Human IL‑18/IL‑1F4 Antibody
R&D Systems | Catalog # MAB9124
Recombinant Monoclonal Antibody.

Key Product Details
Species Reactivity
Human
Applications
Neutralization
Label
Unconjugated
Antibody Source
Monoclonal Rabbit IgG Clone # 1072F
Loading...
Product Specifications
Immunogen
E. coli-derived recombinant human IL-18/IL-1F4
Tyr37-Asp197
Accession # Q14116
Tyr37-Asp197
Accession # Q14116
Specificity
Detects human IL-18/IL-1F4 in direct ELISAs.
Clonality
Monoclonal
Host
Rabbit
Isotype
IgG
Endotoxin Level
<0.10 EU per 1 μg of the antibody by the LAL method.
Scientific Data Images for Human IL‑18/IL‑1F4 Antibody
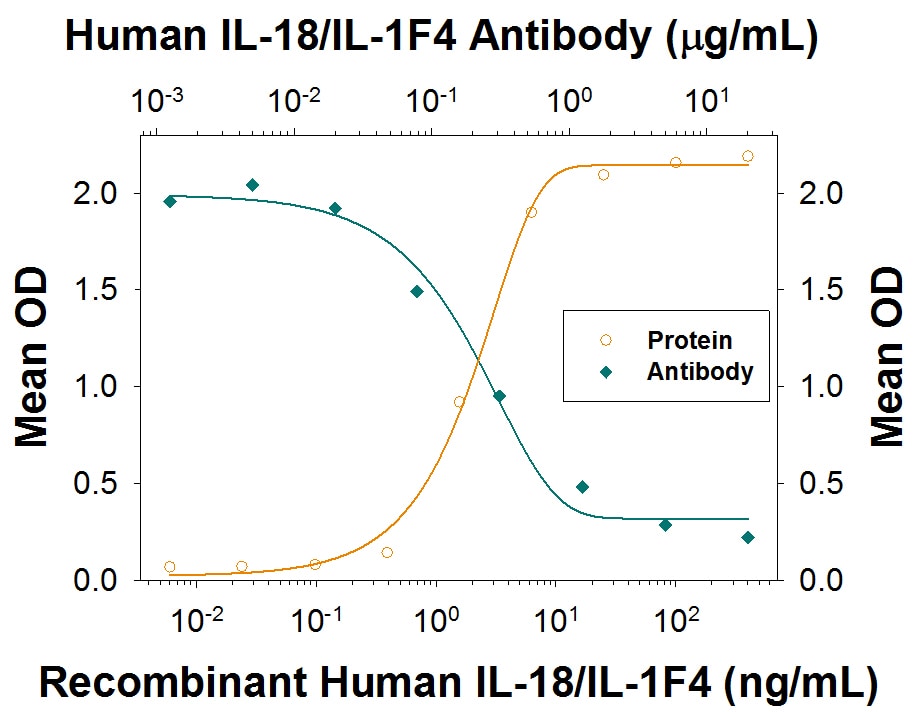
IFN-gamma Secretion Induced by IL-18/IL-1F4 and Neutralization by Human IL-18/IL-1F4 Antibody.
In the presence of Recombinant Human TNF-alpha (20 ng/mL, Catalog # 210-TA), Recombinant Human IL-18/IL-1F4 (Catalog # 9124-IL) stimulates IFN-gamma secretion in the KG-1 human acute myelogenous leukemia cell line in a dose-dependent manner (orange line), as measured by the Human IFN-gamma Quantikine ELISA Kit (Catalog # DIF50). Under these conditions, IFN-gamma secretion elicited by Recombinant Human IL-18/IL-1F4 (10 ng/mL) is neutralized (green line) by increasing concentrations of Rabbit Anti-Human IL-18/IL-1F4 Monoclonal Antibody (Catalog # MAB9124). The ND50 is typically 0.05-0.3 µg/mL.Applications for Human IL‑18/IL‑1F4 Antibody
Application
Recommended Usage
Neutralization
Measured by its ability to neutralize IL‑18/IL‑1F4-induced IFN‑ gamma secretion in the KG‑1 human acute myelogenous leukemia cell line. Novick, D. et al. (1999) Immunity 10(1):127. The Neutralization Dose (ND50) is typically 0.05‑0.3 µg/mL in the presence of 10 ng/mL Recombinant Human IL‑18/IL‑1F4 and 20 ng/mL Recombinant Human TNF‑ alpha.
