Human alpha 2-macroglobulin (h alpha 2M) is a serum glycoprotein that has sequence similarity to other members of the alpha 2M family including complement components C3, C4 and C5 (1). alpha 2M is synthesized as a polypeptide of 1474 amino acids with a signal peptide (23 residues) (2). The mature protein is a tetramer (720 kDa) of 4 identical subunits (180 kDa), which form two disulfide bond-linked dimers. As a general and irreversible protease inhibitor implicated in many processes, alpha 2M is able to inhibit all four classes of proteases by a unique trapping mechanism. The bait region of h alpha 2M (residues 690‑728) contains specific cleavage sites for different proteases. The cleavage of the bait region by a protease induces a conformation change in alpha 2M, which then traps and forms a covalent bond with the protease. The trapped protease remains active against small peptide substrates but loses its ability to interact with large protein substrates or inhibitors.
Human alpha 2‑Macroglobulin Antibody
R&D Systems | Catalog # AF1938

Key Product Details
Species Reactivity
Validated:
Human
Cited:
Human, Rat
Applications
Validated:
Western Blot, Immunocytochemistry, Simple Western, Immunoprecipitation
Cited:
Immunohistochemistry, Western Blot, Immunocytochemistry
Label
Unconjugated
Antibody Source
Polyclonal Goat IgG
Loading...
Product Specifications
Immunogen
Human plasma-derived alpha 2-Macroglobulin
Specificity
Detects human alpha
Clonality
Polyclonal
Host
Goat
Isotype
IgG
Scientific Data Images for Human alpha 2‑Macroglobulin Antibody
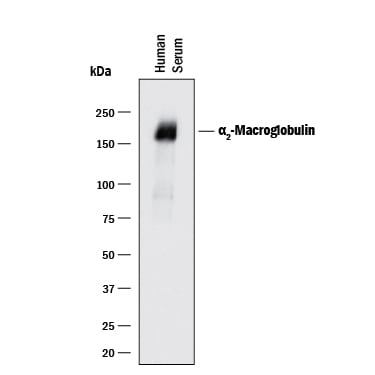
Detection of Human alpha 2‑Macroglobulin by Western Blot.
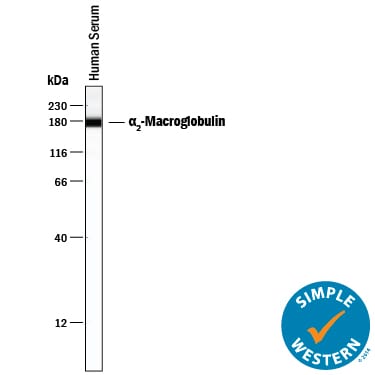
Western blot shows human serum. PVDF membrane was probed with 0.5 µg/mL of Goat Anti-Human a2-Macroglobulin Antigen Affinity-purified Polyclonal Antibody (Catalog # AF1938) followed by HRP-conjugated Anti-Goat IgG Secondary Antibody (Catalog # HAF017). A specific band was detected for a2-Macroglobulin at approximately 180 kDa (as indicated). This experiment was conducted under reducing conditions and using Immunoblot Buffer Group 1.Detection of Human alpha 2‑Macroglobulin by Simple WesternTM.
Simple Western lane view shows human serum, loaded at 0.2 mg/mL. A specific band was detected for a2-Macroglobulin at approximately 178 kDa (as indicated) using 1 µg/mL of Goat Anti-Human a2-Macroglobulin Antigen Affinity-purified Polyclonal Antibody (Catalog # AF1938) followed by 1:50 dilution of HRP-conjugated Anti-Goat IgG Secondary Antibody (Catalog # HAF109). This experiment was conducted under reducing conditions and using the 12-230 kDa separation system.alpha 2‑Macroglobulin in human PBMCs.
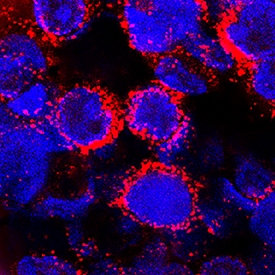
a2-Macroglobulin was detected in immersion fixed human peripheral blood mononuclear cells (PBMCs) using Goat Anti-Human a2-Macroglobulin Antigen Affinity-purified Polyclonal Antibody (Catalog # AF1938) at 15 µg/mL for 3 hours at room temperature. Cells were stained using the NorthernLights™ 557-conjugated Anti-Goat IgG Secondary Antibody (red; Catalog # NL001) and counterstained with DAPI (blue). Specific staining was localized to cytoplasm. View our protocol for Fluorescent ICC Staining of Cells on Coverslips.Applications for Human alpha 2‑Macroglobulin Antibody
Application
Recommended Usage
Immunocytochemistry
5-15 µg/mL
Sample: Immersion fixed human peripheral blood mononuclear cells (PBMCs)
Sample: Immersion fixed human peripheral blood mononuclear cells (PBMCs)
Immunoprecipitation
25 µg/mL
Sample: Conditioned cell culture medium spiked with Human alpha 2-Macroglobulin (Catalog # 1938-PI), see our available Western blot detection antibodies
Sample: Conditioned cell culture medium spiked with Human alpha 2-Macroglobulin (Catalog # 1938-PI), see our available Western blot detection antibodies
Simple Western
1 µg/mL
Sample: Human serum
Sample: Human serum
Western Blot
0.5 µg/mL
Sample: Human serum
Sample: Human serum
Reviewed Applications
Read 4 reviews rated 4.3 using AF1938 in the following applications:
Formulation, Preparation, and Storage
Purification
Antigen Affinity-purified
Reconstitution
Reconstitute at 0.2 mg/mL in sterile PBS. For liquid material, refer to CoA for concentration.
