. Sections of activated ovine spleen, isolated from a sheep with endocarditis. Hematoxylin and eosin (H&E, top left) staining reveals the characteristic splenic red pulp (R) and white pulp (W), with the central arterioles (CA). Sections are stained with antibodies against CD45, CD3, CD64, EGF-like module-containing mucin-like hormone receptor-like 1 (EMR-1), colony stimulating factor-1 receptor (CSF-1R), interleukin-10 (IL-10), inducible nitric oxide synthase (iNOS), CD163, CD200R, CD44, Arginase-1 (Arg-1), CCR-7, CXCR4, and tumor necrosis factor-alpha (TNF-alpha ). Scale bars, 200 μm. Image collected and cropped by CiteAb from the following open publication (https://pubmed.ncbi.nlm.nih.gov/30159315), licensed under a CC-BY license. Not internally tested by R&D Systems.Detection of CXCR4 by Western Blot
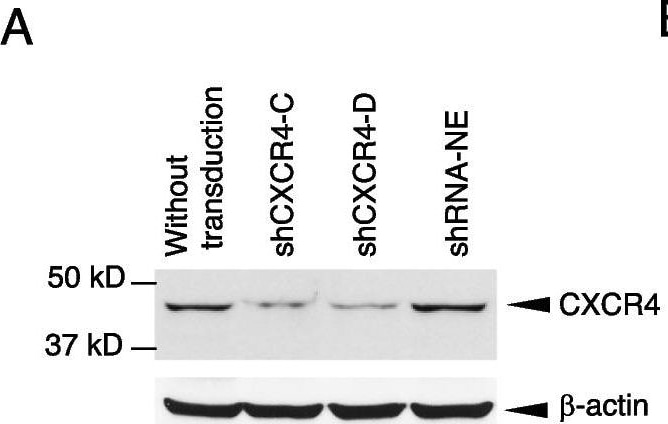
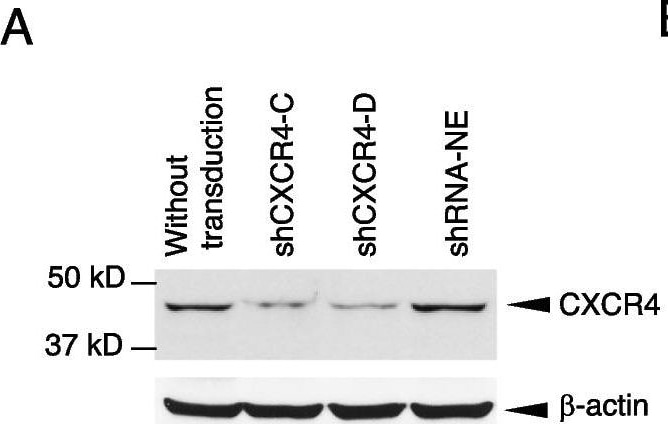
Knockdown of CXCR4 abrogates trastuzumab resistance. CXCR4 was silenced by specific shRNA in HCC1419 cells (“Materials and methods” section). The puromycin-resistant stable colonies were pooled together and named shCXCR4. A pool of cells infected with the lentivirus containing a non-effective vector (shRNA-NE) was selected and used as the control. A Western blot analysis was used to confirm the reduction in CXCR4 expression. B Illustration of co-cultures. C. BCAFs and HMFs were cultured in Dulbecco modified Eagle medium/nutrient mixture/F-12 supplemented with 10% FBS for 72 h. The culture supernatant was collected and tested for SDF-1 alpha using ELISA following the manufacturer’s instructions. The medium used for the cell culture was used as the negative control. The data were analyzed with one-way ANOVA. D, E CXCR4-knockdown cells or non-silent control cells were co-cultured with BCAFs (D) or with BCAFs and PBMCs (E) in 3D, followed by treatment with trastuzumab as illustrated in B. At the endpoint of the study, relative cell viability was quantitatively analyzed using CellTiter-Glo 3D viability assay kit. The data were analyzed with one-way ANOVA (*P < 0.05, **P < 0.01, ***P < 0.001 compared with the non-silent control cells). F, G CXCR4-knockdown cells or non-silent control cells were used for trastuzumab-induced antibody-dependent cellular cytotoxicity (detail in “Materials and methods” section). The cells were stained with propidium iodide and analyzed by flow cytometry (F). Data were analyzed using t-test analysis of variance and are reported as the mean ± SD of triplicates (G) Image collected and cropped by CiteAb from the following open publication (https://pubmed.ncbi.nlm.nih.gov/37280713), licensed under a CC-BY license. Not internally tested by R&D Systems.Detection of CXCR4 by Western Blot
