Granulysin (formerly NKG5 or Lymphokine LAG-2) is a member of the saposin-like protein (SAPLIP) family of membrane disrupting proteins (1). Granulysin is expressed in granules of natural killer and activated cytotoxic T cells. It exhibits cytolytic activity against intracellular or extracellular microbes and also tumors, either alone or in synergy with perforin (2). Human granulysin has structural similarity and 30 - 40% aa identity to granulysins and NK-lysins of other mammals such as bovine, porcine and canine; similar peptides in rodents have not been identified (1). The 15 kDa unglycosylated protein contains five helical domains; helix 2 and 3 contain 9 arginines and one cysteine critical for activity. Peptides of either helix 2 or 3 will lyse bacteria, while helix 3 is needed to lyse tumor targets (3, 4). One isoform designated 519 uses a different start codon, has no signal peptide sequence and is poorly expressed (5). A post-translationally processed 9 kDa form is present in acidified granules and is less lytic than the 15 kDa form at granule pH (6). IL-15 is necessary and sufficient for granulysin upregulation in CD8 T cells (2). Nanomolar granulysin promotes chemotaxis and increases production of chemokines by monocytic cells, while micromolar local concentrations are needed for lysis (7). Experimental evidence supports the following mechanism for activity against intracellular pathogens (8). First, granulysin forms clusters in lipid rafts due to interaction of positive charges in helices 2-3 with acidic sphingolipids. After endocytosis, early endosomes fuse with phagosomes, probably regulated by small GTPase rab5. Granulysin binds microbial membranes through charge interactions and disrupts them, probably via scissoring actions of granulysin molecules (9, 10).
Human Granulysin Antibody
