Heat shock proteins (HSPs) are a family of highly conserved stress response proteins. Heat shock proteins function primarily as molecular chaperones by facilitating the folding of other cellular proteins, preventing protein aggregation or targeting improperly folded proteins to specific degradative pathways. HSPs are typically expressed at low levels under normal physiological conditions but are dramatically up-regulated in response to cellular stress. Elevated levels of HSPs have been observed in association with ischemia/reperfusion, cancer, and chronic heart failure. HSP27 is a member of the small heat shock protein family, which also includes HSP25 and the alpha -crystallins. Ser78 and Ser82 of HSP27 are phosphorylated in vivo in response to growth factors or heat shock, and the extent of phosphorylation plays a role in determining specific functions. HSP27 forms a large oligomer and functions as an anti-apoptotic molecule, regulating apoptosis through direct interaction with key components of the apoptotic pathway. HSP27 binds and sequesters cytochrome c released from the mitochondria in response to an apoptotic stimulus. This prevents the proper assembly of the apoptosome and subsequently, the activation of procaspase-9 and procaspase-3.
Human Phospho-HSP27 (S78/S82) Antibody
R&D Systems | Catalog # MAB23141
Recombinant Monoclonal Antibody.

Key Product Details
Validated by
Biological Validation
Species Reactivity
Validated:
Human
Cited:
Human
Applications
Validated:
Western Blot, Immunocytochemistry
Cited:
Western Blot
Label
Unconjugated
Antibody Source
Recombinant Monoclonal Rabbit IgG Clone # 1026B
Loading...
Product Specifications
Immunogen
Phosphopeptide containing the human HSP27 S78/S82 site
Accession # P04792
Accession # P04792
Specificity
Detects human HSP27 when dually phosphorylated at S78/S82 in Western blots.
Clonality
Monoclonal
Host
Rabbit
Isotype
IgG
Scientific Data Images for Human Phospho-HSP27 (S78/S82) Antibody
Detection of Human Phospho-HSP27 (S78/S82) by Western Blot.
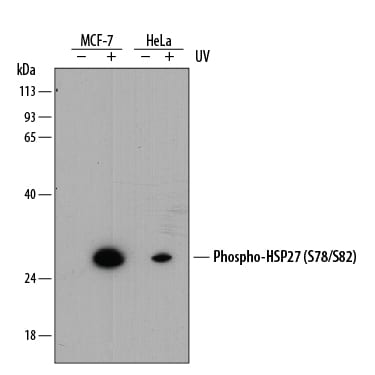
Western blot shows lysates of MCF-7 human breast cancer cell line and HeLa human cervical epithelial carcinoma cell line untreated (-) or treated (+) with 20 mJ/cm2ultraviolet light (UV) with a 30 minute recovery. PVDF membrane was probed with 1 µg/mL of Rabbit Anti-Human Phospho-HSP27 (S78/S82) Monoclonal Antibody (Catalog # MAB23141) followed by HRP-conjugated Anti-Rabbit IgG Secondary Antibody (Catalog # HAF008). A specific band was detected for Phospho-HSP27 (S78/S82) at approximately 27 kDa (as indicated). This experiment was conducted under reducing conditions and using Immunoblot Buffer Group 1.Phospho-HSP27 (S78/S82) in HeLa Human Cell Line.
