Human CD63 Antibody
R&D Systems | Catalog # MAB50482
Recombinant Monoclonal Antibody.

Key Product Details
Species Reactivity
Validated:
Human
Cited:
Human, Porcine
Applications
Validated:
Immunohistochemistry, Western Blot, Flow Cytometry, Simple Western, CyTOF-ready
Cited:
Western Blot, Simple Western
Label
Unconjugated
Antibody Source
Recombinant Monoclonal Rabbit IgG Clone # 2585J
Loading...
Product Specifications
Immunogen
Chinese Hamster Ovary cell line CHO-derived human CD63
Ala103-Val203
Accession # P08962-1
Ala103-Val203
Accession # P08962-1
Specificity
Detects human CD63 in direct ELISAs.
Clonality
Monoclonal
Host
Rabbit
Isotype
IgG
Scientific Data Images for Human CD63 Antibody
Detection of Human CD63 by Western Blot.
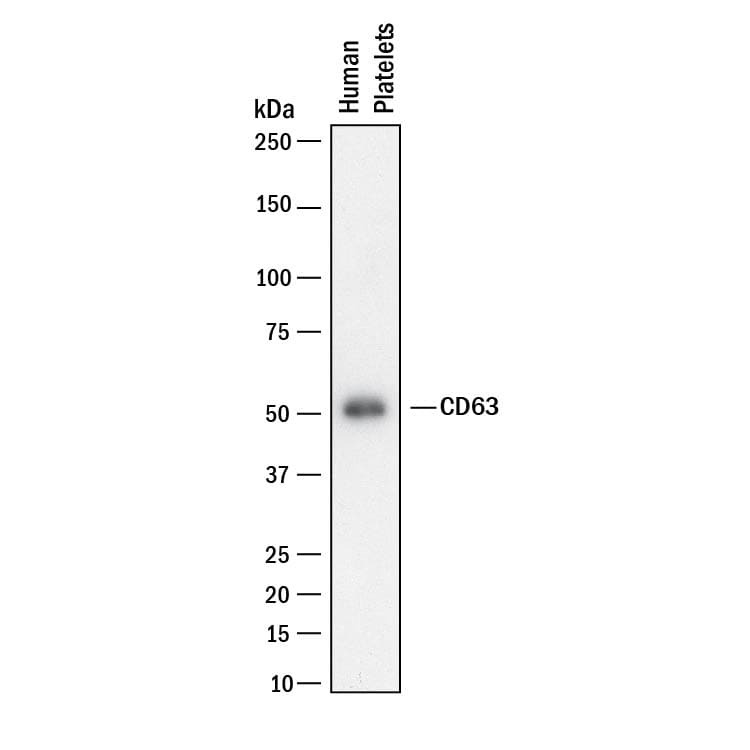
Western blot shows lysates of human platelets. PVDF membrane was probed with 0.5 µg/mL of Rabbit Anti-Human CD63 Monoclonal Antibody (Catalog # MAB50482) followed by HRP-conjugated Anti-Rabbit IgG Secondary Antibody (HAF008). A specific band was detected for CD63 at approximately 55 kDa (as indicated). This experiment was conducted under reducing conditions and using Immunoblot Buffer Group 1.CD63 in Human Lung Cancer Tissue.
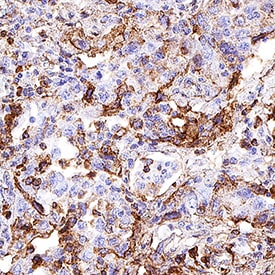
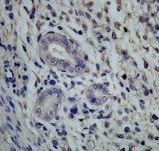
CD63 was detected in immersion fixed paraffin-embedded sections of human lung cancer tissue using Rabbit Anti-Human CD63 Monoclonal Antibody (Catalog # MAB50482) at 0.3 µg/mL for 1 hour at room temperature followed by incubation with the Anti-Rabbit IgG VisUCyte™ HRP Polymer Antibody (VC003). Before incubation with the primary antibody, tissue was subjected to heat-induced epitope retrieval using Antigen Retrieval Reagent-Basic (Catalog # CTS013). Tissue was stained using DAB (brown) and counterstained with hematoxylin (blue). Specific staining was localized to cell membrane in sperm cells. View our protocol for IHC Staining with VisUCyte HRP Polymer Detection Reagents.Detection of CD63 in Human PBMC by Flow Cytometry.