R&D Systems | Catalog # AF3138

Key Product Details
Validated by
Species Reactivity
Validated:
Cited:
Applications
Validated:
Cited:
Label
Antibody Source
Product Specifications
Immunogen
Arg23-Leu145
Accession # P22749
Specificity
Clonality
Host
Isotype
Scientific Data Images for Human Granulysin Antibody
Detection of Granulysin in Human PBMCs by Flow Cytometry.
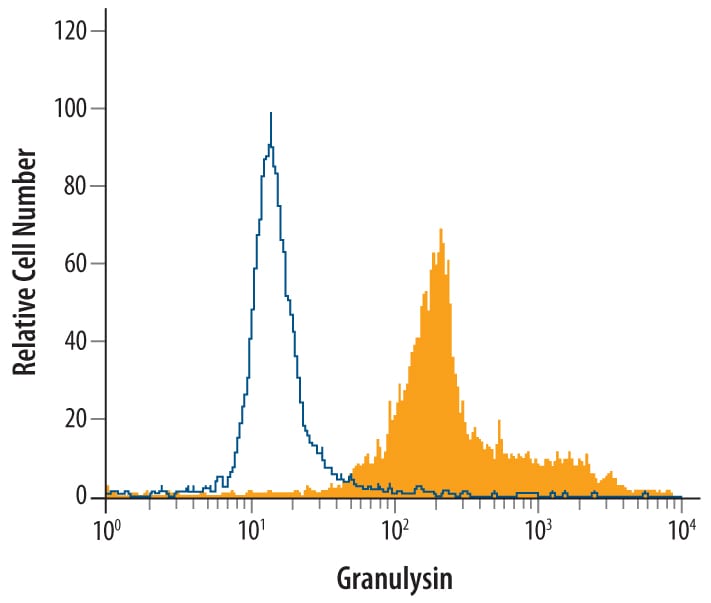
Human peripheral blood mononuclear cells were treated for 24 hr with 50 ng/ml PMA and 200 ng/ml Ca2+ionomycin then stained with Goat Anti-Human Granulysin Antigen Affinity-purified Polyclonal Antibody (Catalog # AF3138, filled histogram) or control antibody (AB-108-C, open histogram), followed by Phycoerythrin-conjugated Anti-Goat IgG Secondary Antibody (F0107).Detection of Granulysin in human PBMCs by Flow Cytometry
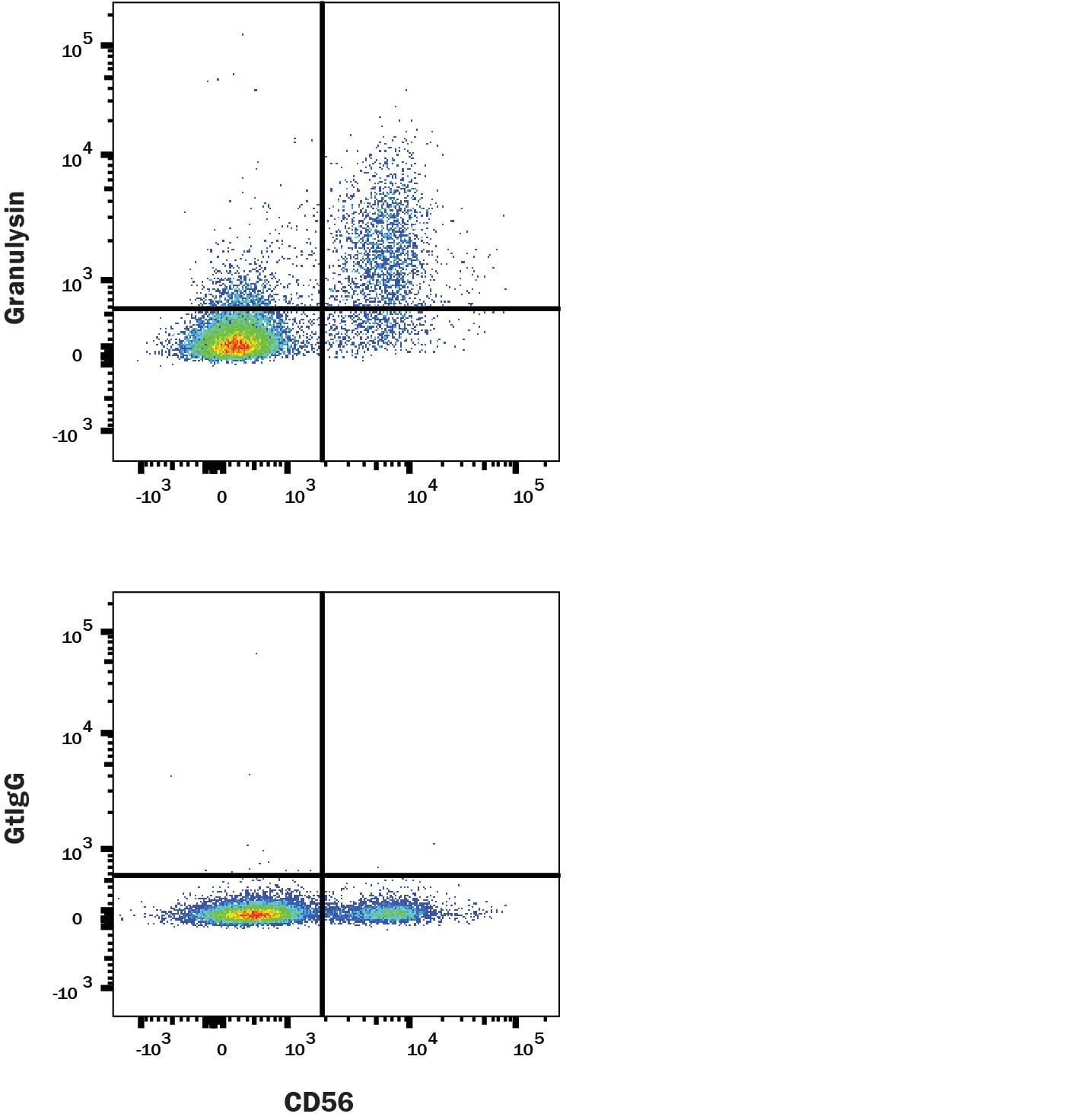
Human PBMCs were stained with Rabbit Anti-Human NCAM‑1/CD56 PE‑conjugated Monoclonal Antibody (Catalog # FAB24086P) and either (A) Goat Anti-Human Granulysin Antigen Affinity-purified Polyclonal Antibody (Catalog # AF3138) or (B) isotype control antibody (Catalog # AB-108-C) followed by Allophycocyanin-conjugated Anti-Goat IgG Secondary Antibody (Catalog # F0108). To facilitate intracellular staining, cells were fixed with Flow Cytometry Fixation Buffer (Catalog # FC004) and permeabilized with Flow Cytometry Permeabilization/Wash Buffer I (Catalog # FC005). View our protocol for Staining Intracellular Molecules.Applications for Human Granulysin Antibody
CyTOF-ready
Intracellular Staining by Flow Cytometry
Sample: Human PBMCs
Western Blot
Sample: Recombinant Human Granulysin (Catalog # 3138-GN/CF)
Flow Cytometry Panel Builder
Bio-Techne Knows Flow Cytometry
Save time and reduce costly mistakes by quickly finding compatible reagents using the Panel Builder Tool.