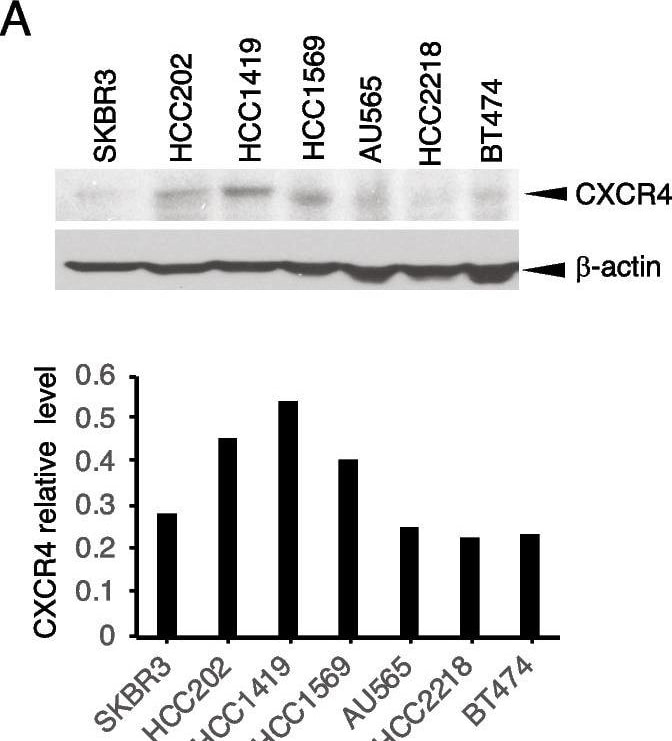
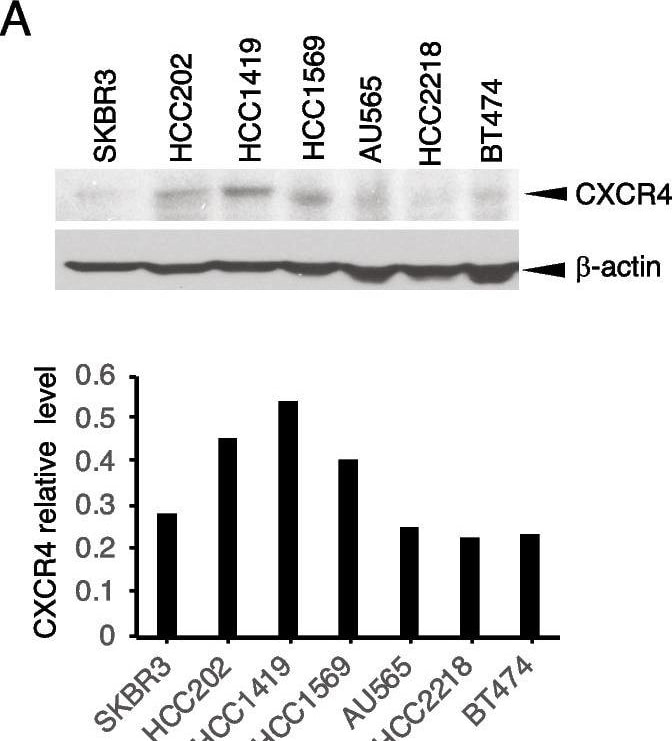
Targeting CXCR4 abrogates trastuzumab resistance. A HER2 + breast cancer cells with or without primary trastuzumab resistance were examined for CXCR4 expression with its antibody (UMB2, Abcam) using western blot analysis. The density of the bands was quantitatively analyzed. B, C Cell lines with high expression of CXCR4 (HCC1419 and HCC202) and low expression of CXCR4 (BT474 and SKBR3) were seeded in 3D Matrigel and treated with trastuzumab (B) or AMD3100 with SDF-1 alpha (4 ng/ml). (C). The total area of the acini was quantitatively analyzed (see “Materials and methods” section). D HCC1419 cells grown in 3D Matrigel culture were treated with trastuzumab (2 µg/ml), AMD3100 (1 µM), SDF-1 alpha (4 ng/ml), or the combination. Photographs were taken on day 13 after the start of treatment. The total area of the acini was quantitatively analyzed using AlphaView SA software (E). F–I Clonogenic assay. HCC1419 (F) and HCC202 (H) cells were seeded at low density and treated with AMD3100 (0.5 µM), SDF-1 alpha (4 ng/ml), trastuzumab (2.5 µg/ml), or the combination. The plates were scanned on day 18 after the start of treatment. Colony formation was quantitatively analyzed using AlphaView SA software. E, G, I Data were analyzed using one-way ANOVA and are reported as mean ± SD of triplicates, representing two independent experiments (*P < 0.0001 compared with vehicle) Image collected and cropped by CiteAb from the following open publication (https://pubmed.ncbi.nlm.nih.gov/37280713), licensed under a CC-BY license. Not internally tested by R&D Systems.Detection of CXCR4 by Western Blot
Knockdown of CXCR4 abrogates trastuzumab resistance. CXCR4 was silenced by specific shRNA in HCC1419 cells (“Materials and methods” section). The puromycin-resistant stable colonies were pooled together and named shCXCR4. A pool of cells infected with the lentivirus containing a non-effective vector (shRNA-NE) was selected and used as the control. A Western blot analysis was used to confirm the reduction in CXCR4 expression. B Illustration of co-cultures. C. BCAFs and HMFs were cultured in Dulbecco modified Eagle medium/nutrient mixture/F-12 supplemented with 10% FBS for 72 h. The culture supernatant was collected and tested for SDF-1 alpha using ELISA following the manufacturer’s instructions. The medium used for the cell culture was used as the negative control. The data were analyzed with one-way ANOVA. D, E CXCR4-knockdown cells or non-silent control cells were co-cultured with BCAFs (D) or with BCAFs and PBMCs (E) in 