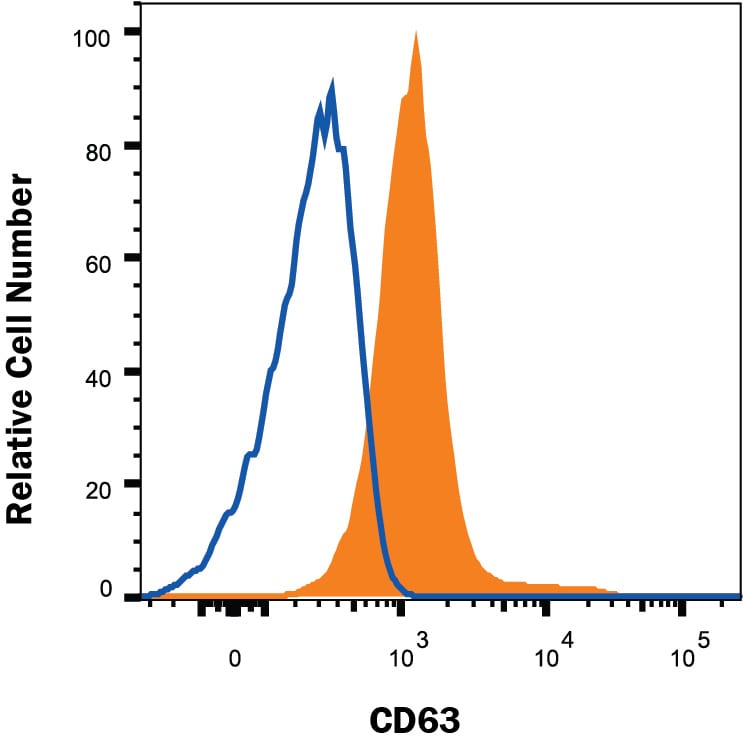
Human PBMC were stained with Rabbit Anti-Human CD63 Monoclonal Antibody (Catalog # MAB50482, filled histogram) or isotype control antibody (Catalog # MAB1050, open histogram), followed by Phycoerythrin-conjugated Anti-Rabbit IgG Secondary Antibody (F0111). To facilitate intracellular staining, cells were fixed and permeabilized with FlowX FoxP3/Transcription Factor Fixation & Perm Buffer Kit (FC012). View our protocol for Staining Intracellular Molecules.Detection of Human CD63 by Simple WesternTM.
