Follistatin-related gene protein (FLRG), also known as follistatin-like 3 (FSTL3) is a glycoprotein belonging to the follistatin-module protein family. Human FLRG cDNA encodes a 263 amino acid (aa) residue protein with a putative 26 aa signal peptide, an N-terminal domain, two cysteine-rich follistatin-like domains (FS) and a C‑terminal acidic domain. Compared to follistatin, FLRG lacks the third FS domain found in follistatin. In addition, FLRG also lacks the heparin-binding domain found within the first amino-terminal FS domain of follistatin. Mouse and human FLRG share approximately 83% aa sequence homology. Like follistatin, FLRG has been shown to bind and inhibit the activities of TGF-beta family ligands including activin, BMP-2, -6, -7 and GDF-8/myostatin. While both FLRG and follistatin are located in a wide and overlapping range of adult and fetal tissue, their sites of peak expression differ: FLRG most highly in heart, lung, kidney, placenta and testis, while follistatin is highest in ovary and pituitary. The expression of FLRG is upregulated by TGF-beta and activin signaling through Smad proteins. Although FLRG is a secreted protein in many cell types, it has also been localized to the nuclear compartment in HeLa, 293 and CHO cells (1‑5).
Human Follistatin‑related Gene Protein/FLRG Antibody
R&D Systems | Catalog # AF1288

Key Product Details
Species Reactivity
Validated:
Cited:
Applications
Validated:
Cited:
Label
Antibody Source
Product Specifications
Immunogen
Met27-Val263
Accession # O95633
Specificity
Clonality
Host
Isotype
Endotoxin Level
Scientific Data Images for Human Follistatin‑related Gene Protein/FLRG Antibody
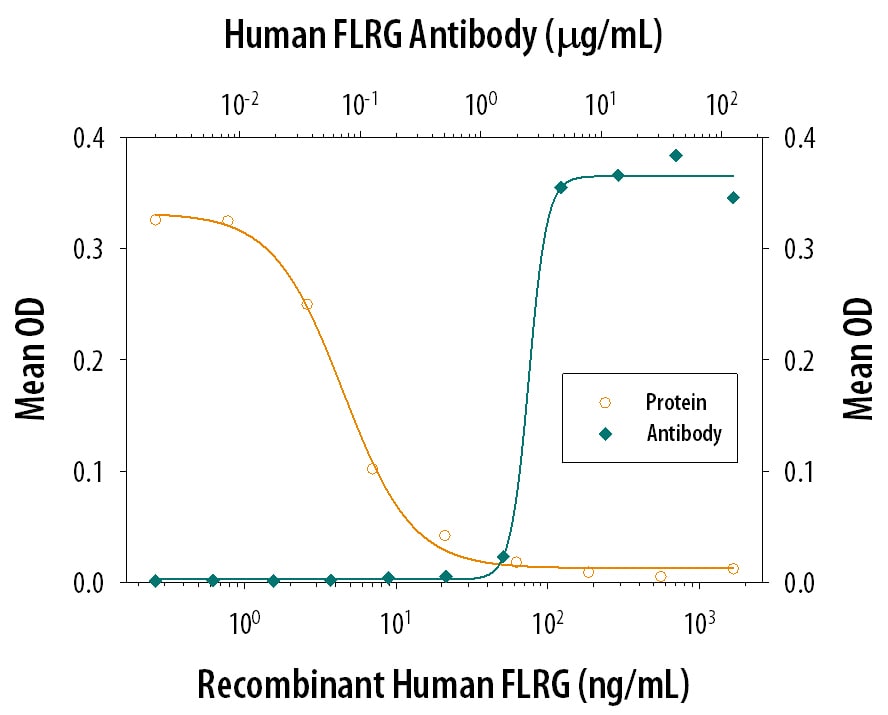
Follistatin-related Gene Protein/FLRG Inhibition of Activin A-induced Hemo-globin Expression and Neutralization by Human Follistatin-related Gene Protein/FLRG Antibody.
