Oct-3/4, also termed Oct-3 or Oct-4, is a POU transcription factor that is expressed in totipotent embryonic stem and germ cells.(2, 3) Oct-3/4 is required to sustain stem cell self-renewal and pluripotency.(4) It is considered a master regulator of pluripotency that controls lineage commitment and is the most widely recognized marker of totipotent embryonic stem cells.

Key Product Details
Validated by
Biological Validation
Species Reactivity
Validated:
Human, Mouse
Cited:
Human, Mouse, Primate - Macaca mulatta (Rhesus Macaque)
Applications
Validated:
Western Blot, Intracellular Staining by Flow Cytometry, Immunocytochemistry, CyTOF-ready
Cited:
Immunohistochemistry, Immunohistochemistry-Frozen, Western Blot, Flow Cytometry, Immunocytochemistry, Immunocytochemistry/ Immunofluorescence, Bioassay
Label
Unconjugated
Antibody Source
Monoclonal Rat IgG2B Clone # 240408
Loading...
Product Specifications
Immunogen
E. coli-derived recombinant human Oct‑3/4
Met1-Asn265 (Met262Leu)
Accession # Q01860
Met1-Asn265 (Met262Leu)
Accession # Q01860
Specificity
Detects human Oct-3/4 in Western blots and detects mouse Oct-3/4 in flow cytometry.
Clonality
Monoclonal
Host
Rat
Isotype
IgG2B
Scientific Data Images for Human/Mouse Oct‑3/4 Antibody
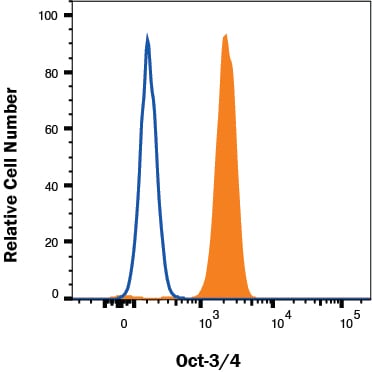
Detection of Oct‑3/4 in NTERA-2 cells by Flow Cytometry
NTERA-2 cells were stained with Rat Anti-Human/Mouse Oct‑3/4 Monoclonal Antibody (Catalog # MAB1759, filled histogram) or isotype control antibody (Catalog # MAB0061, open histogram) followed by Allophycocyanin-conjugated Anti-Rat IgG Secondary Antibody (Catalog # F0113). To facilitate intracellular staining, cells were fixed and permeabilized with FlowX FoxP3 Fixation & Permeabilization Buffer Kit (Catalog # FC012). View our protocol for Staining Intracellular Molecules.Detection of Human Oct-3/4 by Western Blot.
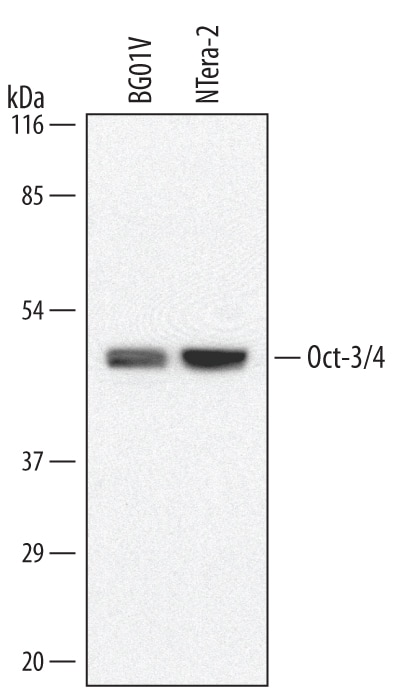
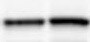
Western blot shows lysates of BG01V human embryonic stem cells and NTera-2 human testicular embryonic carcinoma cell line. PVDF membrane was probed with 1 µg/mL of Rat Anti-Human/Mouse Oct-3/4 Monoclonal Antibody (Catalog # MAB1759) followed by HRP-conjugated Anti-Rat IgG Secondary Antibody (Catalog # HAF005). A specific band was detected for Oct-3/4 at approximately 46 kDa (as indicated). This experiment was conducted under reducing conditions and using Immunoblot Buffer Group 1.Oct‑3/4 in BG01V Human Embryonic Stem Cells.
