Sortilin (neurotensin receptor 3, glycoprotein 95) is a 95 kDa Type I transmembrane monomeric glycoprotein that is one of five known members of the mammalian vacuolar protein sorting 10p domain (Vps10p-D) family of sorting receptors (1, 2). Mouse preprosortilin is processed by signal sequence cleavage followed by propeptide cleavage at a furin recognition site. The cationic propeptide exhibits pH-dependent high affinity binding that blocks the Sortilin ligand binding site both pre‑ and post-cleavage (3). The extracellular/luminal sequence comprises the Vps10p domain, including 10 conserved cysteines (10CC) essential for ligand binding (2). The cytoplasmic domain sorting motifs confer all trafficking during synthesis, targeting to lysosomes, endocytosis and Golgi-endosome transport; as little as 10% may be found on the cell surface (4). Mature mouse Sortilin shares 98% amino acid (aa) identity with rat, and 91% aa identity with human and canine sortilin. During murine development, sortilin is mainly expressed in the nervous system (5), where it is a receptor for neuropeptides including neurotensin, nerve growth factor (NGF) and brain-derived neurotrophic factor (BDNF) (6‑9). ProNGF (or the NGF propeptide alone) binds sortilin with a much higher affinity (KD ~5-8 nM) than does mature NGF (KD ~90 nM). The complex of sortilin, pro‑NGF and the receptor p75ntr results in endocytosis of proNGF and induction of apoptosis (7). Similar results have been obtained with proBDNF and BDNF (8‑9). Sortilin is expressed in other tissues including testis, skeletal muscle and fat (1, 10). It is essential and sufficient for biogenesis of Glut4 storage vesicles necessary for insulin responsiveness in adipocytes (10). Sortilin also binds lipoprotein lipase (11), apoE (2) and RAP (1, 11). Binding is competitive, indicating that although unrelated, targets likely bind the same site.


Key Product Details
Species Reactivity
Validated:
Mouse
Cited:
Human, Mouse, Rat
Applications
Validated:
Immunohistochemistry, Western Blot, Blockade of Receptor-ligand Interaction, Simple Western
Cited:
Immunohistochemistry, Western Blot, Immunocytochemistry, In vivo assay
Label
Unconjugated
Antibody Source
Polyclonal Goat IgG
Loading...
Product Specifications
Immunogen
Mouse myeloma cell line NS0-derived recombinant mouse Sortilin
Gly76-Asn753
Accession # Q6PHU5
Gly76-Asn753
Accession # Q6PHU5
Specificity
Detects mouse Sortilin in direct ELISAs and Western blots. In direct ELISAs and Western blots, approximately 30% cross‑reactivity with recombinant human Sortilin is observed.
Clonality
Polyclonal
Host
Goat
Isotype
IgG
Endotoxin Level
<0.10 EU per 1 μg of the antibody by the LAL method.
Scientific Data Images for Mouse Sortilin Antibody
Detection of Mouse Sortilin by Western Blot.
Western blot shows lysates of mouse brain (total) tissue, mouse brain (cerebellum) tissue, and mouse brain (cortex) tissue. PVDF membrane was probed with 1 µg/mL of Goat Anti-Mouse Sortilin Antigen Affinity-purified Polyclonal Antibody (Catalog # AF2934) followed by HRP-conjugated Anti-Goat IgG Secondary Antibody (Catalog # HAF017). A specific band was detected for Sortilin at approximately 110 kDa (as indicated). This experiment was conducted under reducing conditions and using Immunoblot Buffer Group 1.Sortilin in Mouse Brain.
Sortilin was detected in perfusion fixed frozen sections of mouse brain (rostral ventral medulla) using Goat Anti-Mouse Sortilin Antigen Affinity-purified Polyclonal Antibody (Catalog # AF2934) at 1.7 µg/mL overnight at 4 °C. Tissue was stained using the Anti-Goat HRP-DAB Cell & Tissue Staining Kit (brown; Catalog # CTS008) and counterstained with hematoxylin (blue). View our protocol for Chromogenic IHC Staining of Frozen Tissue Sections.Detection of Mouse Sortilin by Simple WesternTM.