Advanced Features
- Spectra Viewer - Custom analysis of spectra from multiple fluorochromes
- Spillover Popups - Visualize the spectra of individual fluorochromes
- Antigen Density Selector - Match fluorochrome brightness with antigen density
Formulation, Preparation, and Storage
Purification
Reconstitution
Reconstitute at 0.2 mg/mL in sterile PBS. For liquid material, refer to CoA for concentration.
Formulation
*Small pack size (-SP) is supplied either lyophilized or as a 0.2 µm filtered solution in PBS.
Shipping
Stability & Storage
- 12 months from date of receipt, -20 to -70 °C as supplied.
- 1 month, 2 to 8 °C under sterile conditions after reconstitution.
- 6 months, -20 to -70 °C under sterile conditions after reconstitution.
Calculators
Background: Granulysin
References
- Clayberger, C. and A.M. Krensky (2003) Curr. Opin. Immunol. 15:560.
- Ma, L.L. et al. (2002) J. Immunol. 169:5787.
- Wang, Z. et al. (2000) J. Immunol. 165:1486.
- Linde, C.M.A. et al. (2005) Infect. Immun. 73:6332.
- Yabe, T. et al. (1990) J. Exp. Med. 172:1159.
- Hanson, D.A. et al. (1999) Mol. Immunol. 36:413.
- Deng, A. et al. (2005) J. Immunol. 174:5243.
- Walch, M. et al. (2005) J. Immunol. 174:4220.
- Ernst, W.A. et al. (2000) J. Immunol. 165:7102.
- Anderson, D.H. et al. (2003) J. Mol. Biol. 325:355.
Alternate Names
Entrez Gene IDs
Gene Symbol
UniProt
Additional Granulysin Products
Product Documents for Human Granulysin Antibody
Certificate of Analysis
To download a Certificate of Analysis, please enter a lot or batch number in the search box below.
Note: Certificate of Analysis not available for kit components.
Product Specific Notices for Human Granulysin Antibody
For research use only
Citations for Human Granulysin Antibody
Customer Reviews for Human Granulysin Antibody
There are currently no reviews for this product. Be the first to review Human Granulysin Antibody and earn rewards!
Have you used Human Granulysin Antibody?
Submit a review and receive an Amazon gift card!
$25/€18/£15/$25CAN/¥2500 Yen for a review with an image
$10/€7/£6/$10CAN/¥1110 Yen for a review without an image
Submit a review
Protocols
Find general support by application which include: protocols, troubleshooting, illustrated assays, videos and webinars.
- 7-Amino Actinomycin D (7-AAD) Cell Viability Flow Cytometry Protocol
- Cellular Response to Hypoxia Protocols
- Extracellular Membrane Flow Cytometry Protocol
- Flow Cytometry Protocol for Cell Surface Markers
- Flow Cytometry Protocol for Staining Membrane Associated Proteins
- Flow Cytometry Staining Protocols
- Flow Cytometry Troubleshooting Guide
- Intracellular Flow Cytometry Protocol Using Alcohol (Methanol)
- Intracellular Flow Cytometry Protocol Using Detergents
- Intracellular Nuclear Staining Flow Cytometry Protocol Using Detergents
- Intracellular Staining Flow Cytometry Protocol Using Alcohol Permeabilization
- Intracellular Staining Flow Cytometry Protocol Using Detergents to Permeabilize Cells
- Propidium Iodide Cell Viability Flow Cytometry Protocol
- Protocol for Liperfluo
- Protocol for the Characterization of Human Th22 Cells
- Protocol for the Characterization of Human Th9 Cells
- Protocol: Annexin V and PI Staining by Flow Cytometry
- Protocol: Annexin V and PI Staining for Apoptosis by Flow Cytometry
- R&D Systems Quality Control Western Blot Protocol
- Troubleshooting Guide: Fluorokine Flow Cytometry Kits
- Troubleshooting Guide: Western Blot Figures
- Western Blot Conditions
- Western Blot Protocol
- Western Blot Protocol for Cell Lysates
- Western Blot Troubleshooting
- Western Blot Troubleshooting Guide
- View all Protocols, Troubleshooting, Illustrated assays and Webinars