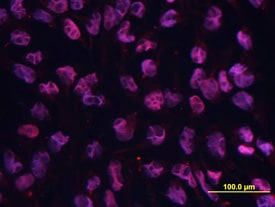
Oct‑3/4 was detected in immersion fixed BG01V human embryonic stem cells using Rat Anti-Human/Mouse Oct‑3/4 Monoclonal Antibody (Catalog # MAB1759) at 10 µg/mL for 3 hours at room temperature. Cells were stained using Rhodamine Red-coupled anti-rat IgG (red) and counterstained with DAPI (blue). Specific staining was localized to nuclei. View our protocol for Fluorescent ICC Staining of Cells on Coverslips.Detection of Human Oct-3/4 by Immunocytochemistry/Immunofluorescence
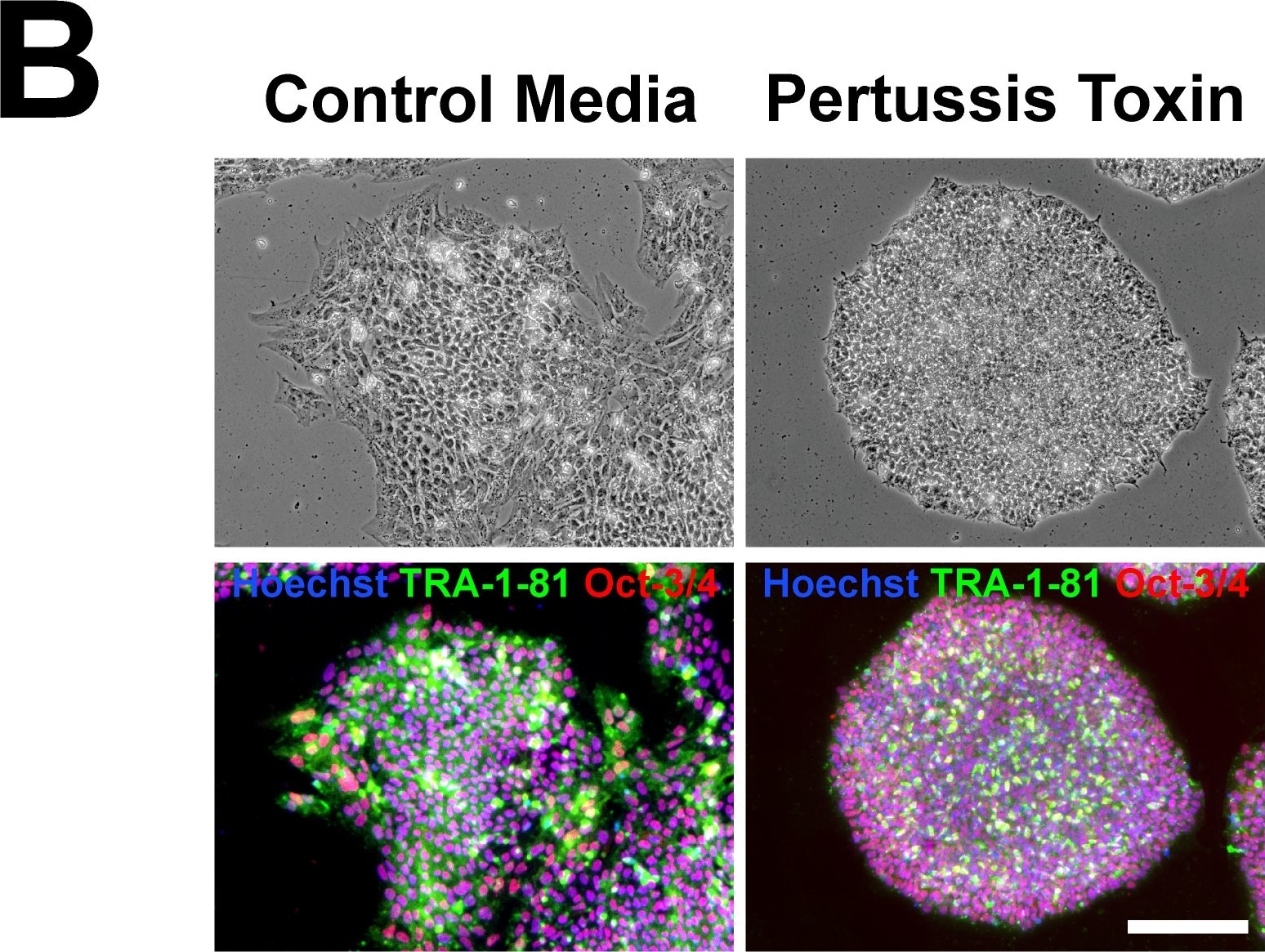
Pertussis toxin does not affect the maintenance of pluripotency pluripotent stem cell colonies or the ability to form all three embryonic germ layers.(A) Pertussis toxin does not affect the maintenance of pluripotency of hES or hiPS cells as assessed by the expression of the pluripotency markers Nanog, Oct4, Sox2, TDGF1, and ZFP42 by quantitative real-time PCR. (B) Immunocytochemistry of TRA-1-81 (green) and Oct-3/4 (red) also did not reveal differences between hES or hiPS cells treated with pertussis toxin and control treatments in pluripotent stem cell colonies. Nuclei were stained with Hoechst (blue). Scale bar, 