Best Seller
Human/Mouse/Rat Olig2 Antibody
R&D Systems | Catalog # AF2418


Key Product Details
Species Reactivity
Validated:
Human, Mouse, Rat
Cited:
Human, Mouse, Rat, Avian - Chicken, Ferret, Hamster - Mesocricetus auratus (Golden Hamster), Primate - Macaca mulatta (Rhesus Macaque), Rabies virus, Transgenic Mouse, Xenograft
Applications
Validated:
Immunohistochemistry, Western Blot, Dual RNAscope ISH-IHC Compatible, Immunocytochemistry, Chromatin Immunoprecipitation (ChIP)
Cited:
Immunohistochemistry, Immunohistochemistry-Paraffin, Immunohistochemistry-Frozen, Western Blot, Flow Cytometry, Immunocytochemistry, Chromatin Immunoprecipitation (ChIP), Bioassay, Cell Culture, Co-Immunoprecipitation, FISH
Label
Unconjugated
Antibody Source
Polyclonal Goat IgG
Loading...
Product Specifications
Immunogen
E. coli-derived recombinant human Olig2
Met1-Lys323
Accession # Q13516
Met1-Lys323
Accession # Q13516
Specificity
Detects human Olig2 in direct ELISAs and Western blots.
Clonality
Polyclonal
Host
Goat
Isotype
IgG
Scientific Data Images for Human/Mouse/Rat Olig2 Antibody
Olig2 and Oligodendrocyte Marker O4 in Rat Cortical Stem Cells.
Olig2 and Oligodendrocyte Marker O4 were detected in 7 day differentiated rat cortical stem cells using 10 µg/mL Goat Anti-Human/Mouse/Rat Olig2 Antigen Affinity-purified Polyclonal Antibody (Catalog # AF2418) and 10 µg/mL Goat Anti-Human/Mouse/Rat/Chicken O4 Monoclonal Antibody (Catalog # MAB1326). Cells were incubated with primary antibodies for 3 hours at room temperature. Cells were stained for Olig2 using the NorthernLights™ 637-conjugated Anti-Goat IgG Secondary Antibody (red; Catalog # NL002), and stained for O4 using an anti-mouse IgM secondary antibody (pseudo-stained green). View our protocol for Fluorescent ICC Staining of Cells on Coverslips.Detection of Olig2-regulated Genes by Chromatin Immunoprecipitation.
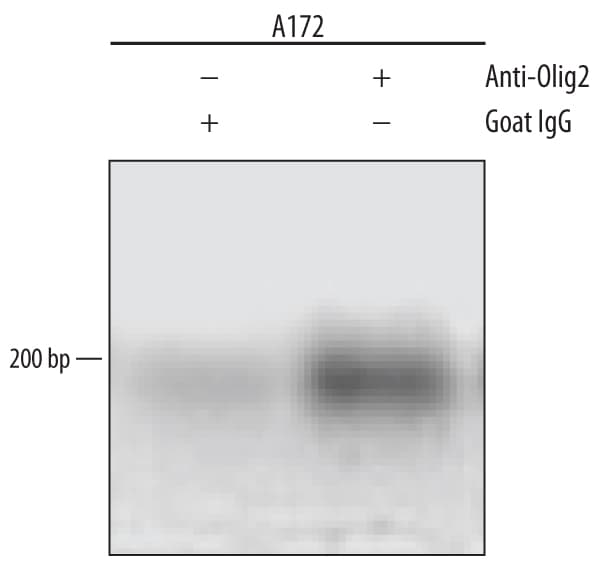
A172 human glioblastoma cell line were fixed using formaldehyde, resuspended in lysis buffer, and sonicated to shear chromatin. Olig2/DNA complexes were immunoprecipitated using 5 µg Goat Anti-Human/Mouse/Rat Olig2 Antigen Affinity-purified Polyclonal Antibody (Catalog # AF2418) or control antibody (Catalog # AB-108-C) for 15 minutes in an ultrasonic bath, followed by Biotinylated Anti-Goat IgG Secondary Antibody (Catalog # BAF109). Immunocomplexes were captured using 50 µL of MagCellect Streptavidin Ferrofluid (Catalog # MAG999) and DNA was purified using chelating resin solution. Thep21promoter was detected by standard PCR.Olig2 in Mouse Embryo.