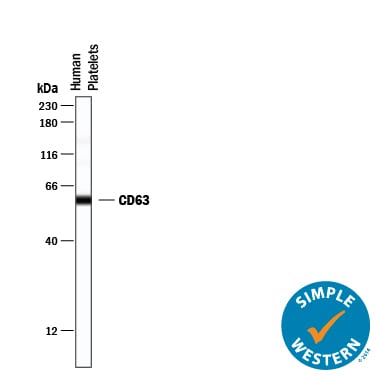
Simple Western lane view shows lysates of human platelets, loaded at 0.2 mg/mL. A specific band was detected for CD63 at approximately 59 kDa (as indicated) using 20 µg/mL of Rabbit Anti-Human CD63 Monoclonal Antibody (Catalog # MAB50482). This experiment was conducted under reducing conditions and using the 12-230 kDa separation system.Detection of CD63 in PBMC monocytes by Flow Cytometry
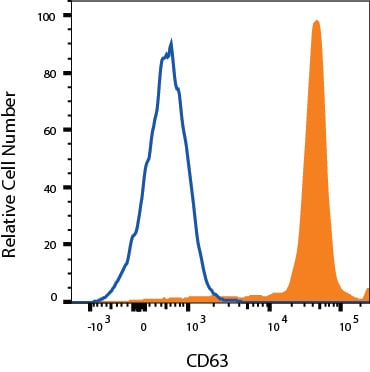
PBMC monocytes were stained with Rabbit Anti-Human CD63 Monoclonal Antibody (Catalog # MAB50482, filled histogram) or isotype control antibody (Catalog # MAB1050, open histogram) followed by Phycoerythrin-conjugated Anti-Rabbit IgG Secondary Antibody (Catalog # F0110). View our protocol for Staining Membrane-associated Proteins.Detection of CD63 by Western Blot
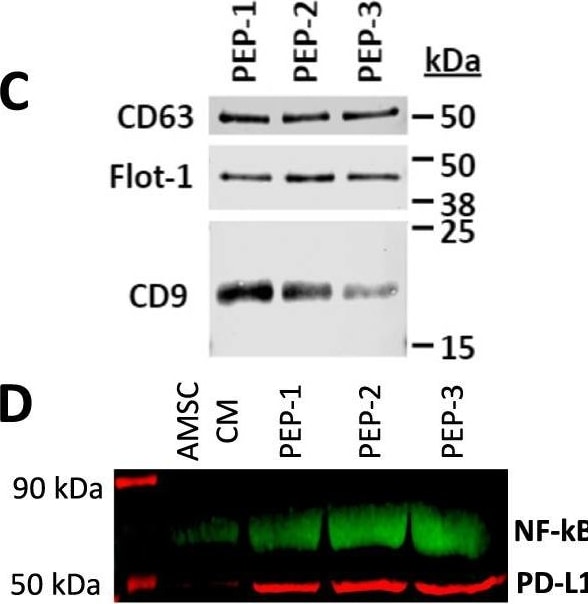
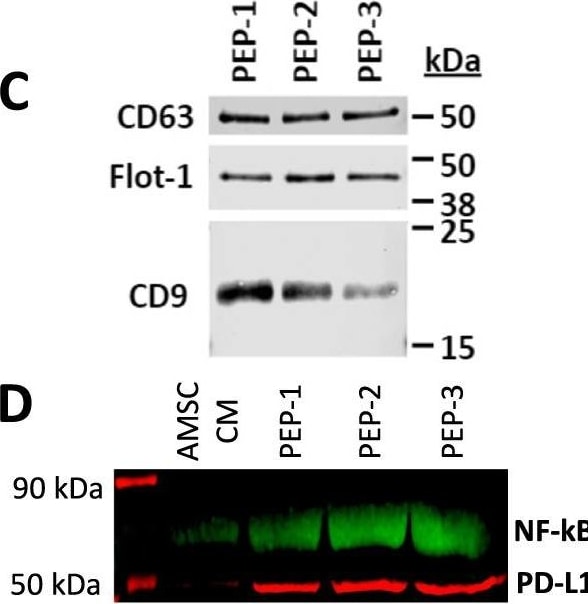
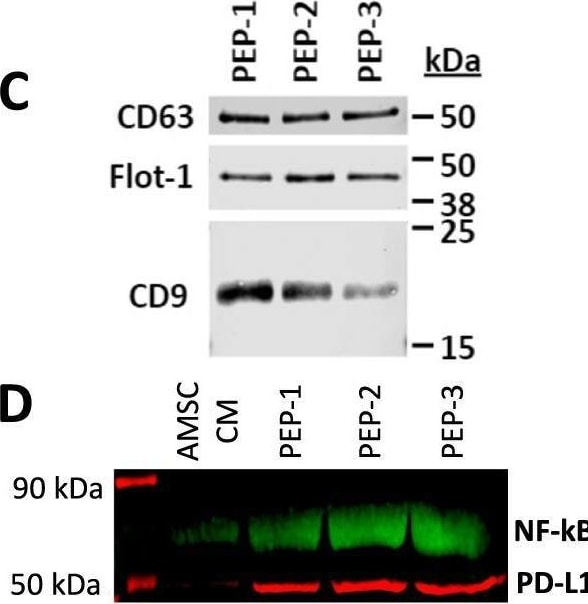
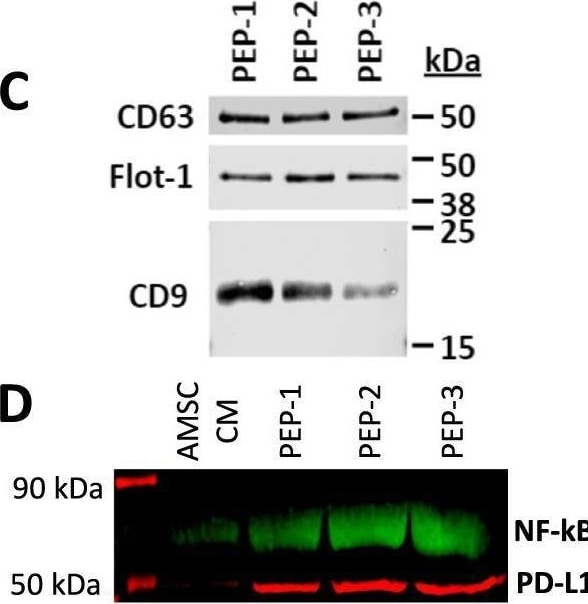
Purified exosome product (PEP) characterization, quantification, and delivery biodistribution. A NanoSight nanoparticle analysis of size distribution and concentration of PEP diluted in phosphate buffered saline 1:1000 documented 6.65 × 1012 ± 1.16 × 1011. B Transmission electron microscopy of PEP. Scale = 200 nm arrow heads pointing to EVs. C Western blot probing for CD63, CD9, and Flotillin-1 in 3 separate CGMP manufactured PEP lots. D Western blot comparison of NF-kappa B p65 and PD-L1 levels, in 3 separate CGMP manufactured PEP lots versus adipose-derived mesenchymal stem cell conditioned media (AMSC-CM). E Atomic-force microscope comparing platelet-conditioned medium EV isolation using centrifugation versus the PEP process, scale bar embedded in the image. F Representative image from Single-particle interferometric reflectance imaging sensing (SP-IRIS) analysis for presence of surface CD41a, CD9, CD63, and CD81 tetraspanins. G Graphical representation of the SP-IRIS analysis. H Quantitation of CD9, CD63 and CD81 on a CD41a captured plate documented vast majority of PEP as CD41a/CD9 positive, with smaller representation from CD63 and background CD81. Data presented as mean ± stdev. N = 3 separate CGMP manufactured PEP lots. I Pie chart representation of the exosome tetraspanin surface marker profile of CD41a captured PEP exosomes. Image collected and cropped by CiteAb from the following open publication (https://pubmed.ncbi.nlm.nih.gov/36175423), licensed under a CC-BY license. Not internally tested by R&D Systems.Detection of CD63 by Western Blot