200 µm. (C) hES or iPS cells treated with pertussis toxin formed embryoid bodies and differentiated into cell types of the all three embryonic germ layers as assessed by immunocytochemistry for alpha -fetoprotein (a marker of endoderm), alpha -smooth muscle actin (mesoderm) and beta III-tubulin (ectoderm). Scale bar, 100 µm. Image collected and cropped by CiteAb from the following publication (https://pubmed.ncbi.nlm.nih.gov/19936228), licensed under a CC-BY license. Not internally tested by R&D Systems.Detection of Human Oct-3/4 by Western Blot
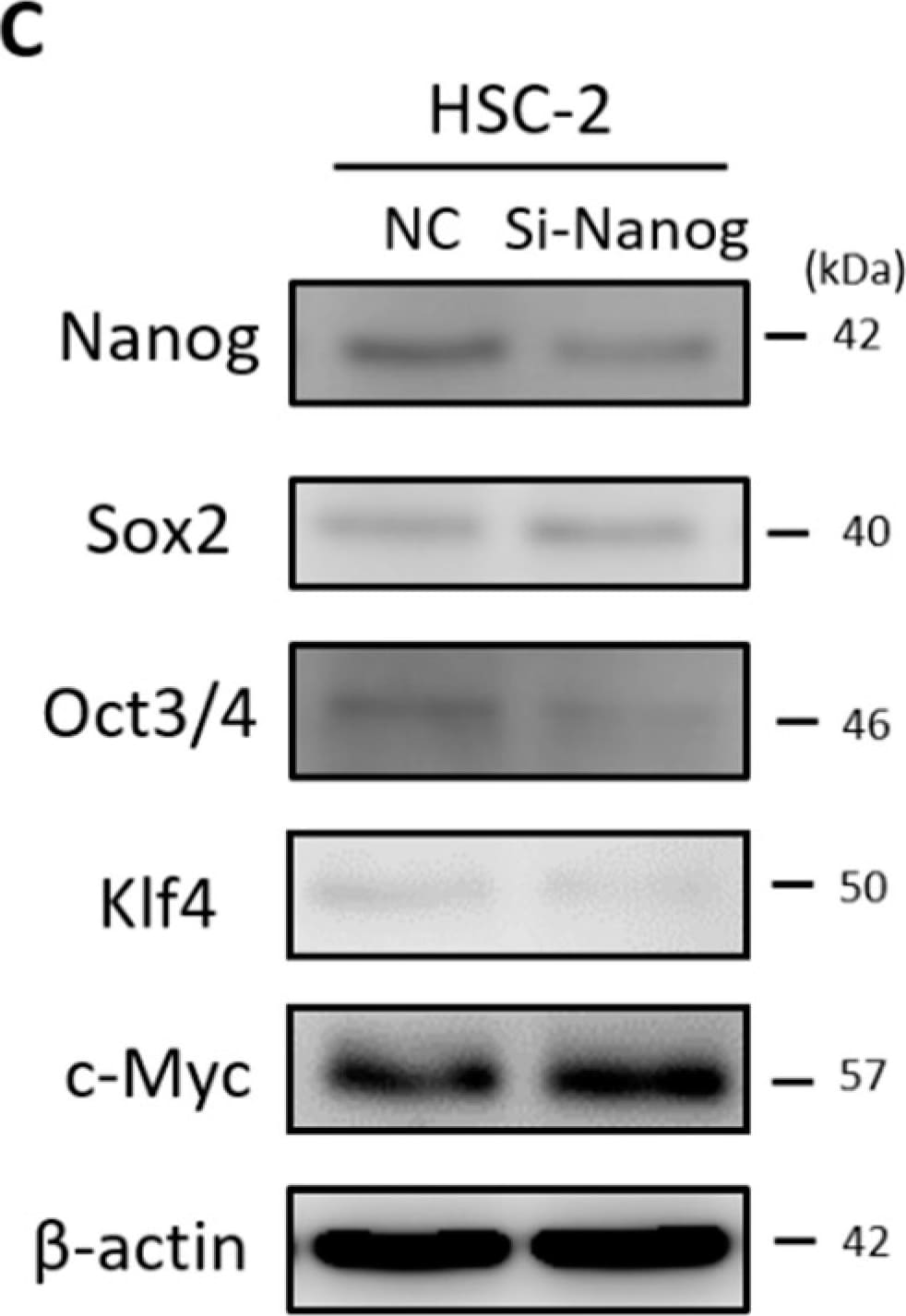
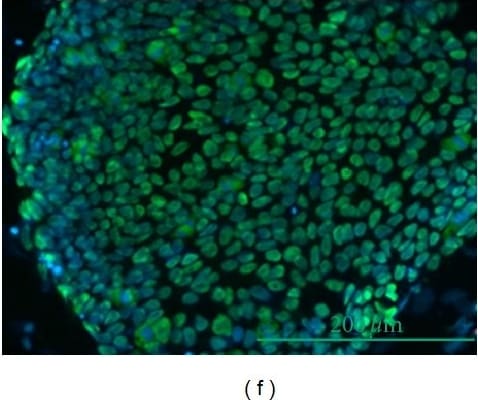
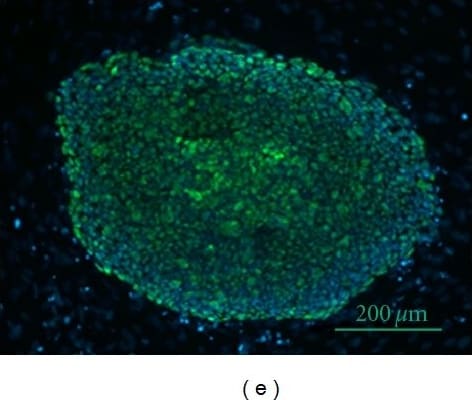
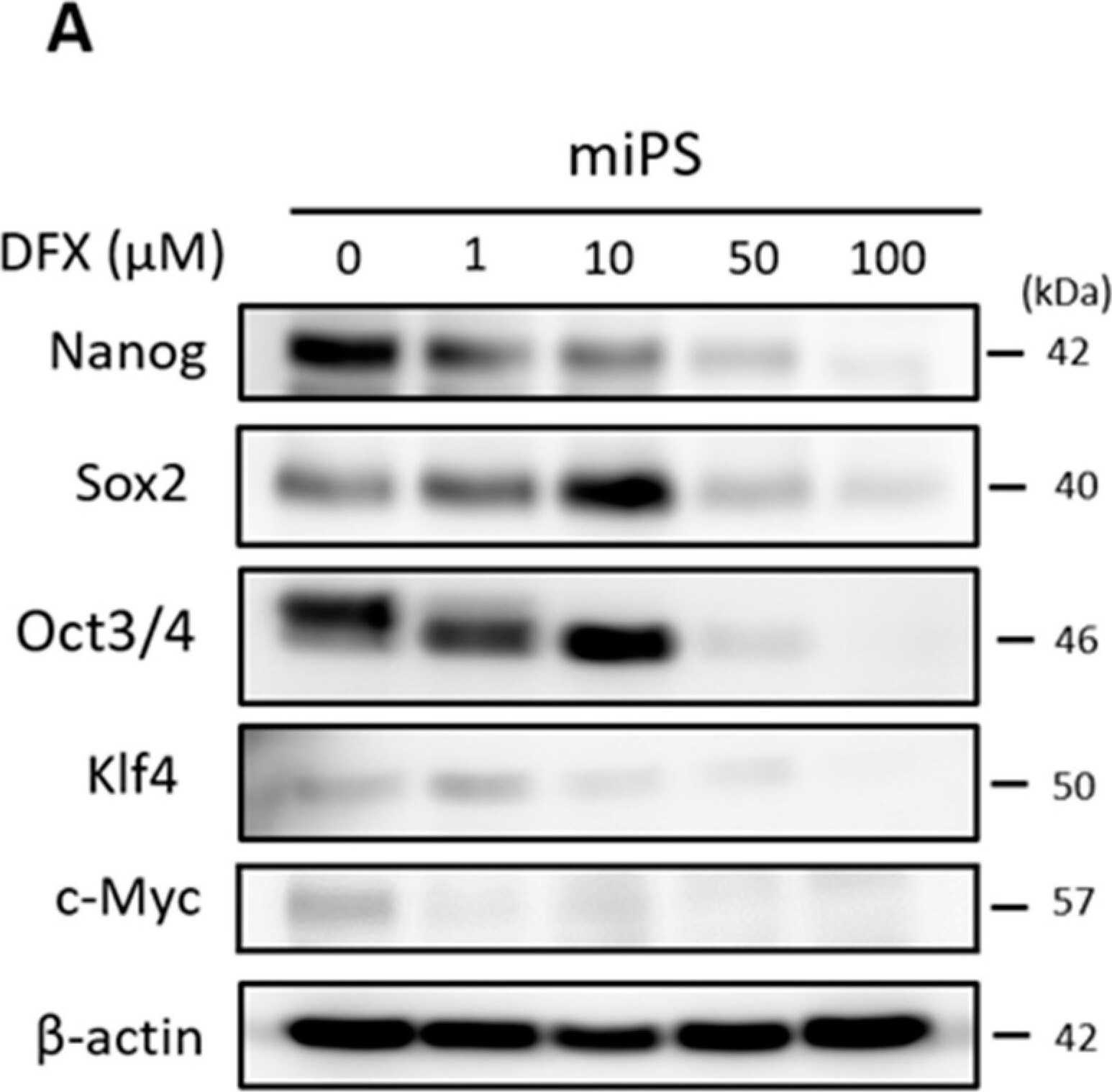
Effect of DFX on spherogenicity of human cancer cell lines and treatment with Nanog siRNA in vitro. (A) After treatment with 0.2% DMSO or 50 μM DFX, a single suspension of HSC-2 cells or OE33 cells was used for the sphere formation assay in a 96-well ultra-low attachment plate. DFX suppressed the spherogenicity of HSC-2 cells and OE33 cells. (B) A single suspension of HSC-2 cells or OE33 cells as described above was used for the spheroid colony assay in a 24-well