Cathepsin D is a lysosomal aspartic protease of the pepsin family (1). Human cathepsin D is synthesized as a precursor protein, consisting of a signal peptide (aa 1‑18), a propeptide (aa 19-64), and a mature chain (aa 65‑412) (2‑4). The mature chain can be processed further to the light (aa 65‑161) and heavy (aa 169‑412) chains. It is expressed in most cells and overexpressed in breast cancer cells (5). It is a major enzyme in protein degradation in lysosomes, and also involved in the presentation of antigenic peptides. Mice deficient in this enzyme showed a progressive atrophy of the intestinal mucosa, a massive destruction of lymphoid organs, and a profound neuronal ceroid lipofucinosis, indicating that cathepsin D is essential for proteolysis of proteins regulating cell growth and tissue homeostasis (6). Cathepsin D secreted from human prostate carcinoma cells are responsible for the generation of angiostatin, a potent endogeneous inhibitor of angiogenesis (6).

Key Product Details
Species Reactivity
Validated:
Human
Cited:
Human
Applications
Validated:
Immunohistochemistry, Western Blot, Simple Western
Cited:
Western Blot
Label
Unconjugated
Antibody Source
Monoclonal Mouse IgG1 Clone # 185111
Loading...
Product Specifications
Immunogen
Mouse myeloma cell line NS0-derived recombinant human Cathepsin D
Leu21-Leu412
Accession # P07339
Leu21-Leu412
Accession # P07339
Specificity
Detects human Cathepsin D in direct ELISAs and Western blots. It recognizes both the pro and mature forms of recombinant human (rh) Cathepsin D. In direct ELISAs, no cross-reactivity with rhCathepsin A, rhCathepsin B, rhCathepsin C, rhCathepsin L, rhCathepsin O, rhCathepsin S, rhCathepsin Z, or recombinant mouse Cathepsin D is observed. In Western blots, 100% cross-reactivity with rhCathepsin E and rmCathepsin D is observed and no cross-reactivity with rhBACE-1 is observed.
Clonality
Monoclonal
Host
Mouse
Isotype
IgG1
Scientific Data Images for Human Cathepsin D Antibody
Detection of Human Cathepsin D by Western Blot.
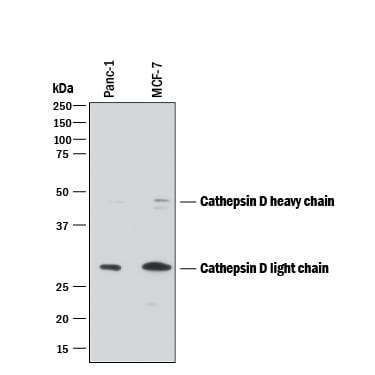
Western blot shows lysates of PANC-1 human pancreatic carcinoma cell line and MCF-7 human breast cancer cell line. PVDF membrane was probed with 0.2 µg/mL of Mouse Anti-Human Cathepsin D Monoclonal Antibody (Catalog # MAB1014) followed by HRP-conjugated Anti-Mouse IgG Secondary Antibody (Catalog # HAF018). Specific bands were detected for Cathepsin D at approximately 28 and 46 kDa (as indicated). This experiment was conducted under reducing conditions and using Immunoblot Buffer Group 1.Cathepsin D in Human Prostate Cancer Tissue.
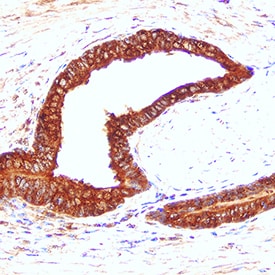
Cathepsin D was detected in immersion fixed paraffin-embedded sections of human prostate cancer tissue using Mouse Anti-Human Cathepsin D Monoclonal Antibody (Catalog # MAB1014) at 5 µg/mL for 1 hour at room temperature followed by incubation with the Anti-Mouse IgG VisUCyte™ HRP Polymer Antibody (Catalog # VC001). Tissue was stained using DAB (brown) and counterstained with hematoxylin (blue). Specific staining was localized to epithelial cell cytoplasm. View our protocol for IHC Staining with VisUCyte HRP Polymer Detection Reagents.Detection of Human Cathepsin D by Simple WesternTM.
