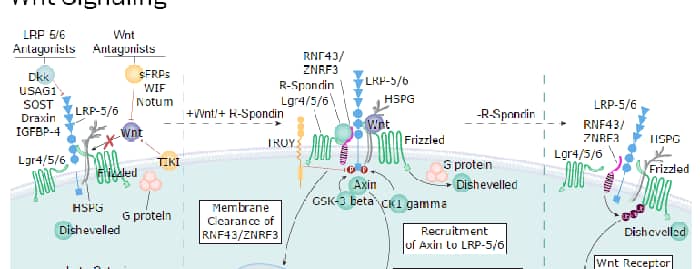
The low-density lipoprotein (LDL) receptor-related protein 5 (LRP-5) and LRP-6 constitute a distinct subgroup of the LDL receptor family. Both LRP-5 and LRP-6 are type I transmembrane proteins that function as co-receptors with Frizzled (FZD) in the canonical Wnt signaling pathway (1, 2). LRP-6 cDNA encodes a 1613 amino acid residue (aa) precursor with a 19 aa signal sequence, a 1353 aa extracellular region, a 23 aa transmembrane (TM) segment and a 218 aa cytoplasmic domain (3). The extracellular region contains four N-terminal cysteine-rich EGF-like repeats, followed by three cysteine-rich LDLR repeats. This pattern of the EGF and LDLR repeat arrangement is different than that typically found in other LDL receptor family proteins. The intracellular region of LRP6 contains protein-protein interaction motifs and is required for canonical Wnt signal transduction (4). Human LRP-6 shares 98% and 74% aa sequence identity with mouse LRP-6 and human LRP-5, respectively. Based on the current model, canonical Wnt signaling requires the interaction of Wnt with FZD and LRP to form a trimeric complex which signals downstream to stabilize cytoplasmic beta -catenin. The stabilized beta -catenin is then translocated to the nucleus where it complexes with the transcription factor LEF/TCF to regulate the transcription of target genes (5). LRP-6 has also been shown to interact with the Dickkopf proteins (DKKs), which are modulators of Wnt signaling (6-8). Binding of DKK-1 to LRP-6 dissociates LRP-6 from FZD, and antagonizes the formation of the functional receptor complex. On cells where the transmembrane proteins Kremens are also present, a ternary complex of LRP-6, DKK-1 and Kremen is formed to trigger the internalization of the complex and removal LRP6 from the cell surface. Thus, DKK-1 and Kremen function synergistically to antagonize LRP-6-mediated Wnt activity. Although DKK-2 also functions as a Wnt antagonist on cells that express Kremen, DKK-2 binding to LRP-6 in the absence of Kremen activates rather than inhibits LRP mediated beta -catenin signaling (9, 10).
Human LRP‑6 Antibody
R&D Systems | Catalog # AF1505

Key Product Details
Species Reactivity
Validated:
Cited:
Applications
Validated:
Cited:
Label
Antibody Source
Product Specifications
Immunogen
Ala20-Pro1368
Accession # O75581
Specificity
Clonality
Host
Isotype
Scientific Data Images for Human LRP‑6 Antibody
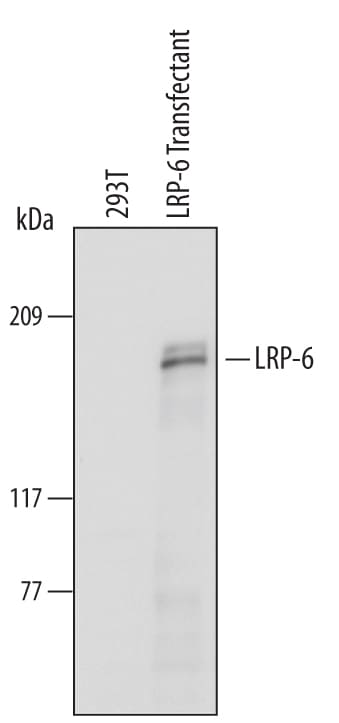
Detection of Human LRP‑6 by Western Blot.
Western blot shows lysates of 293T human embryonic kidney cell line either mock transfected or transfected with human LRP-6. PVDF Membrane was probed with 1 µg/mL of Goat Anti-Human LRP-6 Antigen Affinity-purified Polyclonal Antibody (Catalog # AF1505) followed by HRP-conjugated Anti-Goat IgG Secondary Antibody (Catalog # HAF109). A specific band was detected for LRP-6 at approximately 180 kDa (as indicated). This experiment was conducted under reducing conditions and using Immunoblot Buffer Group 1.Applications for Human LRP‑6 Antibody
CyTOF-ready
Flow Cytometry
Sample: HEK293 human embryonic kidney cell line
Western Blot
Sample: Recombinant Human LRP‑6 Fc Chimera (Catalog # 1505-LR)
Flow Cytometry Panel Builder
Bio-Techne Knows Flow Cytometry
Save time and reduce costly mistakes by quickly finding compatible reagents using the Panel Builder Tool.