ultra-low attachment plate. The number of spheres over 50 μm in diameter was counted. The experiments were performed in triplicate, and means ± S.E.M. of each group are shown. DFX significantly suppressed the number of spheres. * p < 0.05. (C) HSC-2 cells were transfected with control or si-Nanog for 48 h, and the expression of stemness markers (Nanog, Sox2, Oct3/4, Klf4, c-Myc) was determined with western blot analysis. beta -actin was used as a loading control. siRNA suppressed the expression of Nanog, Oct3/4, and Klf4. (D) HSC-2 cells were transfected with control or si-Nanog for 48 h, and the sphere formation assay was performed. No differences were found in spherogenicity between the control and si-Nanog cultures. Image collected and cropped by CiteAb from the following publication (https://pubmed.ncbi.nlm.nih.gov/30717462), licensed under a CC-BY license. Not internally tested by R&D Systems.Detection of Human Oct-3/4 by Immunocytochemistry/Immunofluorescence
Undifferentiated H7 hESC colonies (on passage 15) 3 days after passaging on hFF feeder cells (a) and after 55 passages on MEF feeder cells (b). H7 cells passaged 11 times on hFF feeder cells expressed Nanog (c and d), Oct4 (e and f), SSEA4 (g and h), and TRA-1-60 (i and j). H7 cells on passage 45 on MEF feeder cells expressed Nanog (k and l), Oct4 (m and n), and SSEA4 (o and p). Scale bars 200 μm. Image collected and cropped by CiteAb from the following publication (https://pubmed.ncbi.nlm.nih.gov/22315618), licensed under a CC-BY license. Not internally tested by R&D Systems.Detection of Human Oct-3/4 by Immunocytochemistry/Immunofluorescence
Undifferentiated H7 hESC colonies (on passage 15) 3 days after passaging on hFF feeder cells (a) and after 55 passages on MEF feeder cells (b). H7 cells passaged 11 times on hFF feeder cells expressed Nanog (c and d), Oct4 (e and f), SSEA4 (g and h), and TRA-1-60 (i and j). H7 cells on passage 45 on MEF feeder cells expressed Nanog (k and l), Oct4 (m and n), and SSEA4 (o and p). Scale bars 200 μm. Image collected and cropped by CiteAb from the following publication (https://pubmed.ncbi.nlm.nih.gov/22315618), licensed under a CC-BY license. Not internally tested by R&D Systems.Detection of Mouse Oct-3/4 by Western Blot
