DPP3 (DiPeptidyl Peptidase III; also known as Dipeptidyl arylamidase III and Enkephalinase B) is a cytosolic member of the metallopeptidase family of proteins (1, 2). More specifically, it is classified as the singular member of the M49/Clan M- family of enzymes that possesses an unusual six-amino acid, zinc-binding motif (HExxGH) (1, 3). Notably, DPP # 3 is the only DPP that qualifies as a metallopeptidase, as all other DPPs belong to either the cysteine or serine class of peptidases. DPP3 is widely expressed, being found in numerous hematopoietic cells (RBCs, neutrophils, monocytes) and epithelium-dominated tissues (1, 4, 5). Although DPP3 was initially reported to be an Arg-Arg dipeptidase for non-N-terminally substituted peptides, it is now known to be active on a wide range of amino acid combinations, and thus qualifies as a non-specific peptidase. DPP3 does show restriction when it comes to peptide length, however; peptides shorter than three and longer than ten amino acids are very poor substrates for DPP3. Consistent with its broad range of substrates, DPP3 likely has multiple functions. It has been suggested to be a general mediator of peptidome degradation (i.e. the three-to-24 amino acid cytoplasmic fragments that result from initial proteasome degradation), and is considered particularly important in the degradation of proline-containing peptides (1, 6). Conversely, elevated levels of DDP3 activity will reduce the availability of eight-to-ten amino acid length peptides that are used for MHC presentation, adversely affect this crucial immune surveillance activity. DDP3 is also found extracellularly, and has documented activity against angiotensin II-IV and opioids, suggesting a role for DPP3 in both blood pressure regulation and pain modulation (1, 6-8). Finally, DPP3 appears to play a protective role in oxidative stress. Nrf2 is a Zn-finger transcription factor that stimulates antioxidant enzyme production. Normally, it is sequestered in the cytosol through complex formation with Keap I. Though the details are somewhat unclear, under oxidative stress, DPP3 appears to promote the dissociation of Nrf2 and Keap I, directing Nrf2 into the nucleus with subsequent antioxidant enzyme transcription (1). Human DPP3 is 737 amino acids in length, contains a peptidase region over aa 143-705, and is reported to run as a 93-94 kDa protein on SDS-PAGE (2). Although there is no canonical signal sequence, as noted, it is found extracellularly. Potential sites for myristoylation are known and, if utilized, may account for reports of a DPP3 presence in membranes (1). There are potential isoform variants. One shows a deletion of aa 182-601, while another shows a deletion of aa 91-120. Mouse and rat DPP3 share 93% aa sequence identity with human DPP3.


Key Product Details
Species Reactivity
Human
Applications
Western Blot, Immunocytochemistry
Label
Unconjugated
Antibody Source
Monoclonal Mouse IgG1 Clone # 987030
Loading...
Product Specifications
Immunogen
E. coli-derived recombinant human DPP3
Ala2-Ala737
Accession # Q9NY33
Ala2-Ala737
Accession # Q9NY33
Specificity
Detects human DPP3 in direct ELISAs.
Clonality
Monoclonal
Host
Mouse
Isotype
IgG1
Scientific Data Images for Human DPP3 Antibody
Detection of Human DPP3 by Western Blot.
Western blot shows lysates of human placenta tissue, humand kidney tissue, HepG2 human hepatocellular carcinoma cell line, HeLa human cervical epithelial carcinoma cell line, and Raji human Burkitt's lymphoma cell line. PVDF membrane was probed with 1 µg/mL of Mouse Anti-Human DPP3 Antigen Affinity-purified Monoclonal Antibody (Catalog # MAB80871) followed by HRP-conjugated Anti-Rat IgG Secondary Antibody (Catalog # HAF005). A specific band was detected for DPP3 at approximately 75 kDa (as indicated). This experiment was conducted under reducing conditions and using Immunoblot Buffer Group 1.DPP3 in HeLa Human Cell Line.
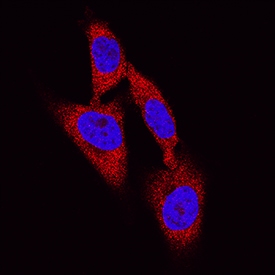
DPP3 was detected in immersion fixed HeLa human cervical epithelial carcinoma cell line using Mouse Anti-Human DPP3 Antigen Affinity-purified Monoclonal Antibody (Catalog # MAB80871) at 8 µg/mL for 3 hours at room temperature. Cells were stained using the NorthernLights™ 557-conjugated Anti-Mouse IgG Secondary Antibody (red; Catalog # NL007) and counterstained with DAPI (blue). Specific staining was localized to cytoplasm. View our protocol for Fluorescent ICC Staining of Cells on Coverslips.Applications for Human DPP3 Antibody
Application
Recommended Usage
Immunocytochemistry
8-25 µg/mL
Sample: Immersion fixed HeLa human cervical epithelial carcinoma cell line
Sample: Immersion fixed HeLa human cervical epithelial carcinoma cell line
Western Blot
1 µg/mL
Sample: Human placenta tissue, Human kidney tissue, HepG2 human hepatocellular carcinoma cell line, HeLa human cervical epithelial carcinoma cell line, and Raji human Burkitt's lymphoma cell line
Sample: Human placenta tissue, Human kidney tissue, HepG2 human hepatocellular carcinoma cell line, HeLa human cervical epithelial carcinoma cell line, and Raji human Burkitt's lymphoma cell line
Formulation, Preparation, and Storage
Purification
Protein A or G purified from hybridoma culture supernatant
Reconstitution
Reconstitute at 0.5 mg/mL in sterile PBS. For liquid material, refer to CoA for concentration.