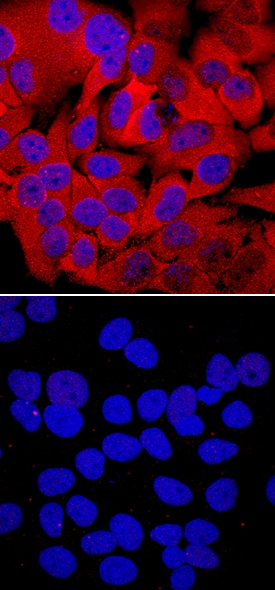
HSP27 phosphorylated at S78/S82 was detected in immersion fixed HeLa human cervical epithelial carcinoma cell line unstimulated (lower panel) or stimulated with 20 mJ/cm2ultraviolet radiation (upper panel) using Rabbit Anti-Human Phospho-HSP27 (S78/S82) Monoclonal Antibody (Catalog # MAB23141) at a 10 µg/mL for 3 hours at room temperature. Cells were stained using the NorthernLights™ 557-conjugated Anti-Rabbit IgG Secondary Antibody (red; Catalog # NL004) and counterstained with DAPI (blue). Specific staining was localized to cytoplasm and nuclei. View our protocol for Fluorescent ICC Staining of Cells on Coverslips.Detection of Phospho-HSP27 (S78/S82) by Western Blot
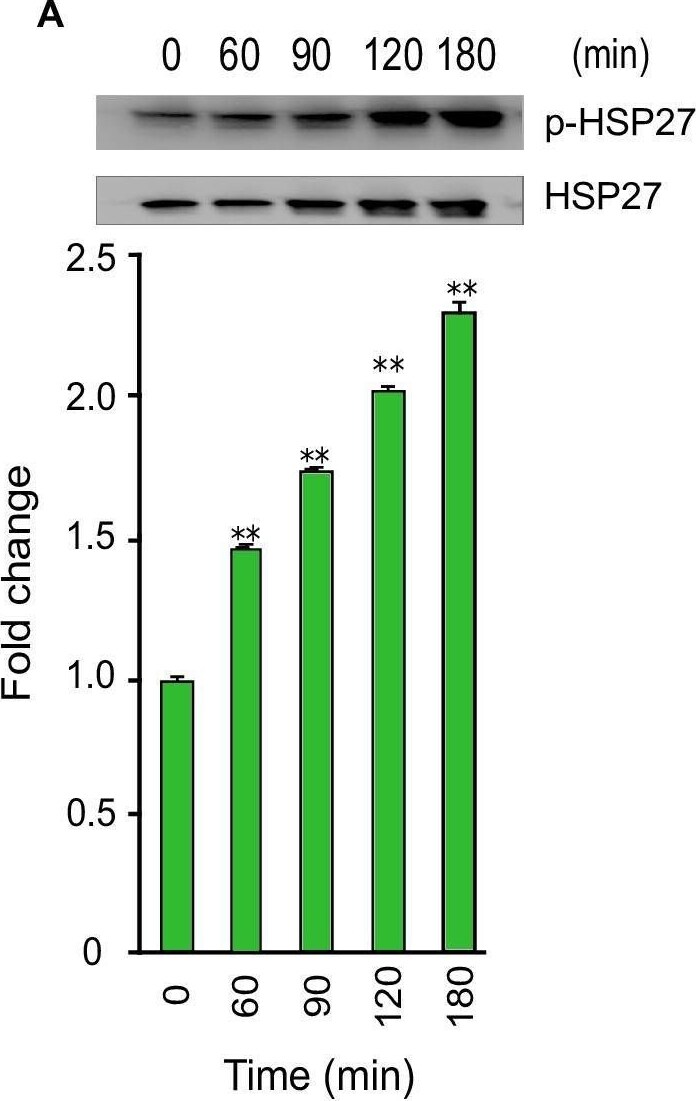
Phosphorylation of Akt, RSK, and CREB during T. cruzi infection. Lysates from primary HCoEpiC infected with T. cruzi at multiple time points were resolved by SDS-PAGE, blotted, and probed with antibodies against (A) p-HSP27 (B) p-Akt T308, (C) p-Akt S473, (D) p-RSK, and (E) p-CREB, and developed as described. The blots were stripped and reprobed with antibodies against the respective total proteins. The blots were developed by chemiluminescence and scanned. The normalized fold change in the level of each phosphorylated protein was determined and plotted in the bar graph below the respective blot. The bar graphs represent mean values ± SE from three independent biological replicates. The value of p<0.05 was considered significant; *p<0.05 **p<0.001. Image collected and cropped by CiteAb from the following open publication (https://pubmed.ncbi.nlm.nih.gov/30222739), licensed under a CC-BY license. Not internally tested by R&D Systems.Applications for Human Phospho-HSP27 (S78/S82) Antibody
Application
Recommended Usage
Immunocytochemistry
5-25 µg/mL
Sample: Immersion fixed HeLa human cervical epithelial carcinoma cell line unstimulated or stimulated with 20 mJ/cm2 ultraviolet radiation
Sample: Immersion fixed HeLa human cervical epithelial carcinoma cell line unstimulated or stimulated with 20 mJ/cm2 ultraviolet radiation
Western Blot
1 µg/mL
Sample: MCF‑7 human breast cancer cell line and HeLa human cervical epithelial carcinoma cell line treated with ultraviolet light (UV)
Sample: MCF‑7 human breast cancer cell line and HeLa human cervical epithelial carcinoma cell line treated with ultraviolet light (UV)
Reviewed Applications
Read 1 review rated 5 using MAB23141 in the following applications:
Formulation, Preparation, and Storage
Purification
Protein A or G purified from cell culture supernatant
Reconstitution
Reconstitute at 0.5 mg/mL in sterile PBS. For liquid material, refer to CoA for concentration.