The effect of DFX against stemness of miPS cells in vitro and cytotoxicity analysis. (A) miPS cells were treated with the indicated dose of DFX (0, 1, 10, 50, 100 μM) and subjected to western blot analysis with antibodies to stemness markers (Nanog, Sox2, Oct3/4, Klf4, c-Myc) or beta -actin (loading control). Stemness markers were suppressed by DFX at concentrations over 50 μM. (B) miPS cells treated with 50 μM DFX were cultured in suspension for 72 h. DFX treatment of miPS cells suppressed spherogenesis and GFP expression, which indicates suppression of Nanog. (C) Micrographs of the fluorescence-based Live/Dead assay showing live and dead miPS cells following treatment with 0.2% DMSO (control) or 50 μM DFX (magnification ×40). The morphology of miPS cells after treatment with DFX changed from round to spindle shaped. Almost all cells were stained green, which indicates live cells. Image collected and cropped by CiteAb from the following publication (https://pubmed.ncbi.nlm.nih.gov/30717462), licensed under a CC-BY license. Not internally tested by R&D Systems.Applications for Human/Mouse Oct‑3/4 Antibody
Application
Recommended Usage
CyTOF-ready
Ready to be labeled using established conjugation methods. No BSA or other carrier proteins that could interfere with conjugation.