3D, followed by treatment with trastuzumab as illustrated in B. At the endpoint of the study, relative cell viability was quantitatively analyzed using CellTiter-Glo 3D viability assay kit. The data were analyzed with one-way ANOVA (*P < 0.05, **P < 0.01, ***P < 0.001 compared with the non-silent control cells). F, G CXCR4-knockdown cells or non-silent control cells were used for trastuzumab-induced antibody-dependent cellular cytotoxicity (detail in “Materials and methods” section). The cells were stained with propidium iodide and analyzed by flow cytometry (F). Data were analyzed using t-test analysis of variance and are reported as the mean ± SD of triplicates (G) Image collected and cropped by CiteAb from the following open publication (https://pubmed.ncbi.nlm.nih.gov/37280713), licensed under a CC-BY license. Not internally tested by R&D Systems.Detection of CXCR4 by Western Blot
Targeting CXCR4 abrogates trastuzumab resistance. A HER2 + breast cancer cells with or without primary trastuzumab resistance were examined for CXCR4 expression with its antibody (UMB2, Abcam) using western blot analysis. The density of the bands was quantitatively analyzed. B, C Cell lines with high expression of CXCR4 (HCC1419 and HCC202) and low expression of CXCR4 (BT474 and SKBR3) were seeded in 3D Matrigel and treated with trastuzumab (B) or AMD3100 with SDF-1 alpha (4 ng/ml). (C). The total area of the acini was quantitatively analyzed (see “Materials and methods” section). D HCC1419 cells grown in 3D Matrigel culture were treated with trastuzumab (2 µg/ml), AMD3100 (1 µM), SDF-1 alpha (4 ng/ml), or the combination. Photographs were taken on day 13 after the start of treatment. The total area of the acini was quantitatively analyzed using AlphaView SA software (E). F–I Clonogenic assay. HCC1419 (F) and HCC202 (H) cells were seeded at low density and treated with AMD3100 (0.5 µM), SDF-1 alpha (4 ng/ml), trastuzumab (2.5 µg/ml), or the combination. The plates were scanned on day 18 after the start of treatment. Colony formation was quantitatively analyzed using AlphaView SA software. E, G, I Data were analyzed using one-way ANOVA and are reported as mean ± SD of triplicates, representing two independent experiments (*P < 0.0001 compared with vehicle) Image collected and cropped by CiteAb from the following open publication (https://pubmed.ncbi.nlm.nih.gov/37280713), licensed under a CC-BY license. Not internally tested by R&D Systems.Applications for Human CXCR4 Antibody
Application
Recommended Usage
CyTOF-ready
Ready to be labeled using established conjugation methods. No BSA or other carrier proteins that could interfere with conjugation.
