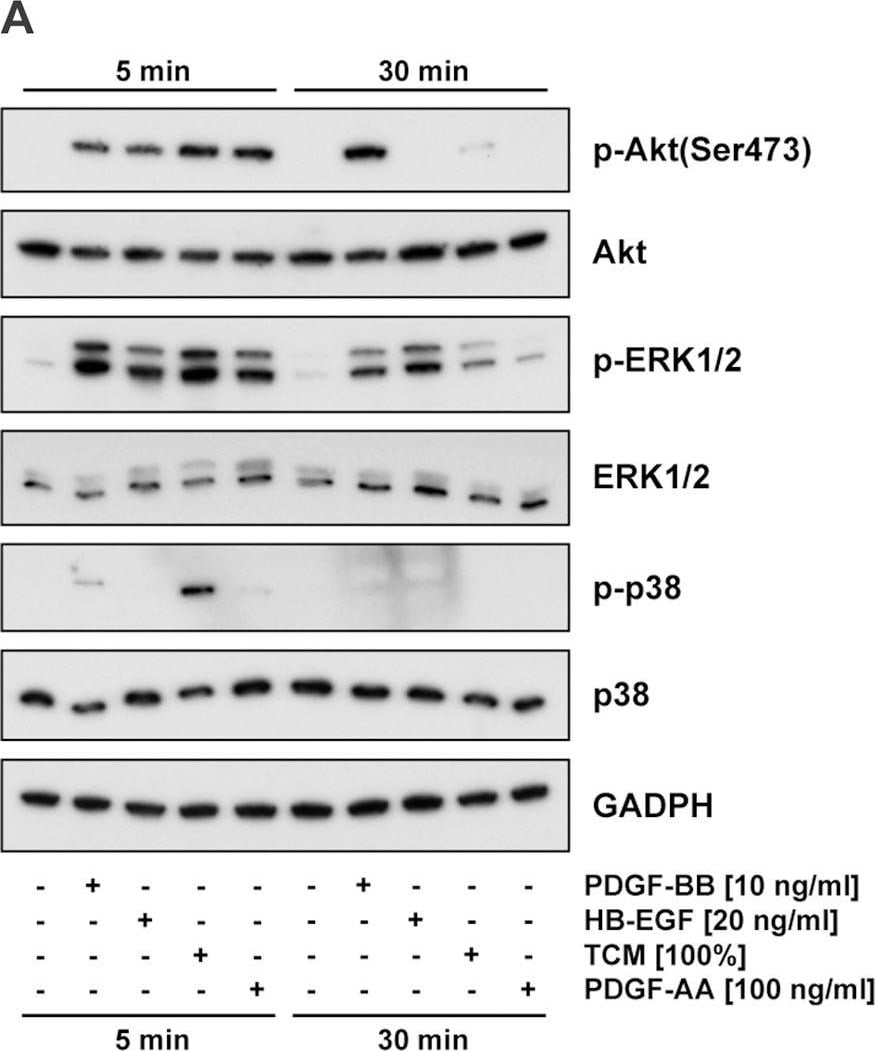
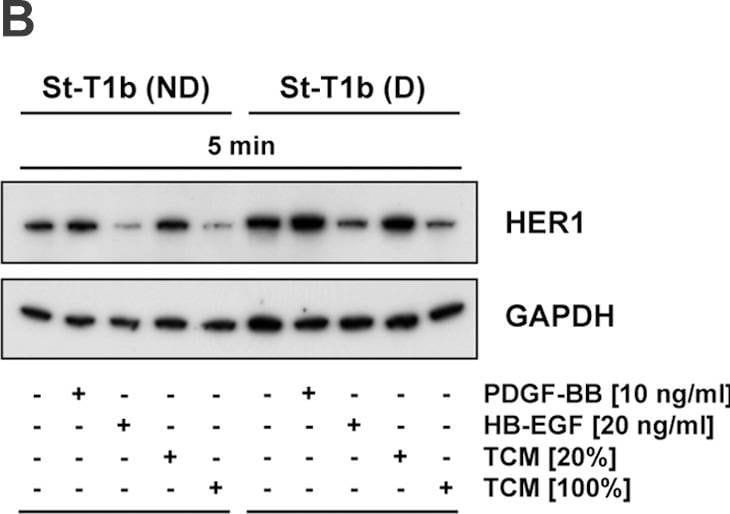
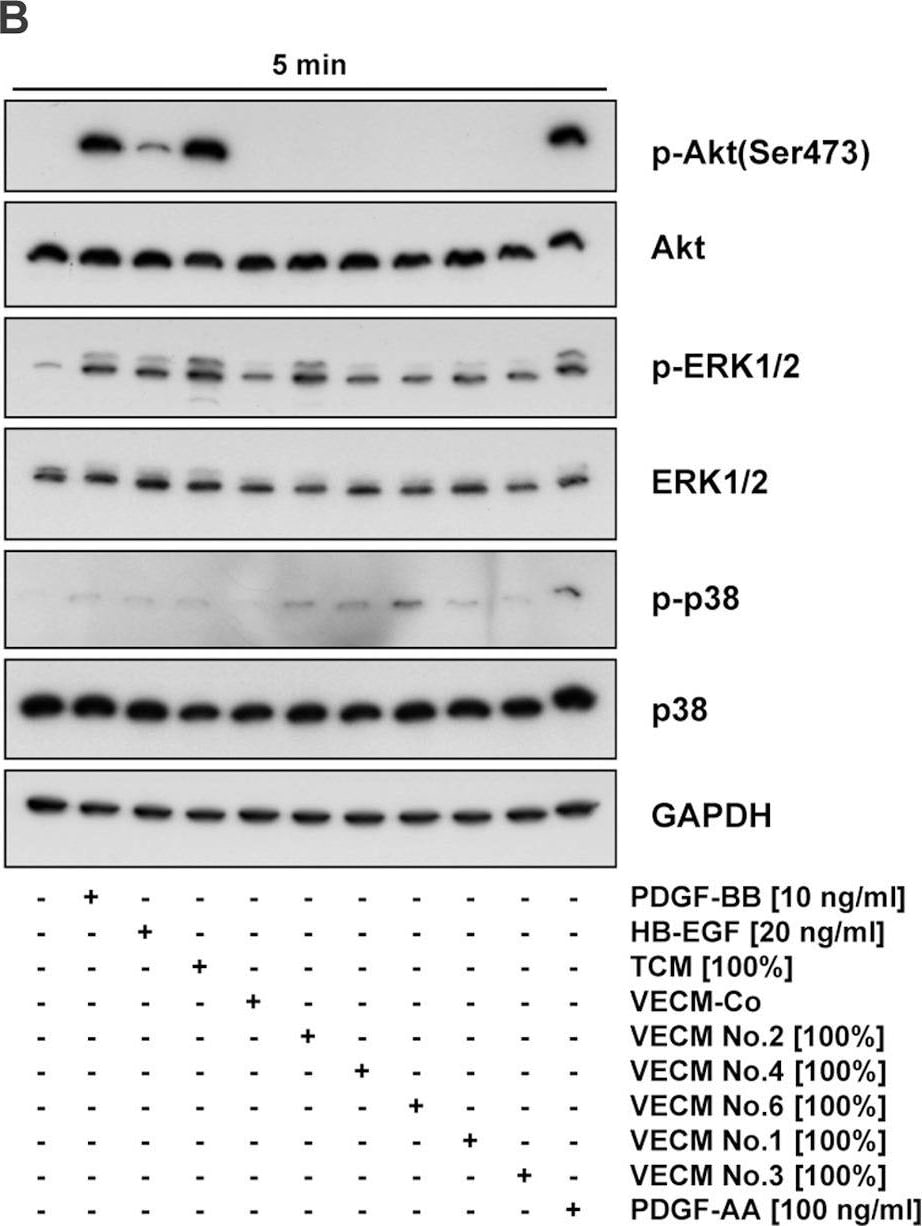
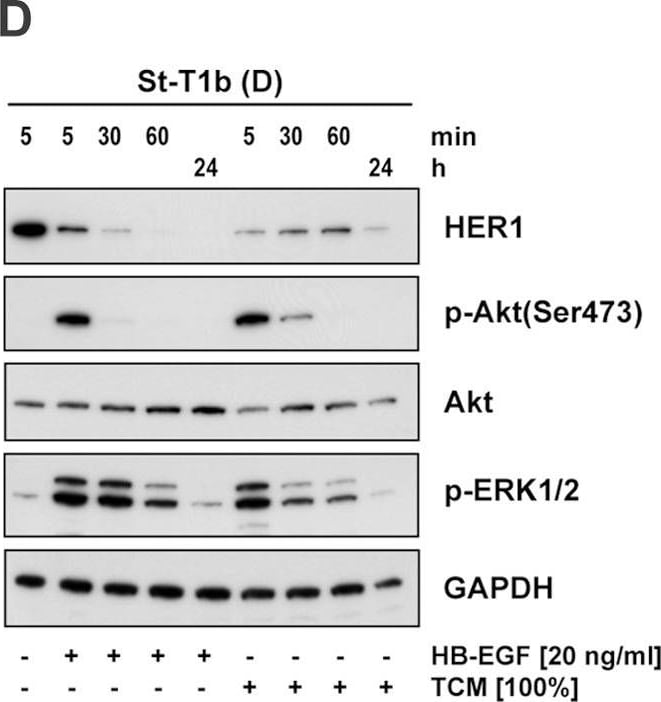
HB-EGF was originally purified based on its heparin-binding property and mitogenic activity on BALB-3T3 fibroblasts from the conditioned medium of the human U-937 histiocytic lymphoma cell line. The natural protein has an apparent molecular mass of 19-23 kDa and exists in multiple forms as a result of heterogenous O‑glycosylation and/or N-terminal truncation. In addition to fibroblasts, HB-EGF is also a potent mitogen for keratinocytes and smooth muscle cells but not for capillary endothelial cells. HB-EGF is produced in monocytes and macrophages. In addition, transcription of HB-EGF can be induced in vascular endothelial cells as well as aortic smooth muscle cells (SMC), suggesting that HB-EGF may have an important role in the pathogenesis of atherosclerosis.
HB-EGF is a member of the EGF family of mitogens which also include transforming growth factor-alpha (TGF-alpha ), amphiregulin (AR), rat schwanoma-derived growth factor (SDGF), vaccinia growth factor (VGF), and the various ligands for the Her2/ErbB2/Neu receptor. All these cytokines are derived from transmembrane precursors that contain one or several EGF structural units in their extracellular domain. Many of these transmembrane precursors are biologically active and seem to play a role in juxtacrine stimulation of adjacent cells. The cDNA for HB-EGF encodes a 204 amino acid residue transmembrane protein that is proteolytically cleaved to generate the soluble HB-EGF. Like EGF, TGF-alpha, and AR; HB-EGF binds to the EGF receptor and activates the receptor tyrosine kinase. HB-EGF is reported to be a more potent SMC mitogen than EGF. It has been suggested that the differential activities found for HB-EGF compared to EGF may be mediated by the heparin-binding properties of HB-EGF. A diphtheria toxin receptor that mediates the endocytosis of the bound toxin has been cloned and found to be identical to the transmembrane HB-EGF precursor.