Immunocytochemistry
8-25 µg/mL
Sample: Immersion fixed NTera-2 human testicular embryonic carcinoma cell line, D3 mouse embryonic stem cell line, and BG01V human embryonic stem cells
Sample: Immersion fixed NTera-2 human testicular embryonic carcinoma cell line, D3 mouse embryonic stem cell line, and BG01V human embryonic stem cells
Intracellular Staining by Flow Cytometry
0.25 µg/106 cells
Sample: NTERA-2 human testicular embryonic carcinoma cell line
Sample: NTERA-2 human testicular embryonic carcinoma cell line
Western Blot
1 µg/mL
Sample: BG01V human embryonic stem cells and NTera‑2 human testicular embryonic carcinoma cell line under reducing conditions only
Sample: BG01V human embryonic stem cells and NTera‑2 human testicular embryonic carcinoma cell line under reducing conditions only
Reviewed Applications
Read 3 reviews rated 5 using MAB1759 in the following applications:
Flow Cytometry Panel Builder
Bio-Techne Knows Flow Cytometry
Save time and reduce costly mistakes by quickly finding compatible reagents using the Panel Builder Tool.
Advanced Features
- Spectra Viewer - Custom analysis of spectra from multiple fluorochromes
- Spillover Popups - Visualize the spectra of individual fluorochromes
- Antigen Density Selector - Match fluorochrome brightness with antigen density
Formulation, Preparation, and Storage
Purification
Protein A or G purified from hybridoma culture supernatant
Reconstitution
Reconstitute at 0.5 mg/mL in sterile PBS. For liquid material, refer to CoA for concentration.
Loading...
Formulation
Lyophilized from a 0.2 μm filtered solution in PBS with Trehalose. See Certificate of Analysis for details.
*Small pack size (-SP) is supplied either lyophilized or as a 0.2 µm filtered solution in PBS.
*Small pack size (-SP) is supplied either lyophilized or as a 0.2 µm filtered solution in PBS.
Shipping
Lyophilized product is shipped at ambient temperature. Liquid small pack size (-SP) is shipped with polar packs. Upon receipt, store immediately at the temperature recommended below.
Stability & Storage
Use a manual defrost freezer and avoid repeated freeze-thaw cycles.
- 12 months from date of receipt, -20 to -70 °C as supplied.
- 1 month, 2 to 8 °C under sterile conditions after reconstitution.
- 6 months, -20 to -70 °C under sterile conditions after reconstitution.
Calculators
Background: Oct-3/4
References
-
Takeda, J. et al. (1992) Nucleic Acids Res. 20:4613.
-
Scholer, H.R. et al. (1989) EMBO J. 8:2543.
-
Rosner, M.H. et al. (1990) Nature 345:686.
- Niwa, H. et al. (2000) Nat. Genet. 24:372.
Long Name
Octamer-binding Protein 3/4
Alternate Names
3-Oct, Oct4, Otf3g, Otf4, Pou5f1
Gene Symbol
POU5F1
UniProt
Additional Oct-3/4 Products
Product Documents for Human/Mouse Oct‑3/4 Antibody
Certificate of Analysis
To download a Certificate of Analysis, please enter a lot or batch number in the search box below.
Note: Certificate of Analysis not available for kit components.
Product Specific Notices for Human/Mouse Oct‑3/4 Antibody
For research use only
Citations for Human/Mouse Oct‑3/4 Antibody
Customer Reviews for Human/Mouse Oct‑3/4 Antibody (3)
5 out of 5
3 Customer Ratings
Have you used Human/Mouse Oct‑3/4 Antibody?
Submit a review and receive an Amazon gift card!