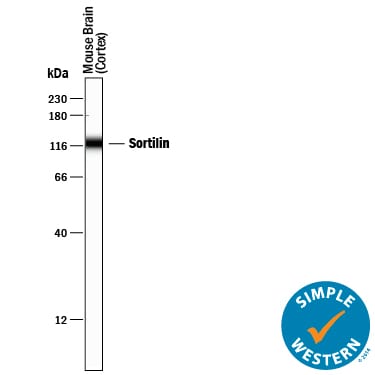
Simple Western lane view shows lysates of mouse brain (cortex) tissue, loaded at 0.2 mg/mL. A specific band was detected for Sortilin at approximately 122 kDa (as indicated) using 10 µg/mL of Goat Anti-Mouse Sortilin Antigen Affinity-purified Polyclonal Antibody (Catalog # AF2934) followed by 1:50 dilution of HRP-conjugated Anti-Goat IgG Secondary Antibody (Catalog # HAF109). This experiment was conducted under reducing conditions and using the 12-230 kDa separation system.Detection of Mouse Sortilin by Western Blot
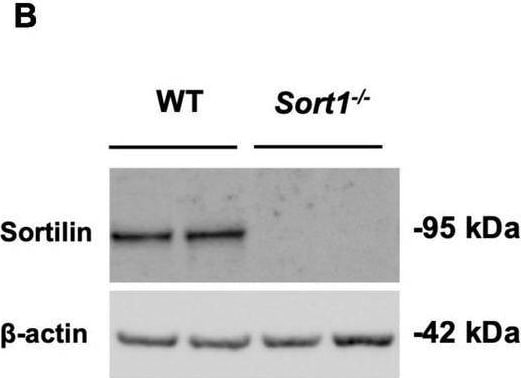
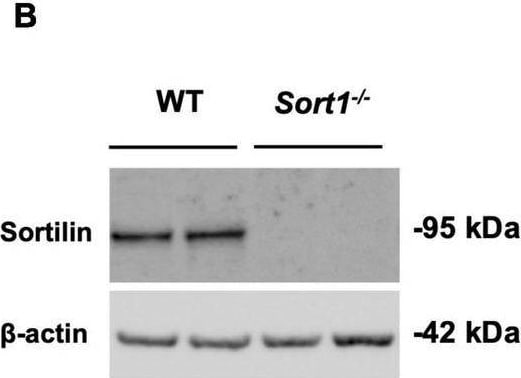
Primary cultured Schwann cells express sortilin. (A) Double-labeled immunofluorescence microscopy of primary cultured Schwann cells show sortilin (green) in wild-type (WT) Schwann cells (red; identified by the Schwann cell marker s100 beta ), but not in cells derived from Sort1–/– rat sciatic nerves. Nuclei are labeled with Hoechst (blue). Scale bar 50 μm. (B) Immunoblot analysis of sortilin in lysates of WT or Sort1–/– primary Schwann cells shows sortilin expression in WT. (C) The viability of Sort1–/– Schwann cells is like WT Schwann cells with (95.89 ± 1.55% vs. 94.30 ± 1.77%, p > 0.99) or without NT-3 stimulation (93.8 ± 1.52% vs. 93.29 ± 1.89%, p > 0.99). Viable cells were identified in Schwann cell cultures 24 h after stimulation with or without NT-3 by calcein (green) fluorescence, while dead cells were identified by ethidium homodimer-1 (red) fluorescence. Scale bar 100 μm. Viability reflects mean% calcein-positive cells ± SEM, n = 4. (D) Schwann cell proliferation (percentage of Ki-67% cells) was not significantly different in cultures of Sort1–/– Schwann cells compared to WT Schwann cell cultures with (4.82 ± 1.54% vs. 1.05 ± 0.25%, p = 0.4381) or without NT-3 stimulation (4.09 ± 1.98% vs. 1.37 ± 0.56%, p > 0.99). The proliferation marker, Ki-67 (green), was identified by immunofluorescence in Schwann cell cultures 48 h after stimulation with or without NT-3. Nuclei are labeled with Hoechst (blue). Scale bar 100 μm. Proliferation rates reflect% Ki-67 positive cells ± S.E.M., n = 3. Statistical significance was analyzed using two-way ANOVA with Bonferroni’s multiple comparisons post hoc test. Image collected and cropped by CiteAb from the following open publication (https://pubmed.ncbi.nlm.nih.gov/35634462), licensed under a CC-BY license. Not internally tested by R&D Systems.Detection of Mouse Sortilin by Western Blot