Flow Cytometry
0.25 µg/106 cells
Sample: Jurkat human acute T cell leukemia cell line
Sample: Jurkat human acute T cell leukemia cell line
Immunohistochemistry
8-25 µg/mL
Sample: Immersion fixed paraffin-embedded sections of human lymph node and human spleen
Sample: Immersion fixed paraffin-embedded sections of human lymph node and human spleen
Neutralization
Measured by its ability to neutralize CXCL12/SDF‑1 alpha -induced chemotaxis in the BaF3 mouse pro‑B cell line transfected with human CXCR4. The Neutralization Dose (ND50) is typically 2.5-12 µg/mL in the presence of 1 ng/mL Recombinant Human/Feline/Rhesus Macaque CXCL12/SDF‑1 alpha.
Reviewed Applications
Read 6 reviews rated 5 using MAB172 in the following applications:
Flow Cytometry Panel Builder
Bio-Techne Knows Flow Cytometry
Save time and reduce costly mistakes by quickly finding compatible reagents using the Panel Builder Tool.
Advanced Features
- Spectra Viewer - Custom analysis of spectra from multiple fluorochromes
- Spillover Popups - Visualize the spectra of individual fluorochromes
- Antigen Density Selector - Match fluorochrome brightness with antigen density
Formulation, Preparation, and Storage
Purification
Protein A or G purified from hybridoma culture supernatant
Reconstitution
Reconstitute at 0.5 mg/mL in sterile PBS. For liquid material, refer to CoA for concentration.
Loading...
Formulation
Lyophilized from a 0.2 μm filtered solution in PBS with Trehalose. *Small pack size (SP) is supplied either lyophilized or as a 0.2 µm filtered solution in PBS.
Shipping
Lyophilized product is shipped at ambient temperature. Liquid small pack size (-SP) is shipped with polar packs. Upon receipt, store immediately at the temperature recommended below.
Stability & Storage
Use a manual defrost freezer and avoid repeated freeze-thaw cycles.
- 12 months from date of receipt, -20 to -70 °C as supplied.
- 1 month, 2 to 8 °C under sterile conditions after reconstitution.
- 6 months, -20 to -70 °C under sterile conditions after reconstitution.
Calculators
Background: CXCR4
References
- Orsini, M.J. et al. (1999) J. Biol. Chem. 274:31076.
- Zagzag, D. et al. (2005) Cancer Res. 65:6178.
- Speetjens, F.M. et al. (2009) Cancer Microenvironment 2:1.
- Wang, L. et al. (2009) Oncology Reports 22:1333.
- Amara, S. et al. (2015) Cancer Biomark. 15:869.
Long Name
C-X-C Motif Chemokine Receptor 4
Alternate Names
CD184, D2S201E, FB22, Fusin, HM89, HSY3RR, LAP-3, LAP3, LCR1, LESTR, NPY3R, NPYRL, NPYY3R, WHIMS
Gene Symbol
CXCR4
Additional CXCR4 Products
Product Documents for Human CXCR4 Antibody
Certificate of Analysis
To download a Certificate of Analysis, please enter a lot or batch number in the search box below.
Note: Certificate of Analysis not available for kit components.
Product Specific Notices for Human CXCR4 Antibody
For research use only
Related Research Areas
Citations for Human CXCR4 Antibody
Customer Reviews for Human CXCR4 Antibody (6)
5 out of 5
6 Customer Ratings
Have you used Human CXCR4 Antibody?
Submit a review and receive an Amazon gift card!
$25/€18/£15/$25CAN/¥2500 Yen for a review with an image
$10/€7/£6/$10CAN/¥1110 Yen for a review without an image
Submit a review
Customer Images
Showing
1
-
5 of
6 reviews
Showing All
Filter By:
-
Application: ImmunohistochemistrySample Tested: Tumor tissue and Ductal carcinomaSpecies: HumanVerified Customer | Posted 08/03/2021
-
Application: MicroarraySample Tested: EDTA PlasmaSpecies: HumanVerified Customer | Posted 12/07/2020Antibody was printed on custom arrays and incubated with fluorescently labeled human EDTA plasma
-
Sample Tested: SUDHL-6 and Ramos human Burkitt's lymphoma cell lineSpecies: HumanVerified Customer | Posted 11/16/2018Cell line stained with human CXCR4 for 45 min and secondary antibody anti-mouse FITC for 45min & Isotype control and unstained cell used in experiment
-
Application: Flow CytometrySample Tested: SUDHL-6 and Ramos cells and SU-DHL-6 cell lineSpecies: HumanVerified Customer | Posted 10/27/2018Cell line stained with human CXCR4 for 45 min and secondary antibody anti-mouse FITC for 45min (Isotype control and unstained cell used in experiment)
-
Application: Flow CytometrySample Tested: Human cell lineSpecies: HumanVerified Customer | Posted 03/15/2018
-
Application: Flow CytometrySample Tested: JurkatSpecies: HumanVerified Customer | Posted 03/09/2018
There are no reviews that match your criteria.