Formulation, Preparation, and Storage
Purification
Protein A or G purified from cell culture supernatant
Reconstitution
Reconstitute at 0.5 mg/mL in sterile PBS. For liquid material, refer to CoA for concentration.
Loading...
Formulation
Lyophilized from a 0.2 μm filtered solution in PBS with Trehalose. *Small pack size (SP) is supplied either lyophilized or as a 0.2 µm filtered solution in PBS.
Shipping
Lyophilized product is shipped at ambient temperature. Liquid small pack size (-SP) is shipped with polar packs. Upon receipt, store immediately at the temperature recommended below.
Stability & Storage
Use a manual defrost freezer and avoid repeated freeze-thaw cycles.
- 12 months from date of receipt, -20 to -70 °C as supplied.
- 1 month, 2 to 8 °C under sterile conditions after reconstitution.
- 6 months, -20 to -70 °C under sterile conditions after reconstitution.
Calculators
Background: IL-18/IL-1F4
References
- Dinarello, C.A. et al. (2013) Front. Immunol. 4:289.
- Smith, D.E. (2011) J. Leukoc. Biol. 89:383.
- Gu, Y. et al. (1997) Science 275:206.
- Torigoe, K. et al. (1997) J. Biol. Chem. 272:25737.
- Cheung, H. et al. (2005) J. Immunol. 174:5351.
- Novick, D. et al. (1999) Immunity 10:127.
- Fehniger, T.A. et al. (1999) J. Immunol. 162:4511.
- Yoshimoto, T. et al. (1998) J. Immunol. 161:3400.
- Yoshimoto, T. et al. (2000) Nat. Immunol. 1:132.
- Kroeger, K.M. et al. (2009) J. Leukoc. Biol. 86:769.
- Lalor, S.J. et al. (2011) J. Immunol. 186:5738.
- Li, J. et al. (2004) Cell. Immunol. 227:103.
- Elbim, C. et al. (2005) Clin. Diagn. Lab. Immunol. 12:436.
- Fabbi, M. et al. (2015) J. Leukoc. Biol. 97:665.
- Ushio, S. et al. (1996) J. Immunol. 156:4274.
- Gaggero, A. et al. (2004) Oncogene 23:7552.
- Bellora, F. et al. (2012) Eur. J. Immunol. 42:1618.
Long Name
Interleukin 18
Alternate Names
IGIF, IL-1F4, IL-1g, IL18
Gene Symbol
IL18
UniProt
Additional IL-18/IL-1F4 Products
Product Documents for Human IL‑18/IL‑1F4 Antibody
Certificate of Analysis
To download a Certificate of Analysis, please enter a lot or batch number in the search box below.
Note: Certificate of Analysis not available for kit components.
Product Specific Notices for Human IL‑18/IL‑1F4 Antibody
For research use only
Citations for Human IL‑18/IL‑1F4 Antibody
Customer Reviews for Human IL‑18/IL‑1F4 Antibody
There are currently no reviews for this product. Be the first to review Human IL‑18/IL‑1F4 Antibody and earn rewards!
Have you used Human IL‑18/IL‑1F4 Antibody?
Submit a review and receive an Amazon gift card!
$25/€18/£15/$25CAN/¥2500 Yen for a review with an image
$10/€7/£6/$10CAN/¥1110 Yen for a review without an image
Submit a review






