Matrix metalloproteinases (MMPs) are a family of zinc and calcium dependent endopeptidases with the combined ability to degrade all the components of the extracellular matrix (ECM). MMP-16 (MT3-MMP) is found in brain, lung, placenta, smooth muscle cells, and malignant tumor tissues including oral melanoma and renal carcinoma (1). MMP-16 has been shown to activate proMMP-2 and degrade various ECM components including native collagens (2, 3). MMP-16 has been proposed to possess the potential to directly enhance the growth and invasiveness of cells in vivo, two critical processes for development and carcinogenesis (4). Structurally, MMP-16 consists of the following domains: a pro domain containing the furin cleavage site, a catalytic domain containing the zinc-binding site, a hinge region, a hemopexin-like domain, a transmembrane domain, and a cytoplamasic tail (1). The structure of the catalytic domain in complex with a hydroxamate inhibitor has been solved (5). The rhMMP-16PC consists of the pro and catalytic domains, which can be activated by treatment with furin.

Key Product Details
Species Reactivity
Human
Applications
Immunohistochemistry, Western Blot, Immunoprecipitation
Label
Unconjugated
Antibody Source
Polyclonal Goat IgG
Loading...
Product Specifications
Immunogen
E. coli-derived recombinant human MMP‑16/MT3‑MMP
Ala32-Gly291 (Ile152Asn)
Accession # P51512
Ala32-Gly291 (Ile152Asn)
Accession # P51512
Specificity
Detects human MMP‑16/MT3‑MMP in direct ELISAs and Western blots.
Clonality
Polyclonal
Host
Goat
Isotype
IgG
Scientific Data Images for Human MMP‑16/MT3‑MMP Antibody
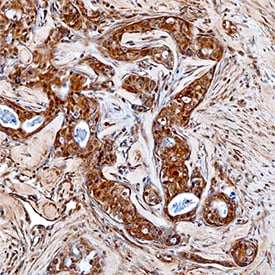
MMP‑16/MT3‑MMP in Human Breast.
MMP-16/MT3-MMP was detected in immersion fixed paraffin-embedded sections of human breast using Goat Anti-Human MMP-16/MT3-MMP Antigen Affinity-purified Polyclonal Antibody (Catalog # AF1785) at 15 µg/mL overnight at 4 °C. Tissue was stained using the Anti-Goat HRP-DAB Cell & Tissue Staining Kit (brown; Catalog # CTS008) and counterstained with hematoxylin (blue). Specific staining was localized to cytoplasm of epithelial cells. View our protocol for Chromogenic IHC Staining of Paraffin-embedded Tissue Sections.Detection of MMP-16/MT3-MMP by Western Blot
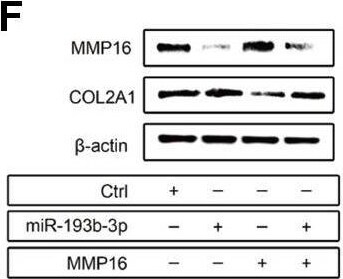
Mechanism: effect of miR-193b-3p’s target gene MMP16 on chondrocyte sheets. (A) Sequence of the predicted miR-193b-3p binding site with the 3’UTR of MMP16 (position 2232-2238). (B) Vectors used in dual-luciferase reporter assay containing the seed sequence of MMP16 3’UTR. (C) Reduced relative luciferase activity transfected with MMP16 and miR-193b-3p, compared with the negative control or MMP16 mutant (p < 0.05) (n=3). (D, E) Gene expression of COL2A1 and MMP16 (p < 0.05) (n=3). (F) Protein expression of COL2A1 and MMP16 for chondrocytes cultured in different groups. (G, H) Quantitative protein level of COL2A1 and MMP16 in different groups (p < 0.05) (n=3). Image collected and cropped by CiteAb from the following open publication (https://pubmed.ncbi.nlm.nih.gov/31492826), licensed under a CC-BY license. Not internally tested by R&D Systems.Applications for Human MMP‑16/MT3‑MMP Antibody
Application
Recommended Usage
Immunohistochemistry
5-15 µg/mL
Sample: Immersion fixed paraffin-embedded sections of human breast
Sample: Immersion fixed paraffin-embedded sections of human breast
Immunoprecipitation
25 µg/mL
Sample: Conditioned cell culture medium spiked with Recombinant Human MMP‑16/MT3-MMP (Catalog # 1785-MP), see our available Western blot detection antibodies
Sample: Conditioned cell culture medium spiked with Recombinant Human MMP‑16/MT3-MMP (Catalog # 1785-MP), see our available Western blot detection antibodies
Western Blot
0.1 µg/mL
Sample: Recombinant Human MMP‑16/MT3-MMP (Catalog # 1785-MP)
Sample: Recombinant Human MMP‑16/MT3-MMP (Catalog # 1785-MP)
Reviewed Applications
Read 1 review rated 5 using AF1785 in the following applications:
Formulation, Preparation, and Storage
Purification
Antigen Affinity-purified
Reconstitution
Reconstitute at 0.2 mg/mL in sterile PBS. For liquid material, refer to CoA for concentration.