Protocols
Find general support by application which include: protocols, troubleshooting, illustrated assays, videos and webinars.
- 7-Amino Actinomycin D (7-AAD) Cell Viability Flow Cytometry Protocol
- Antigen Retrieval Protocol (PIER)
- Antigen Retrieval for Frozen Sections Protocol
- Appropriate Fixation of IHC/ICC Samples
- Cellular Response to Hypoxia Protocols
- Chromogenic IHC Staining of Formalin-Fixed Paraffin-Embedded (FFPE) Tissue Protocol
- Chromogenic Immunohistochemistry Staining of Frozen Tissue
- ClariTSA™ Fluorophore Kits
- Detection & Visualization of Antibody Binding
- Extracellular Membrane Flow Cytometry Protocol
- Flow Cytometry Protocol for Cell Surface Markers
- Flow Cytometry Protocol for Staining Membrane Associated Proteins
- Flow Cytometry Staining Protocols
- Flow Cytometry Troubleshooting Guide
- Fluorescent IHC Staining of Frozen Tissue Protocol
- Graphic Protocol for Heat-induced Epitope Retrieval
- Graphic Protocol for the Preparation and Fluorescent IHC Staining of Frozen Tissue Sections
- Graphic Protocol for the Preparation and Fluorescent IHC Staining of Paraffin-embedded Tissue Sections
- Graphic Protocol for the Preparation of Gelatin-coated Slides for Histological Tissue Sections
- IHC Sample Preparation (Frozen sections vs Paraffin)
- Immunofluorescent IHC Staining of Formalin-Fixed Paraffin-Embedded (FFPE) Tissue Protocol
- Immunohistochemistry (IHC) and Immunocytochemistry (ICC) Protocols
- Immunohistochemistry Frozen Troubleshooting
- Immunohistochemistry Paraffin Troubleshooting
- Intracellular Flow Cytometry Protocol Using Alcohol (Methanol)
- Intracellular Flow Cytometry Protocol Using Detergents
- Intracellular Nuclear Staining Flow Cytometry Protocol Using Detergents
- Intracellular Staining Flow Cytometry Protocol Using Alcohol Permeabilization
- Intracellular Staining Flow Cytometry Protocol Using Detergents to Permeabilize Cells
- Preparing Samples for IHC/ICC Experiments
- Preventing Non-Specific Staining (Non-Specific Binding)
- Primary Antibody Selection & Optimization
- Propidium Iodide Cell Viability Flow Cytometry Protocol
- Protocol for Heat-Induced Epitope Retrieval (HIER)
- Protocol for Liperfluo
- Protocol for Making a 4% Formaldehyde Solution in PBS
- Protocol for VisUCyte™ HRP Polymer Detection Reagent
- Protocol for the Characterization of Human Th22 Cells
- Protocol for the Characterization of Human Th9 Cells
- Protocol for the Preparation & Fixation of Cells on Coverslips
- Protocol for the Preparation and Chromogenic IHC Staining of Frozen Tissue Sections
- Protocol for the Preparation and Chromogenic IHC Staining of Frozen Tissue Sections - Graphic
- Protocol for the Preparation and Chromogenic IHC Staining of Paraffin-embedded Tissue Sections
- Protocol for the Preparation and Chromogenic IHC Staining of Paraffin-embedded Tissue Sections - Graphic
- Protocol for the Preparation and Fluorescent IHC Staining of Frozen Tissue Sections
- Protocol for the Preparation and Fluorescent IHC Staining of Paraffin-embedded Tissue Sections
- Protocol for the Preparation of Gelatin-coated Slides for Histological Tissue Sections
- Protocol: Annexin V and PI Staining by Flow Cytometry
- Protocol: Annexin V and PI Staining for Apoptosis by Flow Cytometry
- TUNEL and Active Caspase-3 Detection by IHC/ICC Protocol
- The Importance of IHC/ICC Controls
- Troubleshooting Guide: Fluorokine Flow Cytometry Kits
- Troubleshooting Guide: Immunohistochemistry
- View all Protocols, Troubleshooting, Illustrated assays and Webinars