Purified exosome product (PEP) characterization, quantification, and delivery biodistribution. A NanoSight nanoparticle analysis of size distribution and concentration of PEP diluted in phosphate buffered saline 1:1000 documented 6.65 × 1012 ± 1.16 × 1011. B Transmission electron microscopy of PEP. Scale = 200 nm arrow heads pointing to EVs. C Western blot probing for CD63, CD9, and Flotillin-1 in 3 separate CGMP manufactured PEP lots. D Western blot comparison of NF-kappa B p65 and PD-L1 levels, in 3 separate CGMP manufactured PEP lots versus adipose-derived mesenchymal stem cell conditioned media (AMSC-CM). E Atomic-force microscope comparing platelet-conditioned medium EV isolation using centrifugation versus the PEP process, scale bar embedded in the image. F Representative image from Single-particle interferometric reflectance imaging sensing (SP-IRIS) analysis for presence of surface CD41a, CD9, CD63, and CD81 tetraspanins. G Graphical representation of the SP-IRIS analysis. H Quantitation of CD9, CD63 and CD81 on a CD41a captured plate documented vast majority of PEP as CD41a/CD9 positive, with smaller representation from CD63 and background CD81. Data presented as mean ± stdev. N = 3 separate CGMP manufactured PEP lots. I Pie chart representation of the exosome tetraspanin surface marker profile of CD41a captured PEP exosomes. Image collected and cropped by CiteAb from the following open publication (https://pubmed.ncbi.nlm.nih.gov/36175423), licensed under a CC-BY license. Not internally tested by R&D Systems.Detection of CD63 by Western Blot
Purified exosome product (PEP) characterization, quantification, and delivery biodistribution. A NanoSight nanoparticle analysis of size distribution and concentration of PEP diluted in phosphate buffered saline 1:1000 documented 6.65 × 1012 ± 1.16 × 1011. B Transmission electron microscopy of PEP. Scale = 200 nm arrow heads pointing to EVs. C Western blot probing for CD63, CD9, and Flotillin-1 in 3 separate CGMP manufactured PEP