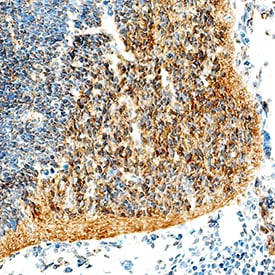
Olig2 was detected in immersion fixed frozen sections of mouse embryo (13 d.p.c.) using Goat Anti-Human/Mouse/Rat Olig2 Antigen Affinity-purified Polyclonal Antibody (Catalog # AF2418) at 8 µg/mL overnight at 4 °C. Tissue was stained using the Anti-Goat HRP-DAB Cell & Tissue Staining Kit (brown; Catalog # CTS008) and counterstained with hematoxylin (blue). Specific staining was localized to developing brain. View our protocol for Chromogenic IHC Staining of Frozen Tissue Sections.Olig2 in Human Brain.
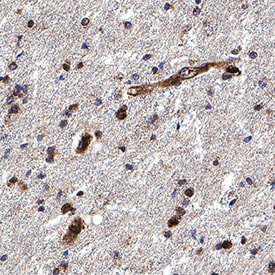
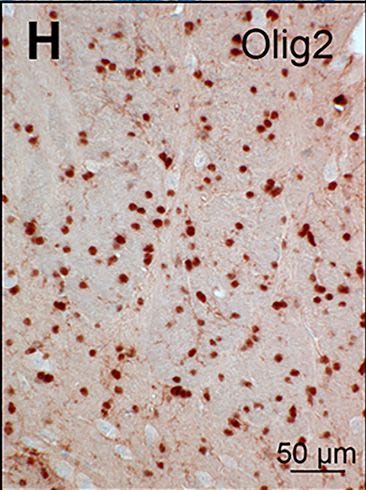
Olig2 was detected in immersion fixed paraffin-embedded sections of human brain (cortex) using Goat Anti-Human/Mouse/Rat Olig2 Antigen Affinity-purified Polyclonal Antibody (Catalog # AF2418) at 10 µg/mL for 1 hour at room temperature followed by incubation with the Anti-Goat IgG VisUCyte™ HRP Polymer Antibody (VC004). Before incubation with the primary antibody, tissue was subjected to heat-induced epitope retrieval using Antigen Retrieval Reagent-Basic (CTS013). Tissue was stained using DAB (brown) and counterstained with hematoxylin (blue). Specific staining was localized to cell nuclei. Staining was performed using our protocol for IHC Staining with VisUCyte HRP Polymer Detection Reagents.Olig2 in Rat Midbrain.