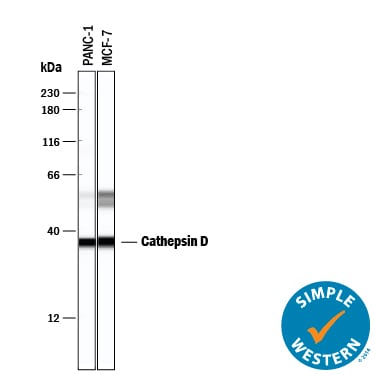
Simple Western lane view shows lysates of PANC‑1 human pancreatic carcinoma cell line and MCF‑7 human breast cancer cell line, loaded at 0.2 mg/mL. Specific bands were detected for Cathepsin D at approximately 36 and 52-57 kDa (as indicated) using 2 µg/mL of Mouse Anti-Human Cathepsin D Monoclonal Antibody (Catalog # MAB1014). This experiment was conducted under reducing conditions and using the 12-230 kDa separation system.Applications for Human Cathepsin D Antibody
Application
Recommended Usage
Immunohistochemistry
5-25 µg/mL
Sample: Immersion fixed paraffin-embedded sections of human prostate cancer tissue
Sample: Immersion fixed paraffin-embedded sections of human prostate cancer tissue
Simple Western
2 µg/mL
Sample: PANC‑1 human pancreatic carcinoma cell line and MCF‑7 human breast cancer cell line
Sample: PANC‑1 human pancreatic carcinoma cell line and MCF‑7 human breast cancer cell line
Western Blot
0.2 µg/mL
Sample: PANC‑1 human pancreatic carcinoma cell line and MCF‑7 human breast cancer cell line
Sample: PANC‑1 human pancreatic carcinoma cell line and MCF‑7 human breast cancer cell line
Reviewed Applications
Read 4 reviews rated 3.8 using MAB1014 in the following applications:
Formulation, Preparation, and Storage
Purification
Protein A or G purified from hybridoma culture supernatant
Reconstitution
Reconstitute at 0.5 mg/mL in sterile PBS. For liquid material, refer to CoA for concentration.
Loading...
Formulation
Lyophilized from a 0.2 μm filtered solution in PBS with Trehalose. *Small pack size (SP) is supplied either lyophilized or as a 0.2 µm filtered solution in PBS.
Shipping
Lyophilized product is shipped at ambient temperature. Liquid small pack size (-SP) is shipped with polar packs. Upon receipt, store immediately at the temperature recommended below.
Stability & Storage
Use a manual defrost freezer and avoid repeated freeze-thaw cycles.
- 12 months from date of receipt, -20 to -70 °C as supplied.
- 1 month, 2 to 8 °C under sterile conditions after reconstitution.
- 6 months, -20 to -70 °C under sterile conditions after reconstitution.
Calculators
Background: Cathepsin D
References
- Conner et al. in Handbook of Proteolytic Enzymes Barrett (1998) Academic Press, San Diego, p. 828.
- Faust, et al. (1985) Proc. Natl. Acad. Sci. USA 82:4910.
- Westley and May (1987) Nucl. Acid Res. 15:3773.
- Redecker, et al. (1991) DNA Cell Biol. 10:423.
- Rochefort, et al. (2000) Clin. Chim. Acta. 291:157.
- Tsukuba, et al. (2000) Mol. Cells 10:601.
Alternate Names
CTSD
Gene Symbol
CTSD
UniProt
Additional Cathepsin D Products
Product Documents for Human Cathepsin D Antibody
Certificate of Analysis
To download a Certificate of Analysis, please enter a lot or batch number in the search box below.
Note: Certificate of Analysis not available for kit components.
Product Specific Notices for Human Cathepsin D Antibody
For research use only
Citations for Human Cathepsin D Antibody
Customer Reviews for Human Cathepsin D Antibody (4)
3.8 out of 5
4 Customer Ratings
Have you used Human Cathepsin D Antibody?