lots. D Western blot comparison of NF-kappa B p65 and PD-L1 levels, in 3 separate CGMP manufactured PEP lots versus adipose-derived mesenchymal stem cell conditioned media (AMSC-CM). E Atomic-force microscope comparing platelet-conditioned medium EV isolation using centrifugation versus the PEP process, scale bar embedded in the image. F Representative image from Single-particle interferometric reflectance imaging sensing (SP-IRIS) analysis for presence of surface CD41a, CD9, CD63, and CD81 tetraspanins. G Graphical representation of the SP-IRIS analysis. H Quantitation of CD9, CD63 and CD81 on a CD41a captured plate documented vast majority of PEP as CD41a/CD9 positive, with smaller representation from CD63 and background CD81. Data presented as mean ± stdev. N = 3 separate CGMP manufactured PEP lots. I Pie chart representation of the exosome tetraspanin surface marker profile of CD41a captured PEP exosomes. Image collected and cropped by CiteAb from the following open publication (https://pubmed.ncbi.nlm.nih.gov/36175423), licensed under a CC-BY license. Not internally tested by R&D Systems.Detection of CD63 by Western Blot
Purified exosome product (PEP) characterization, quantification, and delivery biodistribution. A NanoSight nanoparticle analysis of size distribution and concentration of PEP diluted in phosphate buffered saline 1:1000 documented 6.65 × 1012 ± 1.16 × 1011. B Transmission electron microscopy of PEP. Scale = 200 nm arrow heads pointing to EVs. C Western blot probing for CD63, CD9, and Flotillin-1 in 3 separate CGMP manufactured PEP lots. D Western blot comparison of NF-kappa B p65 and PD-L1 levels, in 3 separate CGMP manufactured PEP lots versus adipose-derived mesenchymal stem cell conditioned media (AMSC-CM). E Atomic-force microscope comparing platelet-conditioned medium EV isolation using centrifugation versus the PEP process, scale bar embedded in the image. F Representative image from Single-particle interferometric reflectance imaging sensing (SP-IRIS) analysis for presence of surface CD41a, CD9, CD63, and CD81 tetraspanins. G Graphical representation of the SP-IRIS analysis. H Quantitation of CD9, CD63 and CD81 on a CD41a captured plate documented vast majority of PEP as CD41a/CD9 positive, with smaller representation from CD63 