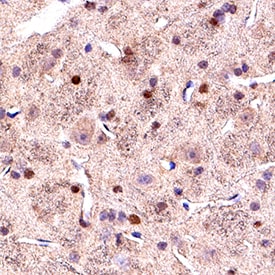
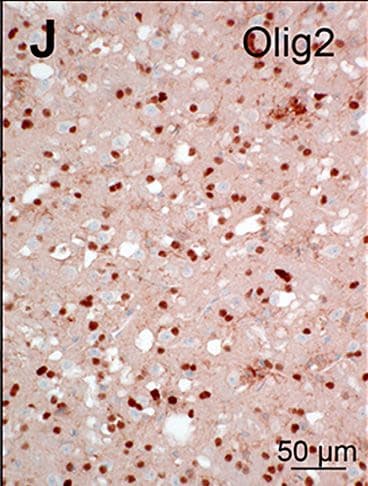
Olig2 was detected in immersion fixed paraffin-embedded sections of rat midbrain using Goat Anti-Human/Mouse/Rat Olig2 Antigen Affinity-purified Polyclonal Antibody (Catalog # AF2418) at 10 µg/mL for 1 hour at room temperature followed by incubation with the Anti-Goat IgG VisUCyte™ HRP Polymer Antibody (VC004). Before incubation with the primary antibody, tissue was subjected to heat-induced epitope retrieval using Antigen Retrieval Reagent-Basic (CTS013). Tissue was stained using DAB (brown) and counterstained with hematoxylin (blue). Specific staining was localized to cell nuclei. Staining was performed using our protocol for IHC Staining with VisUCyte HRP Polymer Detection Reagents.Detection of Mouse Olig2 by Immunocytochemistry/Immunofluorescence
Pathological analysis of l2hgdh-/- mice.The figure compares the histological and ultrastructural appearance of the brain of l2hgdh+/+ (left panels: A, B, E, G, H, K, L) and l2hgdh-/- mice (right panels: C, D, F, I, J, M, N). Hematoxylin and eosin staining shows low-magnification of the lateral part of the brain (A, C) at the level of the lateral ventricle (LV), the caudate putamen (striatum; CPu) and the anterior commissure, anterior part (aca). Higher magnification of pencils of Wilson are shown in panels B and D. Notice the satellite oligodendrocyte at B (arrow). Luxol fast blue staining of myelin is shown in E and F, highlighting the predominant presence of vacuoles in the pencils of Wilson of the striatum of the l2hgdh-/- mouse. Panels G-J are microphotographs from the junction between cortex and white matter in the vicinity of corpus callosum. Immunostaining of NeuN (G and I) shows that most vacuoles in the l2hgdh-/- mice are at distance from the nucleus of neurons (notice the abundance of vacuoles in the lower part of panel I, devoid of neurons) although a few vacuoles indent the nucleus of some neurons (arrowheads). However, the nucleus of satellite oligodendrocytes is also present in close contact to the vacuole indenting the nucleus of some neurons (arrows), preventing to identify clearly which cell type actually contains the peri-neuronal vacuole. In contrast, immunostaining of Olig2 (H and J) clearly shows that most vacuoles are in close contact to the nucleus of oligodendrocytes in the l2hgdh-/- mouse brain. Notice the small size of the nucleus of oligodendrocytes compared to the nucleus of neurons. Panels K-N are Transmission Electron Microscopy photographs of striatum tissue. Ultrastructural analysis (K-N) shows many empty-looking cell processes (asterisks in panels M and N), containing dilated cytoplasmic organelles, in the glia of l2hgdh-/- mice. The