$25/€18/£15/$25CAN/¥2500 Yen for a review with an image
$10/€7/£6/$10CAN/¥1110 Yen for a review without an image
Submit a review
Customer Images
Showing
1
-
3 of
3 reviews
Showing All
Filter By:
-
Application: Western BlotSample Tested: AdipocytesSpecies: HumanVerified Customer | Posted 07/17/2025
-
Application: Western BlotSample Tested: HCT-116 human colorectal carcinoma cell lineSpecies: HumanVerified Customer | Posted 07/10/2025
-
Application: Western BlotSample Tested: NTera-2 human testicular embryonic carcinoma cell lineSpecies: HumanVerified Customer | Posted 02/09/2022
There are no reviews that match your criteria.
Protocols
Find general support by application which include: protocols, troubleshooting, illustrated assays, videos and webinars.
- 7-Amino Actinomycin D (7-AAD) Cell Viability Flow Cytometry Protocol
- Appropriate Fixation of IHC/ICC Samples
- Cellular Response to Hypoxia Protocols
- ClariTSA™ Fluorophore Kits
- Detection & Visualization of Antibody Binding
- Extracellular Membrane Flow Cytometry Protocol
- Flow Cytometry Protocol for Cell Surface Markers
- Flow Cytometry Protocol for Staining Membrane Associated Proteins
- Flow Cytometry Staining Protocols
- Flow Cytometry Troubleshooting Guide
- ICC Cell Smear Protocol for Suspension Cells
- ICC Immunocytochemistry Protocol Videos
- ICC for Adherent Cells
- Immunocytochemistry (ICC) Protocol
- Immunocytochemistry Troubleshooting
- Immunofluorescence of Organoids Embedded in Cultrex Basement Membrane Extract
- Immunohistochemistry (IHC) and Immunocytochemistry (ICC) Protocols
- Intracellular Flow Cytometry Protocol Using Alcohol (Methanol)
- Intracellular Flow Cytometry Protocol Using Detergents
- Intracellular Nuclear Staining Flow Cytometry Protocol Using Detergents
- Intracellular Staining Flow Cytometry Protocol Using Alcohol Permeabilization
- Intracellular Staining Flow Cytometry Protocol Using Detergents to Permeabilize Cells
- Preparing Samples for IHC/ICC Experiments
- Preventing Non-Specific Staining (Non-Specific Binding)
- Primary Antibody Selection & Optimization
- Propidium Iodide Cell Viability Flow Cytometry Protocol
- Protocol for Liperfluo
- Protocol for VisUCyte™ HRP Polymer Detection Reagent
- Protocol for the Characterization of Human Th22 Cells
- Protocol for the Characterization of Human Th9 Cells
- Protocol for the Fluorescent ICC Staining of Cell Smears - Graphic
- Protocol for the Fluorescent ICC Staining of Cultured Cells on Coverslips - Graphic
- Protocol for the Preparation and Fluorescent ICC Staining of Cells on Coverslips
- Protocol for the Preparation and Fluorescent ICC Staining of Non-adherent Cells
- Protocol for the Preparation and Fluorescent ICC Staining of Stem Cells on Coverslips
- Protocol for the Preparation of a Cell Smear for Non-adherent Cell ICC - Graphic
- Protocol: Annexin V and PI Staining by Flow Cytometry
- Protocol: Annexin V and PI Staining for Apoptosis by Flow Cytometry
- R&D Systems Quality Control Western Blot Protocol
- TUNEL and Active Caspase-3 Detection by IHC/ICC Protocol
- The Importance of IHC/ICC Controls
- Troubleshooting Guide: Fluorokine Flow Cytometry Kits
- Troubleshooting Guide: Western Blot Figures
- Western Blot Conditions
- Western Blot Protocol
- Western Blot Protocol for Cell Lysates
- Western Blot Troubleshooting
- Western Blot Troubleshooting Guide
- View all Protocols, Troubleshooting, Illustrated assays and Webinars
Loading...
Associated Pathways