The galectins constitute a large family of carbohydrate-binding proteins with specificity for N-acetyl-lactosamine-containing glycoproteins. At least 14 mammalian galectins, which share structural similarities in their carbohydrate recognition domains (CRD), have been identified to date. The galectins have been classified into the prototype galectins (-1, -2, -5, -7, -10, -11, -13, -14), which contain one CRD and exist either as a monomer or a noncovalent homodimer; the chimera galectins (galectin-3) containing one CRD linked to a nonlectin domain; and the tandem-repeat galectins (-4, -6, -8, -9, -12) consisting of two CRDs joined by a linker peptide. Galectins lack a classical signal peptide and can be localized to the cytosolic compartments where they have intracellular functions. However, via one or more as yet unidentified non-classical secretory pathways, galectins can also be secreted to function extracellularly. Individual members of the galectin family have different tissue distribution profiles and exhibit subtle differences in their carbohydrate-binding specificities. Each family member may preferentially bind to a unique subset of cell-surface glycoproteins.
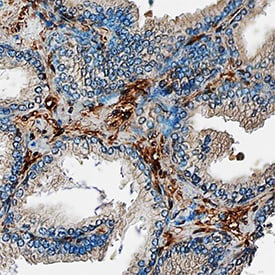
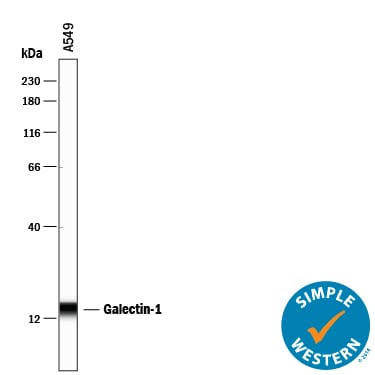
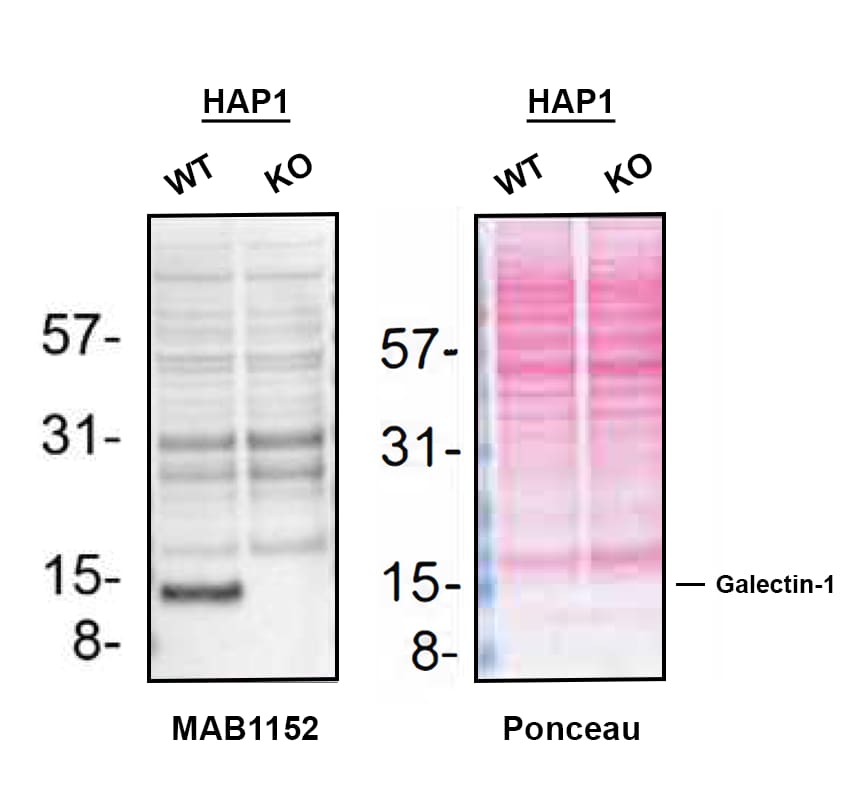
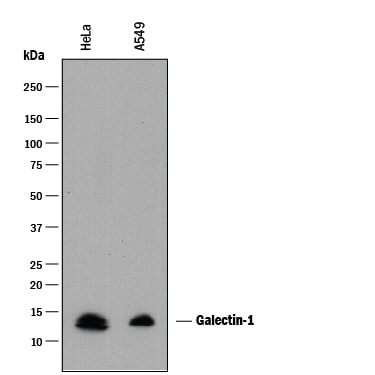
Galectin-1, also known as L-14, BHL and galaptin, is a monomeric or homodimeric prototype galectin that is expressed in a variety of cells and tissues including muscle, heart, liver, prostate, lymph nodes, spleen, thymus, placenta, testis, retina, macrophages, B cells, T cells, dendritic cells, and tumor cells. It preferentially binds laminin, fibronectin, 90K/Mac-2BP, CD45, CD43, CD7, CD2, CD3, and ganglioside GM1. Galectin-1 modulates cell growth and proliferation, either positively or negatively, depending on the cell type and activation status. It controls cell survival by inducing apoptosis of activated T cells and immature thymocytes. It modulates cytokine secretion by inducing Th2 type cytokines and inhibiting pro-inflammatory cytokine production. Galectin-1 can also modulate cell-cell as well as cell-matrix interactions and depending on the cell type and developmental stage, promote cell attachment or detachment. Galectin-1 has immunosuppressive and anti-inflammatory properties and has been shown to suppress acute and chronic inflammation and autoimmunity. Human and mouse galectin-1 share about 88% amino acid sequence similarity.