and background CD81. Data presented as mean ± stdev. N = 3 separate CGMP manufactured PEP lots. I Pie chart representation of the exosome tetraspanin surface marker profile of CD41a captured PEP exosomes. Image collected and cropped by CiteAb from the following open publication (https://pubmed.ncbi.nlm.nih.gov/36175423), licensed under a CC-BY license. Not internally tested by R&D Systems.Detection of CD63 by Western Blot
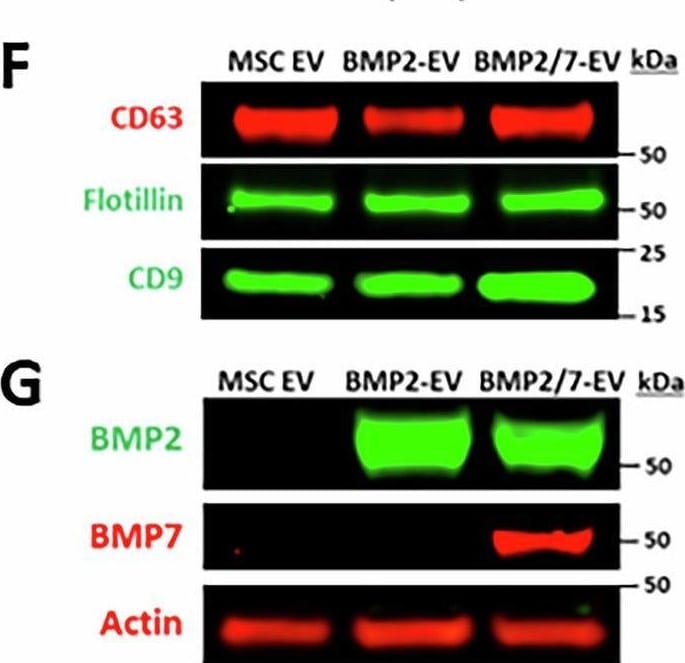
Production and characterization of engineered BMP2- and BMP7-EV.A Schematic of lentiviral vector with BMP2 (top) or BMP2 and BMP7 (bottom) joined with T2A linker. Acronyms are as follows: LTR (Long Terminal Repeat), Ψ (viral packaging), RRE (Rev response element), cPPT (central polypurine tract), WPRE (post-regulatory element), delta U3 (U3 deletion in 3’ LTR). B Real-time quantitative reverse-transcriptase PCR to show delta CT ratio to GAPDH in BMP2 or BMP7 mRNA in MSCs transfected with either LV-BMP2 or LV-BMP2/7 plasmid. Statistical significance determined by one-way ANOVA with Tukey post hoc analysis. (+/- SD, n = 3). C Western blot of MSCs that were untransfected or transfected with LV-BMP2 or LV-BMP2/7. Actin was used as a loading control. D NanoSight analysis demonstrating mean size profile (+/- SD) of extracellular vesicles isolated from LV-BMP2 or LV-BMP2/7 transfected MSCs. E Transmission electron microscopy (TEM) demonstrating size profile of BMP2-EV or BMP2/7-EV. Scale bar 200 nm. F Western blot of small extracellular vesicles collected from MSCs that were untransfected or transfected with LV-BMP2 or LV-BMP2/7. Samples were probed for small extracellular vesicle markers CD63, Flotillin, and CD9. G Western blot of small extracellular vesicles collected from MSCs that were untransfected or transfected with LV-BMP2 or LV-BMP2/7. Samples were probed for BMP2 and BMP7 with actin as a loading control. Image collected and cropped by CiteAb from the following open publication (https://pubmed.ncbi.nlm.nih.gov/40461558), licensed under a CC-BY license. Not internally tested by R&D Systems.Applications for Human CD63 Antibody
Application
Recommended Usage
CyTOF-ready
Ready to be labeled using established conjugation methods. No BSA or other carrier proteins that could interfere with conjugation.