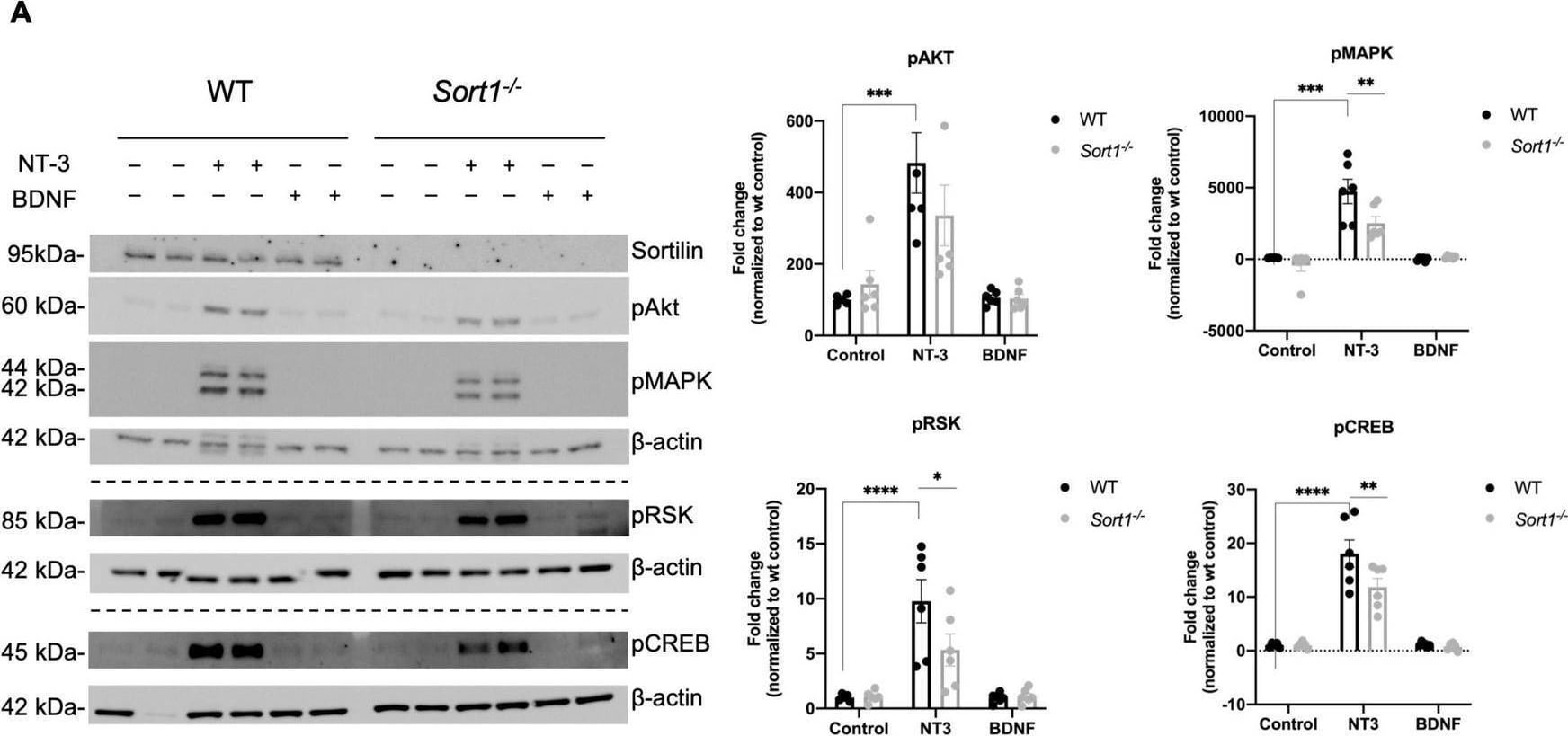
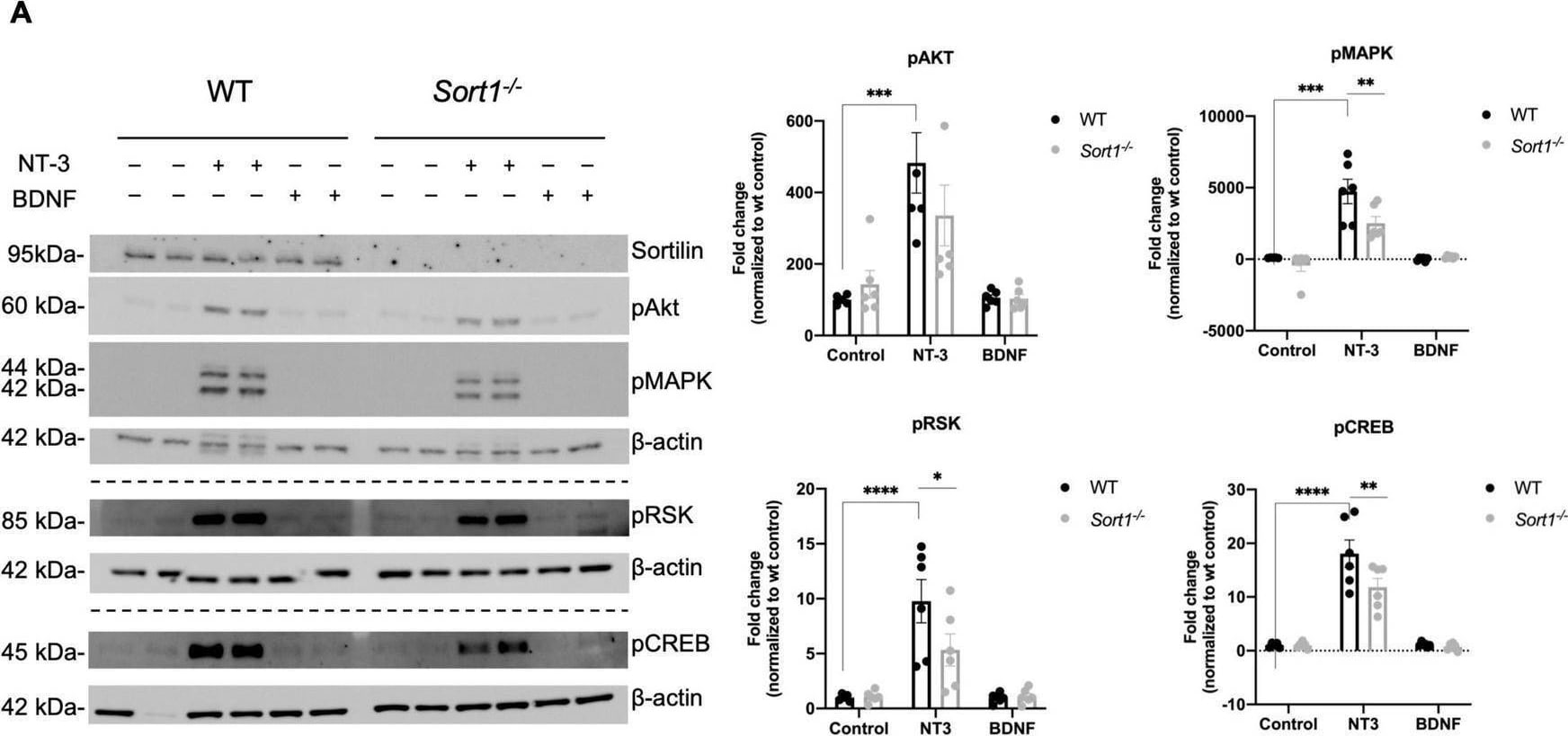
Neurotrophin signaling is decreased in Sort1–/– Schwann cells. (A) pAkt, pMAPK, pRSK, and pCREB were quantified by densitometry of immunoblots from lysates of NT-3 or BDNF stimulated wild-type (WT) or Sort1–/– primary cultures of Schwann cells and normalized against beta -actin. pMAPK, pRSK, and pCREB levels are significantly lower in Sort1–/– Schwann cells compared to WT Schwann cells. Data represent the mean ± SEM for n = 3. (B) The relative wound healing closure time was similar in Sort1–/– Schwann cells relative to WT Schwann cells with or without 10% FBS (positive control) or NT-3. Furthermore, the relative wound healing closure time indicates that NT-3 does not increase the relative wound healing closure time significantly in WT Schwann cells compared to unstimulated controls; 10% FBS significantly increased the relative wound healing closure time at 27–39 h (*p < 0.05). (C)Sort1–/– Schwann cells showed similar myelin formation relative to WT Schwann cells. Co-cultures of DRG neurons and Schwann cells were fixed 11 days after myelin induction with or without NT-3 stimulation. The myelin segments were identified with an antibody against myelin basic protein (MBP; green) and the neurites were identified with antibodies against neurofilament (red). Hoechst identifies nuclei (blue). Statistical significance was determined by two-way ANOVA with Bonferroni’s multiple comparisons post hoc test. Data represents mean ± SEM, n = 3 (*p < 0.05, **p < 0.005, ***p < 0.001, ****p < 0.0001). Image collected and cropped by CiteAb from the following open publication (https://pubmed.ncbi.nlm.nih.gov/35634462), licensed under a CC-BY license. Not internally tested by R&D Systems.Detection of Mouse Sortilin by Western Blot