Advanced Features
- Spectra Viewer - Custom analysis of spectra from multiple fluorochromes
- Spillover Popups - Visualize the spectra of individual fluorochromes
- Antigen Density Selector - Match fluorochrome brightness with antigen density
Formulation, Preparation, and Storage
Purification
Reconstitution
Reconstitute at 0.2 mg/mL in sterile PBS. For liquid material, refer to CoA for concentration.
Formulation
Shipping
Stability & Storage
- 12 months from date of receipt, -20 to -70 °C as supplied.
- 1 month, 2 to 8 °C under sterile conditions after reconstitution.
- 6 months, -20 to -70 °C under sterile conditions after reconstitution.
Calculators
Background: LRP-6
References
- Howell, B.W. and J. Herz (2001) Curr. Op. Neurobiology 11:74.
- Pinson, K.I. et al. (2000) Nature 407:535.
- Brown, S.D. et al. (1998) Biochem. Biophys. Res. Commun. 248:879.
- Tamai, K. et al. (2000) Nature 407:530.
- Schweizer, L. and H. Varmus (2003) BMC Cell Biology 4:4.
- Mao, B. et al. (2001) Nature 411:321.
- Semenov, M.V. et al. (2001) Curr. Biol. 11:951.
- Bafico, A. et al. (2001) Nature Cell Biol. 3:683.
- Zorn, A.M. (2001) Curr. Biol. 11:R592.
- Mao, B. et al. (2002) Nature 417:664.
Long Name
Alternate Names
Gene Symbol
UniProt
Additional LRP-6 Products
Product Documents for Human LRP‑6 Antibody
Certificate of Analysis
To download a Certificate of Analysis, please enter a lot or batch number in the search box below.
Note: Certificate of Analysis not available for kit components.
Product Specific Notices for Human LRP‑6 Antibody
For research use only
Related Research Areas
Citations for Human LRP‑6 Antibody
Customer Reviews for Human LRP‑6 Antibody
There are currently no reviews for this product. Be the first to review Human LRP‑6 Antibody and earn rewards!
Have you used Human LRP‑6 Antibody?
Submit a review and receive an Amazon gift card!
$25/€18/£15/$25CAN/¥2500 Yen for a review with an image
$10/€7/£6/$10CAN/¥1110 Yen for a review without an image
Submit a review
Protocols
Find general support by application which include: protocols, troubleshooting, illustrated assays, videos and webinars.
- 7-Amino Actinomycin D (7-AAD) Cell Viability Flow Cytometry Protocol
- Cellular Response to Hypoxia Protocols
- Extracellular Membrane Flow Cytometry Protocol
- Flow Cytometry Protocol for Cell Surface Markers
- Flow Cytometry Protocol for Staining Membrane Associated Proteins
- Flow Cytometry Staining Protocols
- Flow Cytometry Troubleshooting Guide
- Intracellular Flow Cytometry Protocol Using Alcohol (Methanol)
- Intracellular Flow Cytometry Protocol Using Detergents
- Intracellular Nuclear Staining Flow Cytometry Protocol Using Detergents
- Intracellular Staining Flow Cytometry Protocol Using Alcohol Permeabilization
- Intracellular Staining Flow Cytometry Protocol Using Detergents to Permeabilize Cells
- Propidium Iodide Cell Viability Flow Cytometry Protocol
- Protocol for Liperfluo
- Protocol for the Characterization of Human Th22 Cells
- Protocol for the Characterization of Human Th9 Cells
- Protocol: Annexin V and PI Staining by Flow Cytometry
- Protocol: Annexin V and PI Staining for Apoptosis by Flow Cytometry
- R&D Systems Quality Control Western Blot Protocol
- Troubleshooting Guide: Fluorokine Flow Cytometry Kits
- Troubleshooting Guide: Western Blot Figures
- Western Blot Conditions
- Western Blot Protocol
- Western Blot Protocol for Cell Lysates
- Western Blot Troubleshooting
- Western Blot Troubleshooting Guide
- View all Protocols, Troubleshooting, Illustrated assays and Webinars
Associated Pathways
 Wnt Signaling Pathways: beta-Catenin-dependent Wnt Signaling
Wnt Signaling Pathways: beta-Catenin-dependent Wnt Signaling