Flow Cytometry
0.25 µg/106 cells
Sample: Human PBMCs fixed and permeabilized with FlowX FoxP3/Transcription Factor Fixation & Perm Buffer Kit (Catalog # FC012); PBMC monocytes (surface stain)
Sample: Human PBMCs fixed and permeabilized with FlowX FoxP3/Transcription Factor Fixation & Perm Buffer Kit (Catalog # FC012); PBMC monocytes (surface stain)
Immunohistochemistry
0.3-25 µg/mL
Sample: Immersion fixed paraffin-embedded sections of human lung cancer tissue
Sample: Immersion fixed paraffin-embedded sections of human lung cancer tissue
Simple Western
20 µg/mL
Sample: Human platelets
Sample: Human platelets
Western Blot
0.5 µg/mL
Sample: Human platelets
Sample: Human platelets
Reviewed Applications
Read 1 review rated 5 using MAB50482 in the following applications:
Flow Cytometry Panel Builder
Bio-Techne Knows Flow Cytometry
Save time and reduce costly mistakes by quickly finding compatible reagents using the Panel Builder Tool.
Advanced Features
- Spectra Viewer - Custom analysis of spectra from multiple fluorochromes
- Spillover Popups - Visualize the spectra of individual fluorochromes
- Antigen Density Selector - Match fluorochrome brightness with antigen density
Formulation, Preparation, and Storage
Purification
Protein A or G purified from cell culture supernatant
Reconstitution
Reconstitute at 0.5 mg/mL in sterile PBS. For liquid material, refer to CoA for concentration.
Loading...
Formulation
Lyophilized from a 0.2 μm filtered solution in PBS with Trehalose. *Small pack size (SP) is supplied either lyophilized or as a 0.2 µm filtered solution in PBS.
Shipping
Lyophilized product is shipped at ambient temperature. Liquid small pack size (-SP) is shipped with polar packs. Upon receipt, store immediately at the temperature recommended below.
Stability & Storage
Use a manual defrost freezer and avoid repeated freeze-thaw cycles.
- 12 months from date of receipt, -20 to -70 °C as supplied.
- 1 month, 2 to 8 °C under sterile conditions after reconstitution.
- 6 months, -20 to -70 °C under sterile conditions after reconstitution.
Calculators
Background: CD63
Alternate Names
CD63, Granulophysin, Lamp-3, ME491, OMA81H, Tspan30
Gene Symbol
CD63
UniProt
Additional CD63 Products
Product Documents for Human CD63 Antibody
Certificate of Analysis
To download a Certificate of Analysis, please enter a lot or batch number in the search box below.
Note: Certificate of Analysis not available for kit components.
Product Specific Notices for Human CD63 Antibody
For research use only
Related Research Areas
Citations for Human CD63 Antibody
Customer Reviews for Human CD63 Antibody (1)
5 out of 5
1 Customer Rating
Have you used Human CD63 Antibody?
Submit a review and receive an Amazon gift card!
$25/€18/£15/$25CAN/¥2500 Yen for a review with an image
$10/€7/£6/$10CAN/¥1110 Yen for a review without an image
Submit a review
Customer Images
Showing
1
-
1 of
1 review
Showing All
Filter By:
-
Application: ImmunohistochemistrySample Tested: Uterine epitheliumSpecies: HumanVerified Customer | Posted 11/25/2021
There are no reviews that match your criteria.
Protocols
Find general support by application which include: protocols, troubleshooting, illustrated assays, videos and webinars.