Primary cultured Schwann cells express sortilin. (A) Double-labeled immunofluorescence microscopy of primary cultured Schwann cells show sortilin (green) in wild-type (WT) Schwann cells (red; identified by the Schwann cell marker s100 beta ), but not in cells derived from Sort1–/– rat sciatic nerves. Nuclei are labeled with Hoechst (blue). Scale bar 50 μm. (B) Immunoblot analysis of sortilin in lysates of WT or Sort1–/– primary Schwann cells shows sortilin expression in WT. (C) The viability of Sort1–/– Schwann cells is like WT Schwann cells with (95.89 ± 1.55% vs. 94.30 ± 1.77%, p > 0.99) or without NT-3 stimulation (93.8 ± 1.52% vs. 93.29 ± 1.89%, p > 0.99). Viable cells were identified in Schwann cell cultures 24 h after stimulation with or without NT-3 by calcein (green) fluorescence, while dead cells were identified by ethidium homodimer-1 (red) fluorescence. Scale bar 100 μm. Viability reflects mean% calcein-positive cells ± SEM, n = 4. (D) Schwann cell proliferation (percentage of Ki-67% cells) was not significantly different in cultures of Sort1–/– Schwann cells compared to WT Schwann cell cultures with (4.82 ± 1.54% vs. 1.05 ± 0.25%, p = 0.4381) or without NT-3 stimulation (4.09 ± 1.98% vs. 1.37 ± 0.56%, p > 0.99). The proliferation marker, Ki-67 (green), was identified by immunofluorescence in Schwann cell cultures 48 h after stimulation with or without NT-3. Nuclei are labeled with Hoechst (blue). Scale bar 100 μm. Proliferation rates reflect% Ki-67 positive cells ± S.E.M., n = 3. Statistical significance was analyzed using two-way ANOVA with Bonferroni’s multiple comparisons post hoc test. Image collected and cropped by CiteAb from the following open publication (https://pubmed.ncbi.nlm.nih.gov/35634462), licensed under a CC-BY license. Not internally tested by R&D Systems.Detection of Mouse Sortilin by Western Blot
Neurotrophin signaling is decreased in Sort1–/– Schwann cells. (A) pAkt, pMAPK, pRSK, and pCREB were quantified by densitometry of immunoblots from lysates of NT-3 or BDNF stimulated wild-type (WT) or Sort1–/– primary cultures of Schwann cells and normalized against beta -actin. pMAPK, pRSK, and pCREB levels are significantly lower in Sort1–/– Schwann cells compared to WT Schwann cells. Data