Loading...
Formulation
Lyophilized from a 0.2 μm filtered solution in PBS with Trehalose. *Small pack size (SP) is supplied either lyophilized or as a 0.2 µm filtered solution in PBS.
Shipping
Lyophilized product is shipped at ambient temperature. Liquid small pack size (-SP) is shipped with polar packs. Upon receipt, store immediately at the temperature recommended below.
Stability & Storage
Use a manual defrost freezer and avoid repeated freeze-thaw cycles.
- 12 months from date of receipt, -20 to -70 °C as supplied.
- 1 month, 2 to 8 °C under sterile conditions after reconstitution.
- 6 months, -20 to -70 °C under sterile conditions after reconstitution.
Calculators
Background: MMP-16/MT3-MMP
References
- Takino, T. et al. (1995) J. Biol. Chem. 270:23013.
- Shofuda, K. et al. (1997) J. Biol. Chem. 272:9749.
- Shimada, T. et al. (1999) Eur. J. Biochem. 262:907.
- Kang, T. et al. (2000) FASEB J. 14:2559.
- Lang, R. et al. (2004) J. Mol. Biol. 336:213.
Long Name
Matrix Metalloproteinase 16/Membrane Type 3 MMP
Alternate Names
MMP16, MT3-MMP
Entrez Gene IDs
4325 (Human)
Gene Symbol
MMP16
UniProt
Additional MMP-16/MT3-MMP Products
Product Documents for Human MMP‑16/MT3‑MMP Antibody
Certificate of Analysis
To download a Certificate of Analysis, please enter a lot or batch number in the search box below.
Note: Certificate of Analysis not available for kit components.
Product Specific Notices for Human MMP‑16/MT3‑MMP Antibody
For research use only
Related Research Areas
Citations for Human MMP‑16/MT3‑MMP Antibody
Customer Reviews for Human MMP‑16/MT3‑MMP Antibody (1)
5 out of 5
1 Customer Rating
Have you used Human MMP‑16/MT3‑MMP Antibody?
Submit a review and receive an Amazon gift card!
$25/€18/£15/$25CAN/¥2500 Yen for a review with an image
$10/€7/£6/$10CAN/¥1110 Yen for a review without an image
Submit a review
Customer Images
Showing
1
-
1 of
1 review
Showing All
Filter By:
-
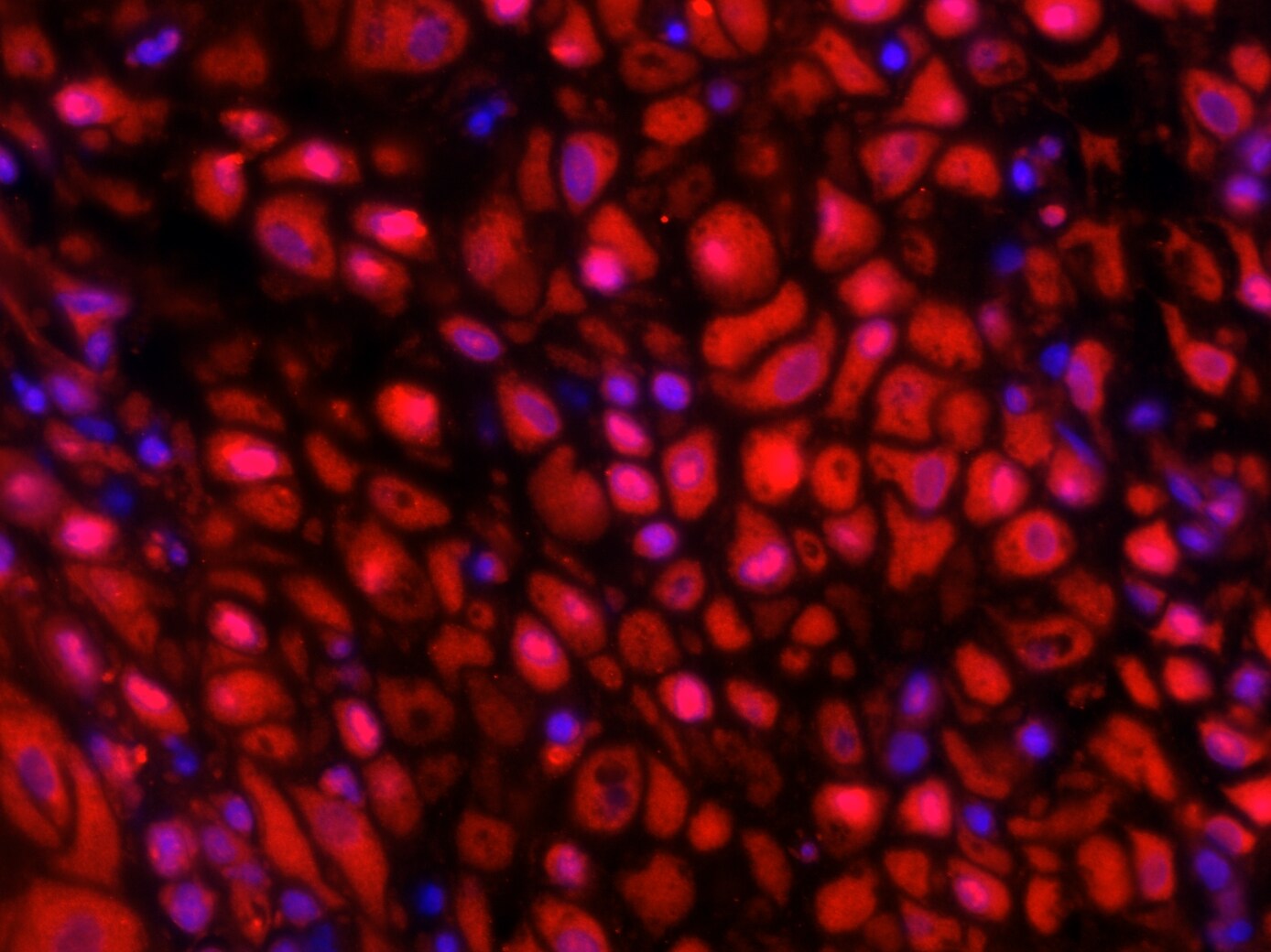
Application: immunofluorescence - paraffinSample Tested: First trimester deciduaSpecies: HumanVerified Customer | Posted 03/18/201740X magnification. MMP16 is expressed by decidual stromal cells.Sodium citrate antigen retrieval. Permeabilized with 0.5% triton X. Primary antibody concentration - 10 ug/ml Secondary antibody - Rabbit Anti-Goat Alexa Fluor® 568 at 4 ug/ml.
There are no reviews that match your criteria.
Protocols
Find general support by application which include: protocols, troubleshooting, illustrated assays, videos and webinars.
- Antigen Retrieval Protocol (PIER)
- Antigen Retrieval for Frozen Sections Protocol