- 7-Amino Actinomycin D (7-AAD) Cell Viability Flow Cytometry Protocol
- Antigen Retrieval Protocol (PIER)
- Antigen Retrieval for Frozen Sections Protocol
- Appropriate Fixation of IHC/ICC Samples
- Cellular Response to Hypoxia Protocols
- Chromogenic IHC Staining of Formalin-Fixed Paraffin-Embedded (FFPE) Tissue Protocol
- Chromogenic Immunohistochemistry Staining of Frozen Tissue
- ClariTSA™ Fluorophore Kits
- Detection & Visualization of Antibody Binding
- Extracellular Membrane Flow Cytometry Protocol
- Flow Cytometry Protocol for Cell Surface Markers
- Flow Cytometry Protocol for Staining Membrane Associated Proteins
- Flow Cytometry Staining Protocols
- Flow Cytometry Troubleshooting Guide
- Fluorescent IHC Staining of Frozen Tissue Protocol
- Graphic Protocol for Heat-induced Epitope Retrieval
- Graphic Protocol for the Preparation and Fluorescent IHC Staining of Frozen Tissue Sections
- Graphic Protocol for the Preparation and Fluorescent IHC Staining of Paraffin-embedded Tissue Sections
- Graphic Protocol for the Preparation of Gelatin-coated Slides for Histological Tissue Sections
- IHC Sample Preparation (Frozen sections vs Paraffin)
- Immunofluorescent IHC Staining of Formalin-Fixed Paraffin-Embedded (FFPE) Tissue Protocol
- Immunohistochemistry (IHC) and Immunocytochemistry (ICC) Protocols
- Immunohistochemistry Frozen Troubleshooting
- Immunohistochemistry Paraffin Troubleshooting
- Intracellular Flow Cytometry Protocol Using Alcohol (Methanol)
- Intracellular Flow Cytometry Protocol Using Detergents
- Intracellular Nuclear Staining Flow Cytometry Protocol Using Detergents
- Intracellular Staining Flow Cytometry Protocol Using Alcohol Permeabilization
- Intracellular Staining Flow Cytometry Protocol Using Detergents to Permeabilize Cells
- Preparing Samples for IHC/ICC Experiments
- Preventing Non-Specific Staining (Non-Specific Binding)
- Primary Antibody Selection & Optimization
- Propidium Iodide Cell Viability Flow Cytometry Protocol
- Protocol for Heat-Induced Epitope Retrieval (HIER)
- Protocol for Liperfluo
- Protocol for Making a 4% Formaldehyde Solution in PBS
- Protocol for VisUCyte™ HRP Polymer Detection Reagent
- Protocol for the Characterization of Human Th22 Cells
- Protocol for the Characterization of Human Th9 Cells
- Protocol for the Preparation & Fixation of Cells on Coverslips
- Protocol for the Preparation and Chromogenic IHC Staining of Frozen Tissue Sections
- Protocol for the Preparation and Chromogenic IHC Staining of Frozen Tissue Sections - Graphic
- Protocol for the Preparation and Chromogenic IHC Staining of Paraffin-embedded Tissue Sections
- Protocol for the Preparation and Chromogenic IHC Staining of Paraffin-embedded Tissue Sections - Graphic
- Protocol for the Preparation and Fluorescent IHC Staining of Frozen Tissue Sections
- Protocol for the Preparation and Fluorescent IHC Staining of Paraffin-embedded Tissue Sections
- Protocol for the Preparation of Gelatin-coated Slides for Histological Tissue Sections
- Protocol: Annexin V and PI Staining by Flow Cytometry
- Protocol: Annexin V and PI Staining for Apoptosis by Flow Cytometry
- R&D Systems Quality Control Western Blot Protocol
- TUNEL and Active Caspase-3 Detection by IHC/ICC Protocol
- The Importance of IHC/ICC Controls
- Troubleshooting Guide: Fluorokine Flow Cytometry Kits
- Troubleshooting Guide: Immunohistochemistry
- Troubleshooting Guide: Western Blot Figures
- Western Blot Conditions
- Western Blot Protocol
- Western Blot Protocol for Cell Lysates
- Western Blot Troubleshooting
- Western Blot Troubleshooting Guide
- View all Protocols, Troubleshooting, Illustrated assays and Webinars
Loading...