Loading...
Formulation
Lyophilized from a 0.2 μm filtered solution in PBS with Trehalose. *Small pack size (SP) is supplied either lyophilized or as a 0.2 µm filtered solution in PBS.
Shipping
Lyophilized product is shipped at ambient temperature. Liquid small pack size (-SP) is shipped with polar packs. Upon receipt, store immediately at the temperature recommended below.
Stability & Storage
Use a manual defrost freezer and avoid repeated freeze-thaw cycles.
- 12 months from date of receipt, -20 to -70 °C as supplied.
- 1 month, 2 to 8 °C under sterile conditions after reconstitution.
- 6 months, -20 to -70 °C under sterile conditions after reconstitution.
Calculators
Background: alpha 2-Macroglobulin
References
- Sottrup-Jensen, L. et al. (1985) Proc. Natl. Acad. Sci. USA 82:9.
- Kan, C.C. et al. (1985) Proc. Natl. Acad. Sci. USA 82:2282.
Alternate Names
A2M, alpha 2Macroglobulin, CPAMD5
Entrez Gene IDs
2 (Human)
Gene Symbol
A2M
Additional alpha 2-Macroglobulin Products
Product Documents for Human alpha 2‑Macroglobulin Antibody
Certificate of Analysis
To download a Certificate of Analysis, please enter a lot or batch number in the search box below.
Note: Certificate of Analysis not available for kit components.
Product Specific Notices for Human alpha 2‑Macroglobulin Antibody
For research use only
Citations for Human alpha 2‑Macroglobulin Antibody
Customer Reviews for Human alpha 2‑Macroglobulin Antibody (4)
4.3 out of 5
4 Customer Ratings
Have you used Human alpha 2‑Macroglobulin Antibody?
Submit a review and receive an Amazon gift card!
$25/€18/£15/$25CAN/¥2500 Yen for a review with an image
$10/€7/£6/$10CAN/¥1110 Yen for a review without an image
Submit a review
Customer Images
Showing
1
-
4 of
4 reviews
Showing All
Filter By:
-
Application: MicroarraysSample Tested: EDTA PlasmaSpecies: HumanVerified Customer | Posted 03/11/2019
-
Application: ELISASample Tested: EDTA PlasmaSpecies: HumanVerified Customer | Posted 12/20/2018
-
Application: ELISASample Tested: Plasma and SerumSpecies: Human and MouseVerified Customer | Posted 11/07/2018
-
Application: MicroarraySample Tested: EDTA PlasmaSpecies: HumanVerified Customer | Posted 10/31/2018
There are no reviews that match your criteria.
Protocols
Find general support by application which include: protocols, troubleshooting, illustrated assays, videos and webinars.
- Appropriate Fixation of IHC/ICC Samples
- Cellular Response to Hypoxia Protocols
- ClariTSA™ Fluorophore Kits
- Detection & Visualization of Antibody Binding
- ICC Cell Smear Protocol for Suspension Cells
- ICC Immunocytochemistry Protocol Videos
- ICC for Adherent Cells
- Immunocytochemistry (ICC) Protocol
- Immunocytochemistry Troubleshooting
- Immunofluorescence of Organoids Embedded in Cultrex Basement Membrane Extract
- Immunohistochemistry (IHC) and Immunocytochemistry (ICC) Protocols
- Immunoprecipitation Protocol
- Preparing Samples for IHC/ICC Experiments
- Preventing Non-Specific Staining (Non-Specific Binding)
- Primary Antibody Selection & Optimization
- Protocol for VisUCyte™ HRP Polymer Detection Reagent
- Protocol for the Fluorescent ICC Staining of Cell Smears - Graphic
- Protocol for the Fluorescent ICC Staining of Cultured Cells on Coverslips - Graphic
- Protocol for the Preparation and Fluorescent ICC Staining of Cells on Coverslips
- Protocol for the Preparation and Fluorescent ICC Staining of Non-adherent Cells
- Protocol for the Preparation and Fluorescent ICC Staining of Stem Cells on Coverslips
- Protocol for the Preparation of a Cell Smear for Non-adherent Cell ICC - Graphic
- R&D Systems Quality Control Western Blot Protocol
- TUNEL and Active Caspase-3 Detection by IHC/ICC Protocol
- The Importance of IHC/ICC Controls
- Troubleshooting Guide: Western Blot Figures
- Western Blot Conditions
- Western Blot Protocol
- Western Blot Protocol for Cell Lysates
- Western Blot Troubleshooting
- Western Blot Troubleshooting Guide
- View all Protocols, Troubleshooting, Illustrated assays and Webinars
Loading...
Associated Pathways
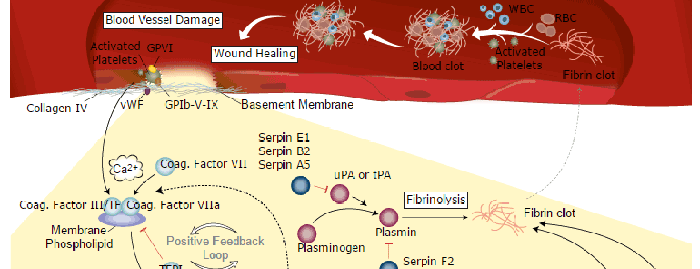
 Blood Coagulation Signaling Pathways
Blood Coagulation Signaling Pathways