Submit a review and receive an Amazon gift card!
$25/€18/£15/$25CAN/¥2500 Yen for a review with an image
$10/€7/£6/$10CAN/¥1110 Yen for a review without an image
Submit a review
Customer Images
Showing
1
-
4 of
4 reviews
Showing All
Filter By:
-
Application: ImmunohistochemistrySample Tested: Spinal cordSpecies: HumanVerified Customer | Posted 10/29/2021
-
Application: MicroarraysSample Tested: EDTA PlasmaSpecies: HumanVerified Customer | Posted 03/11/2019
-
Application: MicroarraySample Tested: EDTA PlasmaSpecies: HumanVerified Customer | Posted 11/20/2018
-
Application: ELISASample Tested: Serum and PlasmaSpecies: Human and MouseVerified Customer | Posted 11/07/2018
There are no reviews that match your criteria.
Protocols
Find general support by application which include: protocols, troubleshooting, illustrated assays, videos and webinars.
- Antigen Retrieval Protocol (PIER)
- Antigen Retrieval for Frozen Sections Protocol
- Appropriate Fixation of IHC/ICC Samples
- Cellular Response to Hypoxia Protocols
- Chromogenic IHC Staining of Formalin-Fixed Paraffin-Embedded (FFPE) Tissue Protocol
- Chromogenic Immunohistochemistry Staining of Frozen Tissue
- ClariTSA™ Fluorophore Kits
- Detection & Visualization of Antibody Binding
- Fluorescent IHC Staining of Frozen Tissue Protocol
- Graphic Protocol for Heat-induced Epitope Retrieval
- Graphic Protocol for the Preparation and Fluorescent IHC Staining of Frozen Tissue Sections
- Graphic Protocol for the Preparation and Fluorescent IHC Staining of Paraffin-embedded Tissue Sections
- Graphic Protocol for the Preparation of Gelatin-coated Slides for Histological Tissue Sections
- IHC Sample Preparation (Frozen sections vs Paraffin)
- Immunofluorescent IHC Staining of Formalin-Fixed Paraffin-Embedded (FFPE) Tissue Protocol
- Immunohistochemistry (IHC) and Immunocytochemistry (ICC) Protocols
- Immunohistochemistry Frozen Troubleshooting
- Immunohistochemistry Paraffin Troubleshooting
- Preparing Samples for IHC/ICC Experiments
- Preventing Non-Specific Staining (Non-Specific Binding)
- Primary Antibody Selection & Optimization
- Protocol for Heat-Induced Epitope Retrieval (HIER)
- Protocol for Making a 4% Formaldehyde Solution in PBS
- Protocol for VisUCyte™ HRP Polymer Detection Reagent
- Protocol for the Preparation & Fixation of Cells on Coverslips
- Protocol for the Preparation and Chromogenic IHC Staining of Frozen Tissue Sections
- Protocol for the Preparation and Chromogenic IHC Staining of Frozen Tissue Sections - Graphic
- Protocol for the Preparation and Chromogenic IHC Staining of Paraffin-embedded Tissue Sections
- Protocol for the Preparation and Chromogenic IHC Staining of Paraffin-embedded Tissue Sections - Graphic
- Protocol for the Preparation and Fluorescent IHC Staining of Frozen Tissue Sections
- Protocol for the Preparation and Fluorescent IHC Staining of Paraffin-embedded Tissue Sections
- Protocol for the Preparation of Gelatin-coated Slides for Histological Tissue Sections
- R&D Systems Quality Control Western Blot Protocol
- TUNEL and Active Caspase-3 Detection by IHC/ICC Protocol
- The Importance of IHC/ICC Controls
- Troubleshooting Guide: Immunohistochemistry
- Troubleshooting Guide: Western Blot Figures
- Western Blot Conditions
- Western Blot Protocol
- Western Blot Protocol for Cell Lysates
- Western Blot Troubleshooting
- Western Blot Troubleshooting Guide
- View all Protocols, Troubleshooting, Illustrated assays and Webinars
Loading...
Associated Pathways