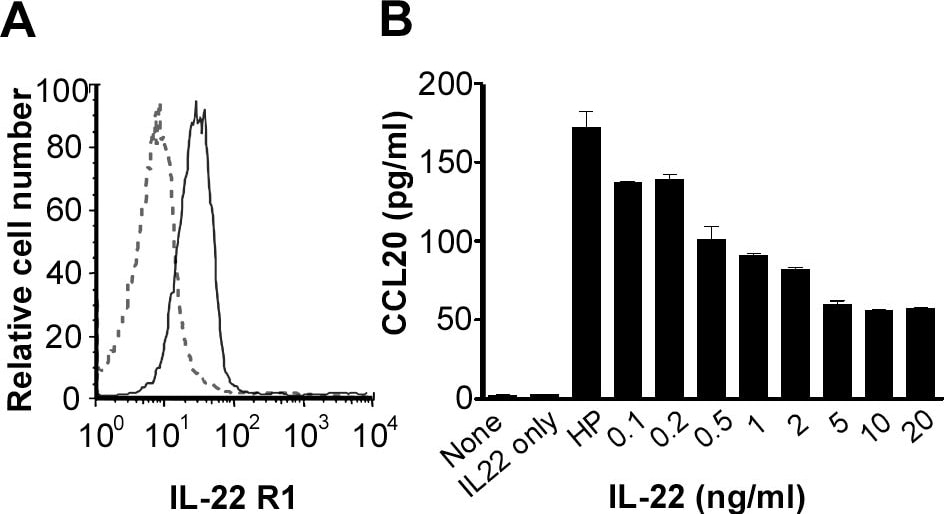
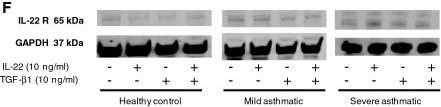
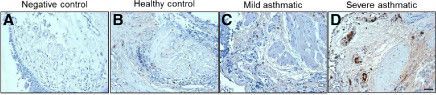
IL-22 receptor, also known as IL-22 R alpha 1 and CRF2-9, is an approximately 65 kDa transmembrane glycoprotein in the type II cytokine receptor family (CRF). IL-22 R alpha 1 contains a 211 amino acid (aa) extracellular domain (ECD) with two fibronectin type III repeats, and a 323 aa cytoplasmic domain. IL-22 R alpha 1 associates with either IL-10 R beta or IL-20 R beta to form receptor complexes with distinct ligand selectivities. IL-10 R beta is a shared subunit of the IL-10, -22, -26, -28, and -29 receptors, while IL-20 R beta is a shared subunit of the IL-19, -20, -22R and -24 receptors (1). IL-22 R alpha 1/IL-10 R beta is an IL-22 responsive receptor (2, 3), and IL-22 R alpha 1/IL-20 R beta is an IL-20 or IL-24 responsive receptor (4, 5). IL-22 R alpha 1 contains cytoplasmic motifs for interactions with signal transduction molecules, but formation of ternary complexes with IL-10 R beta or IL-20 R beta and the respective ligands is required for signal transduction (2, 6). IL-22BP functions as a competitive antagonist by binding
IL‑22 and preventing its association with IL-22 R alpha 1 (7, 9). Even though it is a receptor for interleukins, IL-22 R alpha 1 is not expressed on hematopoietic cells (6, 10, 11). Instead, IL-22 R alpha 1 expression is restricted to epithelial and stromal cells (6, 10‑13). IL-22 R alpha 1 signaling promotes innate immune responses and wound healing at sites of infection and inflammation. This includes upregulation of antimicrobial, acute phase, proinflammatory, and extracellular matrix proteins as well as proteases (3, 11, 13, 14). IL-22 R alpha 1 signaling also promotes downregulation of proteins involved in keratinocyte differentiation (3, 14). Within the ECD, human IL-22 R alpha 1 shares 78%, 76%, and 83% aa sequence identity with mouse, rat, and canine IL-22 R, respectively. It shares 22% - 25% aa sequence identity with the ECDs of other class II receptors IL-10 R, IL-20 R, and IL-28 R.