Loading...
Formulation
Lyophilized from a 0.2 μm filtered solution in PBS with Trehalose. *Small pack size (SP) is supplied either lyophilized or as a 0.2 µm filtered solution in PBS.
Shipping
Lyophilized product is shipped at ambient temperature. Liquid small pack size (-SP) is shipped with polar packs. Upon receipt, store immediately at the temperature recommended below.
Stability & Storage
Store the unopened product at -20 to -70 °C. Use a manual defrost freezer and avoid repeated freeze-thaw cycles. Do not use past expiration date.
Calculators
Background: HSP27
References
- Gusev, N.B. et al. (2002) Biochemistry (Moscow) 67:511.
- Garrido, C. et al. (2001) Biochem. Biophys. Res. Commun. 286:433.
- Garrido, C. (2002) Cell Death Diffr. 9:483.
- Brvey, J-M. et al. (2000) Nat. Cell Biol. 2:645.
Long Name
Heat Shock Protein 27
Alternate Names
HSP25, HSPB1
Gene Symbol
HSPB1
UniProt
Additional HSP27 Products
Product Documents for Human Phospho-HSP27 (S78/S82) Antibody
Certificate of Analysis
To download a Certificate of Analysis, please enter a lot or batch number in the search box below.
Note: Certificate of Analysis not available for kit components.
Product Specific Notices for Human Phospho-HSP27 (S78/S82) Antibody
For research use only
Related Research Areas
Citations for Human Phospho-HSP27 (S78/S82) Antibody
Customer Reviews for Human Phospho-HSP27 (S78/S82) Antibody (1)
5 out of 5
1 Customer Rating
Have you used Human Phospho-HSP27 (S78/S82) Antibody?
Submit a review and receive an Amazon gift card!
$25/€18/£15/$25CAN/¥2500 Yen for a review with an image
$10/€7/£6/$10CAN/¥1110 Yen for a review without an image
Submit a review
Customer Images
Showing
1
-
1 of
1 review
Showing All
Filter By:
-
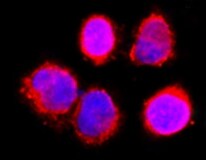
Application: Immunocytochemistry/ImmunofluorescenceSample Tested: HeLa cellsSpecies: HumanVerified Customer | Posted 05/17/2022
There are no reviews that match your criteria.
Protocols
Find general support by application which include: protocols, troubleshooting, illustrated assays, videos and webinars.
- Appropriate Fixation of IHC/ICC Samples
- Cellular Response to Hypoxia Protocols
- ClariTSA™ Fluorophore Kits
- Detection & Visualization of Antibody Binding
- ICC Cell Smear Protocol for Suspension Cells
- ICC Immunocytochemistry Protocol Videos
- ICC for Adherent Cells
- Immunocytochemistry (ICC) Protocol
- Immunocytochemistry Troubleshooting
- Immunofluorescence of Organoids Embedded in Cultrex Basement Membrane Extract
- Immunohistochemistry (IHC) and Immunocytochemistry (ICC) Protocols
- Preparing Samples for IHC/ICC Experiments
- Preventing Non-Specific Staining (Non-Specific Binding)
- Primary Antibody Selection & Optimization
- Protocol for VisUCyte™ HRP Polymer Detection Reagent
- Protocol for the Fluorescent ICC Staining of Cell Smears - Graphic
- Protocol for the Fluorescent ICC Staining of Cultured Cells on Coverslips - Graphic
- Protocol for the Preparation and Fluorescent ICC Staining of Cells on Coverslips
- Protocol for the Preparation and Fluorescent ICC Staining of Non-adherent Cells
- Protocol for the Preparation and Fluorescent ICC Staining of Stem Cells on Coverslips
- Protocol for the Preparation of a Cell Smear for Non-adherent Cell ICC - Graphic
- R&D Systems Quality Control Western Blot Protocol
- TUNEL and Active Caspase-3 Detection by IHC/ICC Protocol
- The Importance of IHC/ICC Controls
- Troubleshooting Guide: Western Blot Figures
- Western Blot Conditions
- Western Blot Protocol
- Western Blot Protocol for Cell Lysates
- Western Blot Troubleshooting
- Western Blot Troubleshooting Guide
- View all Protocols, Troubleshooting, Illustrated assays and Webinars