represent the mean ± SEM for n = 3. (B) The relative wound healing closure time was similar in Sort1–/– Schwann cells relative to WT Schwann cells with or without 10% FBS (positive control) or NT-3. Furthermore, the relative wound healing closure time indicates that NT-3 does not increase the relative wound healing closure time significantly in WT Schwann cells compared to unstimulated controls; 10% FBS significantly increased the relative wound healing closure time at 27–39 h (*p < 0.05). (C)Sort1–/– Schwann cells showed similar myelin formation relative to WT Schwann cells. Co-cultures of DRG neurons and Schwann cells were fixed 11 days after myelin induction with or without NT-3 stimulation. The myelin segments were identified with an antibody against myelin basic protein (MBP; green) and the neurites were identified with antibodies against neurofilament (red). Hoechst identifies nuclei (blue). Statistical significance was determined by two-way ANOVA with Bonferroni’s multiple comparisons post hoc test. Data represents mean ± SEM, n = 3 (*p < 0.05, **p < 0.005, ***p < 0.001, ****p < 0.0001). Image collected and cropped by CiteAb from the following open publication (https://pubmed.ncbi.nlm.nih.gov/35634462), licensed under a CC-BY license. Not internally tested by R&D Systems.Applications for Mouse Sortilin Antibody
Application
Recommended Usage
Blockade of Receptor-ligand Interaction
Immunohistochemistry
1-15 µg/mL
Sample: Perfusion fixed frozen sections of mouse brain (rostral ventral medulla)
Sample: Perfusion fixed frozen sections of mouse brain (rostral ventral medulla)
Simple Western
10 µg/mL
Sample: Mouse brain (cortex) tissue
Sample: Mouse brain (cortex) tissue
Western Blot
1 µg/mL
Sample: Mouse brain (total) tissue, mouse brain (cerebellum) tissue, and mouse brain (cortex) tissue
Sample: Mouse brain (total) tissue, mouse brain (cerebellum) tissue, and mouse brain (cortex) tissue
Reviewed Applications
Read 8 reviews rated 5 using AF2934 in the following applications:
Formulation, Preparation, and Storage
Purification
Antigen Affinity-purified
Reconstitution
Reconstitute at 0.2 mg/mL in sterile PBS. For liquid material, refer to CoA for concentration.