Recombinant Human Follistatin-related Gene Protein/FLRG (Catalog # 1288-F3) inhibits Recombinant Human/Mouse/Rat Activin A (Catalog # 338-AC) induced hemoglobin expression in the K562 human chronic myelogenous leukemia cell line in a dose-dependent manner (orange line), as measured by the psuedoperoxidase activity. Inhibition of Recombinant Human/Mouse/Rat Activin A (7.5 ng/mL) activity elicited by Recombinant Human Follistatin-related Gene Protein/ FLRG (0.1 µg/mL) is neutralized (green line) by increasing concentrations of Goat Anti-Human Follistatin-related Gene Protein/FLRG Antigen Affinity-purified Polyclonal Antibody (Catalog # AF1288). The ND50 is typically 1-4 µg/mL.Applications for Human Follistatin‑related Gene Protein/FLRG Antibody
ELISA
ELISA detection - This antibody can be used as a detection reagent in a human FLRG sandwich immunoassay in combination with the human FLRG capture reagent (Cat. # MAB1288) and recombinant human FLRG (Cat. # 1288-F3) as the standard. The suggested concentration range for this detection reagent is 0.1 - 0.4 μg/mL and should be titrated to determine the optimal concentration. In this format, less than 1% cross-reactivity is observed with rmFLRG.
Neutralization
Formulation, Preparation, and Storage
Purification
Reconstitution
Reconstitute at 0.2 mg/mL in sterile PBS. For liquid material, refer to CoA for concentration.
Formulation
Shipping
Stability & Storage
- 12 months from date of receipt, -20 to -70 °C as supplied.
- 1 month, 2 to 8 °C under sterile conditions after reconstitution.
- 6 months, -20 to -70 °C under sterile conditions after reconstitution.
Calculators
Background: Follistatin-related Gene Protein/FLRG
References
- Tsuchida, K. et al. (2000) J. Biol. Chem. 275:40778.
- Sidis, Y. et al. (2002) Endocrinology 143:1613.
- Tortoriello, D.V. et al. (2001) Endocrinology 142:3426.
- Hill, J. et al. (2002) J. Biol. Chem. 277:40735.
- Bartholin, L. et al. (2001) Oncogene 20:5409.
Alternate Names
Gene Symbol
UniProt
Additional Follistatin-related Gene Protein/FLRG Products
Product Documents for Human Follistatin‑related Gene Protein/FLRG Antibody
Certificate of Analysis
To download a Certificate of Analysis, please enter a lot or batch number in the search box below.
Note: Certificate of Analysis not available for kit components.
Product Specific Notices for Human Follistatin‑related Gene Protein/FLRG Antibody
For research use only
Related Research Areas
Citations for Human Follistatin‑related Gene Protein/FLRG Antibody
Customer Reviews for Human Follistatin‑related Gene Protein/FLRG Antibody
There are currently no reviews for this product. Be the first to review Human Follistatin‑related Gene Protein/FLRG Antibody and earn rewards!
Have you used Human Follistatin‑related Gene Protein/FLRG Antibody?
Submit a review and receive an Amazon gift card!
$25/€18/£15/$25CAN/¥2500 Yen for a review with an image
$10/€7/£6/$10CAN/¥1110 Yen for a review without an image
Submit a review
Protocols
Find general support by application which include: protocols, troubleshooting, illustrated assays, videos and webinars.
- ELISA Sample Preparation & Collection Guide
- ELISA Troubleshooting Guide
- How to Run an R&D Systems DuoSet ELISA
- How to Run an R&D Systems Quantikine ELISA
- How to Run an R&D Systems Quantikine™ QuicKit™ ELISA
- Quantikine HS ELISA Kit Assay Principle, Alkaline Phosphatase
- Quantikine HS ELISA Kit Principle, Streptavidin-HRP Polymer
- Sandwich ELISA (Colorimetric) – Biotin/Streptavidin Detection Protocol
- Sandwich ELISA (Colorimetric) – Direct Detection Protocol
- Troubleshooting Guide: ELISA
- View all Protocols, Troubleshooting, Illustrated assays and Webinars