Loading...
Formulation
Lyophilized from a 0.2 μm filtered solution in PBS with Trehalose. *Small pack size (SP) is supplied either lyophilized or as a 0.2 µm filtered solution in PBS.
Shipping
Lyophilized product is shipped at ambient temperature. Liquid small pack size (-SP) is shipped with polar packs. Upon receipt, store immediately at the temperature recommended below.
Stability & Storage
Use a manual defrost freezer and avoid repeated freeze-thaw cycles.
- 12 months from date of receipt, -20 to -70 °C as supplied.
- 1 month, 2 to 8 °C under sterile conditions after reconstitution.
- 6 months, -20 to -70 °C under sterile conditions after reconstitution.
Calculators
Background: DPP3
References
- Prajapati, S.C. & S. S. Chauhan (2011) FEBS J. 278:3256.
- Fukasawa, K. et al. (1998) Biochem. J. 329:275.
- Hirose, J. et al. (2004) Arch. Biochem. Biophys. 431:1.
- Jones, T.H & A. Kapralou (1982) Anal. Biochem. 119:418.
- Hashimoto, J. et al. (2000) Biochem. Biophys. Res. Commun. 273:393.
- Barsun, M. et al. (2007) Biol. Chem. 388:343.
- Allard, M. et al. (1987) J. Neurochem. 48:1553.
- Bezerra, G.A. et al. (2012) Proc. Natl. Acad. Sci. USA 109:6525.
Long Name
Dipeptidyl-peptidase 3
Alternate Names
DPPIII
Gene Symbol
DPP3
UniProt
Additional DPP3 Products
Product Documents for Human DPP3 Antibody
Certificate of Analysis
To download a Certificate of Analysis, please enter a lot or batch number in the search box below.
Note: Certificate of Analysis not available for kit components.
Product Specific Notices for Human DPP3 Antibody
For research use only
Related Research Areas
Customer Reviews for Human DPP3 Antibody
There are currently no reviews for this product. Be the first to review Human DPP3 Antibody and earn rewards!
Have you used Human DPP3 Antibody?
Submit a review and receive an Amazon gift card!
$25/€18/£15/$25CAN/¥2500 Yen for a review with an image
$10/€7/£6/$10CAN/¥1110 Yen for a review without an image
Submit a review
Protocols
Find general support by application which include: protocols, troubleshooting, illustrated assays, videos and webinars.
- Appropriate Fixation of IHC/ICC Samples
- Cellular Response to Hypoxia Protocols
- ClariTSA™ Fluorophore Kits
- Detection & Visualization of Antibody Binding
- ICC Cell Smear Protocol for Suspension Cells
- ICC Immunocytochemistry Protocol Videos
- ICC for Adherent Cells
- Immunocytochemistry (ICC) Protocol
- Immunocytochemistry Troubleshooting
- Immunofluorescence of Organoids Embedded in Cultrex Basement Membrane Extract
- Immunohistochemistry (IHC) and Immunocytochemistry (ICC) Protocols
- Preparing Samples for IHC/ICC Experiments
- Preventing Non-Specific Staining (Non-Specific Binding)
- Primary Antibody Selection & Optimization
- Protocol for VisUCyte™ HRP Polymer Detection Reagent
- Protocol for the Fluorescent ICC Staining of Cell Smears - Graphic
- Protocol for the Fluorescent ICC Staining of Cultured Cells on Coverslips - Graphic
- Protocol for the Preparation and Fluorescent ICC Staining of Cells on Coverslips
- Protocol for the Preparation and Fluorescent ICC Staining of Non-adherent Cells
- Protocol for the Preparation and Fluorescent ICC Staining of Stem Cells on Coverslips
- Protocol for the Preparation of a Cell Smear for Non-adherent Cell ICC - Graphic
- R&D Systems Quality Control Western Blot Protocol
- TUNEL and Active Caspase-3 Detection by IHC/ICC Protocol
- The Importance of IHC/ICC Controls
- Troubleshooting Guide: Western Blot Figures
- Western Blot Conditions
- Western Blot Protocol
- Western Blot Protocol for Cell Lysates
- Western Blot Troubleshooting
- Western Blot Troubleshooting Guide
- View all Protocols, Troubleshooting, Illustrated assays and Webinars
Loading...