Loading...
Formulation
Lyophilized from a 0.2 μm filtered solution in PBS with Trehalose. *Small pack size (SP) is supplied either lyophilized or as a 0.2 µm filtered solution in PBS.
Shipping
Lyophilized product is shipped at ambient temperature. Liquid small pack size (-SP) is shipped with polar packs. Upon receipt, store immediately at the temperature recommended below.
Stability & Storage
Use a manual defrost freezer and avoid repeated freeze-thaw cycles.
- 12 months from date of receipt, -20 to -70 °C as supplied.
- 1 month, 2 to 8 °C under sterile conditions after reconstitution.
- 6 months, -20 to -70 °C under sterile conditions after reconstitution.
Calculators
Background: Sortilin
References
- Petersen, C.M. et al. (1997) J. Biol. Chem. 272:3599.
- Westergaard, U.B. et al. (2004) J. Biol. Chem. 279:50221.
- Petersen, C.M. et al. (1998) EMBO J. 18:595.
- Nielsen, M.S. et al. (2001) EMBO J. 20:2180.
- Hermans-Borgmeyer, I. et al. (1999) Mol. Brain Res. 65:216.
- Mazella, J. et al. (1998) J. Biol. Chem. 273:26273.
- Nykjaer, A et al. (2004) Nature 427:843.
- Teng, H.K. et al. (2005) J. Neurosci. 25:5455.
- Chen, Z.-Y. et al. (2004) J. Neurosci. 25:6156.
- Shi, J and K.V. Kandror (2005) Dev. Cell 9:99.
- Nielsen, M.S. et al. (1999) J. Biol. Chem. 274:8832.
Alternate Names
Gp95, Ntr3, SORT1
Gene Symbol
SORT1
UniProt
Additional Sortilin Products
Product Documents for Mouse Sortilin Antibody
Certificate of Analysis
To download a Certificate of Analysis, please enter a lot or batch number in the search box below.
Note: Certificate of Analysis not available for kit components.
Product Specific Notices for Mouse Sortilin Antibody
For research use only
Related Research Areas
Citations for Mouse Sortilin Antibody
Customer Reviews for Mouse Sortilin Antibody (8)
5 out of 5
8 Customer Ratings
Have you used Mouse Sortilin Antibody?
Submit a review and receive an Amazon gift card!
