The superfamily of insulin-like growth factor (IGF) binding proteins include the six high-affinity IGF binding proteins (IGFBP) and at least four additional low-affinity binding proteins referred to as IGFBP related proteins (IGFBP-rP). All IGFBP superfamily members are cysteine-rich proteins with conserved cysteine residues, which are clustered in the amino- and carboxy-terminal thirds of the molecule. IGFBPs modulate the biological activities of IGF proteins. Some IGFBPs may also have intrinsic bioactivity that is independent of their ability to bind IGF proteins. Post-translational modifications of IGFBPs, including glycosylation, phosphorylation and proteolysis, have been shown to modify the affinities of the binding proteins to IGF.
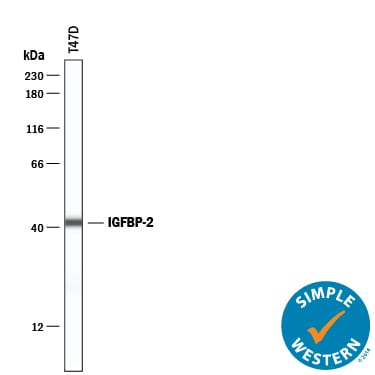
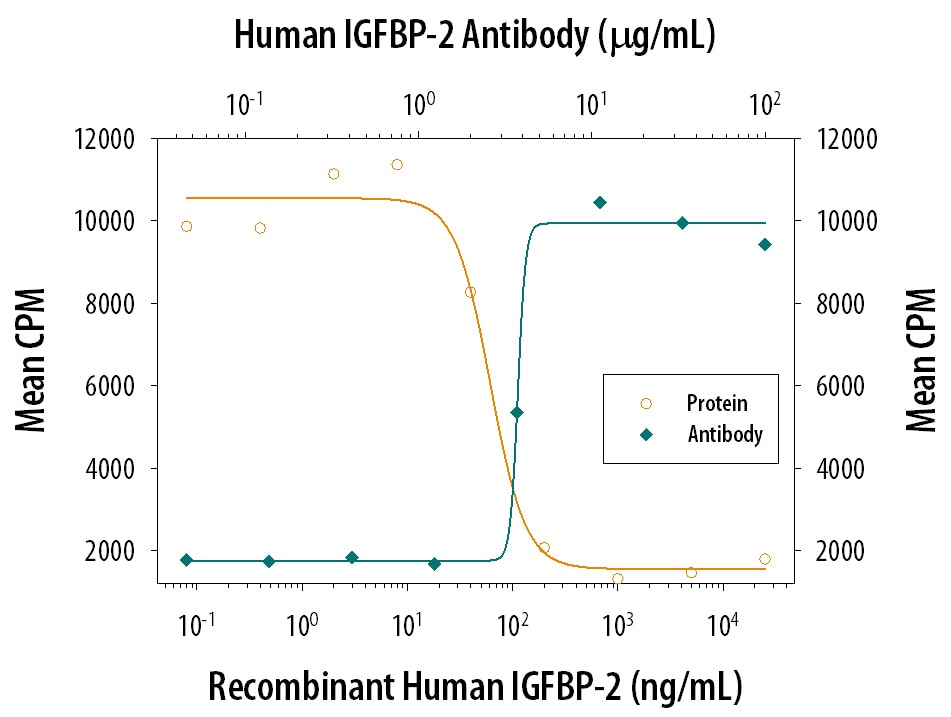
Human IGFBP-2 cDNA encodes a 328 amino acid (aa) residue precursor protein with a putative 39 aa residue signal peptide that is processed to generate the 289 aa residue mature protein. IGFBP-2 contains an integrin receptor recognition sequence (RGD sequence) but lacks potential N-linked glycosylation sites. During development, IGFBP-2 is expressed in a number of tissues. The highest expression level is found in the central nervous system. In adults, high expression levels are also detected in the central nervous system and in a number of reproductive tissues. IGFBP-2 binds preferentially to IGF-II, exhibiting a 2-10 fold higher affinity for IGF-II than for IGF-I.