- Appropriate Fixation of IHC/ICC Samples
- Cellular Response to Hypoxia Protocols
- Chromogenic IHC Staining of Formalin-Fixed Paraffin-Embedded (FFPE) Tissue Protocol
- Chromogenic Immunohistochemistry Staining of Frozen Tissue
- ClariTSA™ Fluorophore Kits
- Detection & Visualization of Antibody Binding
- Fluorescent IHC Staining of Frozen Tissue Protocol
- Graphic Protocol for Heat-induced Epitope Retrieval
- Graphic Protocol for the Preparation and Fluorescent IHC Staining of Frozen Tissue Sections
- Graphic Protocol for the Preparation and Fluorescent IHC Staining of Paraffin-embedded Tissue Sections
- Graphic Protocol for the Preparation of Gelatin-coated Slides for Histological Tissue Sections
- IHC Sample Preparation (Frozen sections vs Paraffin)
- Immunofluorescent IHC Staining of Formalin-Fixed Paraffin-Embedded (FFPE) Tissue Protocol
- Immunohistochemistry (IHC) and Immunocytochemistry (ICC) Protocols
- Immunohistochemistry Frozen Troubleshooting
- Immunohistochemistry Paraffin Troubleshooting
- Immunoprecipitation Protocol
- Preparing Samples for IHC/ICC Experiments
- Preventing Non-Specific Staining (Non-Specific Binding)
- Primary Antibody Selection & Optimization
- Protocol for Heat-Induced Epitope Retrieval (HIER)
- Protocol for Making a 4% Formaldehyde Solution in PBS
- Protocol for VisUCyte™ HRP Polymer Detection Reagent
- Protocol for the Preparation & Fixation of Cells on Coverslips
- Protocol for the Preparation and Chromogenic IHC Staining of Frozen Tissue Sections
- Protocol for the Preparation and Chromogenic IHC Staining of Frozen Tissue Sections - Graphic
- Protocol for the Preparation and Chromogenic IHC Staining of Paraffin-embedded Tissue Sections
- Protocol for the Preparation and Chromogenic IHC Staining of Paraffin-embedded Tissue Sections - Graphic
- Protocol for the Preparation and Fluorescent IHC Staining of Frozen Tissue Sections
- Protocol for the Preparation and Fluorescent IHC Staining of Paraffin-embedded Tissue Sections
- Protocol for the Preparation of Gelatin-coated Slides for Histological Tissue Sections
- R&D Systems Quality Control Western Blot Protocol
- TUNEL and Active Caspase-3 Detection by IHC/ICC Protocol
- The Importance of IHC/ICC Controls
- Troubleshooting Guide: Immunohistochemistry
- Troubleshooting Guide: Western Blot Figures
- Western Blot Conditions
- Western Blot Protocol
- Western Blot Protocol for Cell Lysates
- Western Blot Troubleshooting
- Western Blot Troubleshooting Guide
- View all Protocols, Troubleshooting, Illustrated assays and Webinars
Loading...