$25/€18/£15/$25CAN/¥2500 Yen for a review with an image
$10/€7/£6/$10CAN/¥1110 Yen for a review without an image
Submit a review
Customer Images
Showing
1
-
5 of
8 reviews
Showing All
Filter By:
-
Application: Immunohistochemistry-FrozenSample Tested: Dorsal root gangliaSpecies: MouseVerified Customer | Posted 12/19/2014
-
Application: ImmunofluorescenceSample Tested: Primary dorsal root ganglia neuronsSpecies: MouseVerified Customer | Posted 12/19/2014
-
Application: Immunohistochemistry-FrozenSample Tested: Spinal cord cryosections and dorsal root gangliaSpecies: MouseVerified Customer | Posted 12/19/2014
-
Application: ImmunoprecipitationSample Tested: Endogenous mouse sortilinSpecies: MouseVerified Customer | Posted 12/19/2014
-
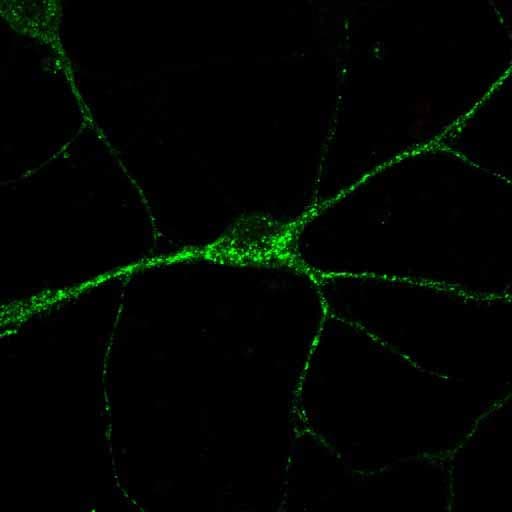
Application: ImmunocytochemistrySample Tested: Primary neuronsSpecies: MouseVerified Customer | Posted 12/19/2014ICC analysis of Sortilin in a DIV12 hippocampal neuron
-
Application: Immunohistochemistry-FrozenSample Tested: CryosectionsSpecies: MouseVerified Customer | Posted 12/19/2014
-
Application: ImmunoprecipitationSample Tested: mouse cryosectionsSpecies: MouseVerified Customer | Posted 12/16/2014
-
Application: ImmunocytochemistrySample Tested: mouse cryosectionsSpecies: MouseVerified Customer | Posted 12/16/2014
There are no reviews that match your criteria.
Protocols
Find general support by application which include: protocols, troubleshooting, illustrated assays, videos and webinars.
- Antigen Retrieval Protocol (PIER)
- Antigen Retrieval for Frozen Sections Protocol
- Appropriate Fixation of IHC/ICC Samples
- Cellular Response to Hypoxia Protocols
- Chromogenic IHC Staining of Formalin-Fixed Paraffin-Embedded (FFPE) Tissue Protocol
- Chromogenic Immunohistochemistry Staining of Frozen Tissue
- ClariTSA™ Fluorophore Kits
- Detection & Visualization of Antibody Binding
- Fluorescent IHC Staining of Frozen Tissue Protocol
- Graphic Protocol for Heat-induced Epitope Retrieval
- Graphic Protocol for the Preparation and Fluorescent IHC Staining of Frozen Tissue Sections
- Graphic Protocol for the Preparation and Fluorescent IHC Staining of Paraffin-embedded Tissue Sections
- Graphic Protocol for the Preparation of Gelatin-coated Slides for Histological Tissue Sections
- IHC Sample Preparation (Frozen sections vs Paraffin)
- Immunofluorescent IHC Staining of Formalin-Fixed Paraffin-Embedded (FFPE) Tissue Protocol
- Immunohistochemistry (IHC) and Immunocytochemistry (ICC) Protocols
- Immunohistochemistry Frozen Troubleshooting
- Immunohistochemistry Paraffin Troubleshooting
- Preparing Samples for IHC/ICC Experiments
- Preventing Non-Specific Staining (Non-Specific Binding)
- Primary Antibody Selection & Optimization
- Protocol for Heat-Induced Epitope Retrieval (HIER)
- Protocol for Making a 4% Formaldehyde Solution in PBS
- Protocol for VisUCyte™ HRP Polymer Detection Reagent
- Protocol for the Preparation & Fixation of Cells on Coverslips
- Protocol for the Preparation and Chromogenic IHC Staining of Frozen Tissue Sections
- Protocol for the Preparation and Chromogenic IHC Staining of Frozen Tissue Sections - Graphic
- Protocol for the Preparation and Chromogenic IHC Staining of Paraffin-embedded Tissue Sections
- Protocol for the Preparation and Chromogenic IHC Staining of Paraffin-embedded Tissue Sections - Graphic
- Protocol for the Preparation and Fluorescent IHC Staining of Frozen Tissue Sections
- Protocol for the Preparation and Fluorescent IHC Staining of Paraffin-embedded Tissue Sections
- Protocol for the Preparation of Gelatin-coated Slides for Histological Tissue Sections
- R&D Systems Quality Control Western Blot Protocol
- TUNEL and Active Caspase-3 Detection by IHC/ICC Protocol
- The Importance of IHC/ICC Controls
- Troubleshooting Guide: Immunohistochemistry
- Troubleshooting Guide: Western Blot Figures
- Western Blot Conditions
- Western Blot Protocol
- Western Blot Protocol for Cell Lysates
- Western Blot Troubleshooting
- Western Blot Troubleshooting Guide
- View all Protocols, Troubleshooting, Illustrated assays and Webinars
Loading...