myelin sheath of many axons appears focally altered or disrupted (arrows at M and N), sometimes at the vicinity of empty-looking spaces. C, capillary lumen. The length of the scale bar is indicated in each panel. Image collected and cropped by CiteAb from the following publication (https://pubmed.ncbi.nlm.nih.gov/25763823), licensed under a CC-BY license. Not internally tested by R&D Systems.Detection of Rat Olig2 by Immunocytochemistry/Immunofluorescence
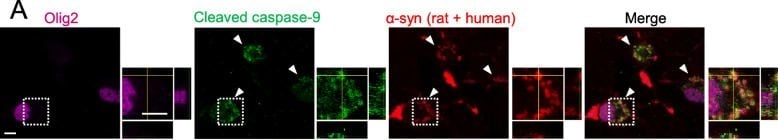
alpha -Syn PFF exposure to immature OLGs during differentiation-induced abnormal oligodendrogenesis. a Confocal images of oligodendroglial cells on day 4 after differentiation induction showing the colocalization of alpha -syn immunoreactivity and cleaved caspase-9 expression. The cells were incubated with 1 μM alpha -syn PFFs for 24 h (day 3–4) before fixation. White arrowheads indicate intracellular inclusions. The regions marked by dotted squares are highlighted in the magnified views. Scale bar = 5 μm. b Scheme showing the experimental protocols of 3 μM alpha -syn PFF exposure to oligodendroglial cells during two different phases (day 0–1, or day 3–4) of differentiation. c Cell viability analysis (WST assay) of day 8 OLGs, which were obtained through different protocols of alpha -syn PFF exposure during differentiation (no exposure, exposure on day 0–1, or exposure on days 3–4). N = 5, respectively, independent culture, Kruskal–Wallis, p* < 0.05. d Immunoblot analysis of day 8 OLGs obtained via different protocols of alpha -syn PFF exposure during differentiation (no exposure, exposure on day 0–1, or exposure on days 3–4), showing the protein expression levels of oligodendroglial cell markers. e, f Quantification of the relative protein expression levels of MBP (e) and BCAS1 (f) in day 8 OLGs obtained using the above-mentioned protocols. The differences between the no exposure group and day 0–1 group in (E) (p = 0.0636) and in (F) (p = 0.1980) were not statistically significant. N = 5, respectively, independent culture, Kruskal–Wallis, p* < 0.05, p** < 0.01. g Graphical representation of in vitro analysis showing two patterns of alpha -syn PFF-induced maturation inhibition. alpha -Syn PFF exposure to differentiating OLGs during the BCAS1(+) cell-dominant phase resulted in decreased cell viability (left), while exposure during the OPC-dominant phase resulted in abnormal protein expression levels of OLG markers (right) Image collected and cropped by CiteAb from the following publication (https://pubmed.ncbi.nlm.nih.gov/32727582), licensed under a CC-BY license. Not internally tested by R&D Systems.Detection of Mouse Olig2 by Immunocytochemistry/Immunofluorescence
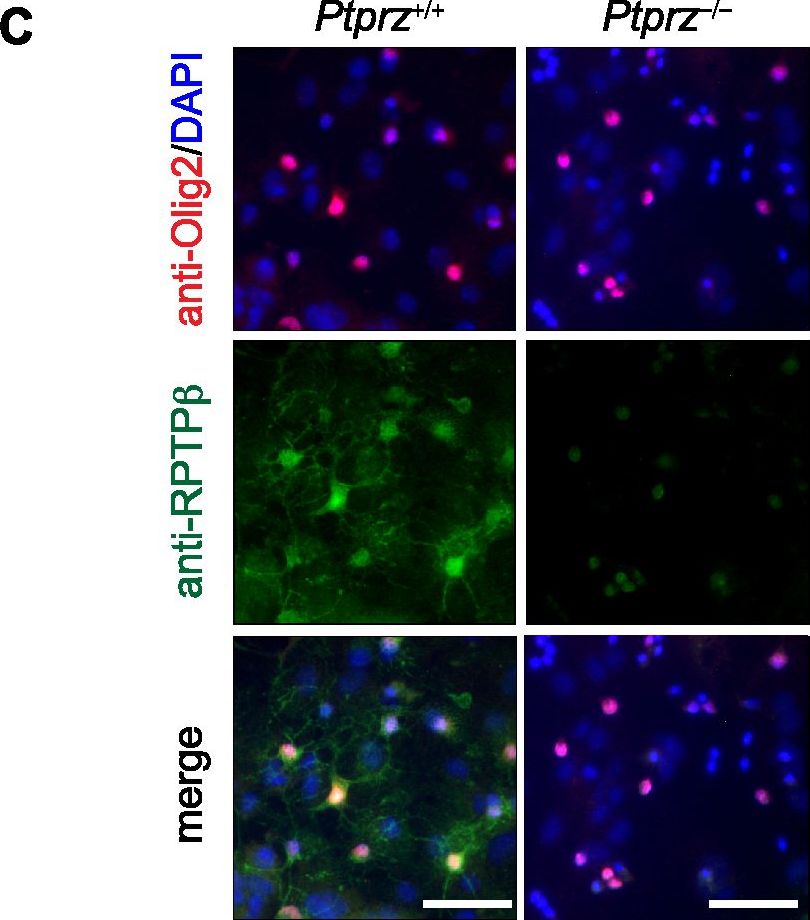
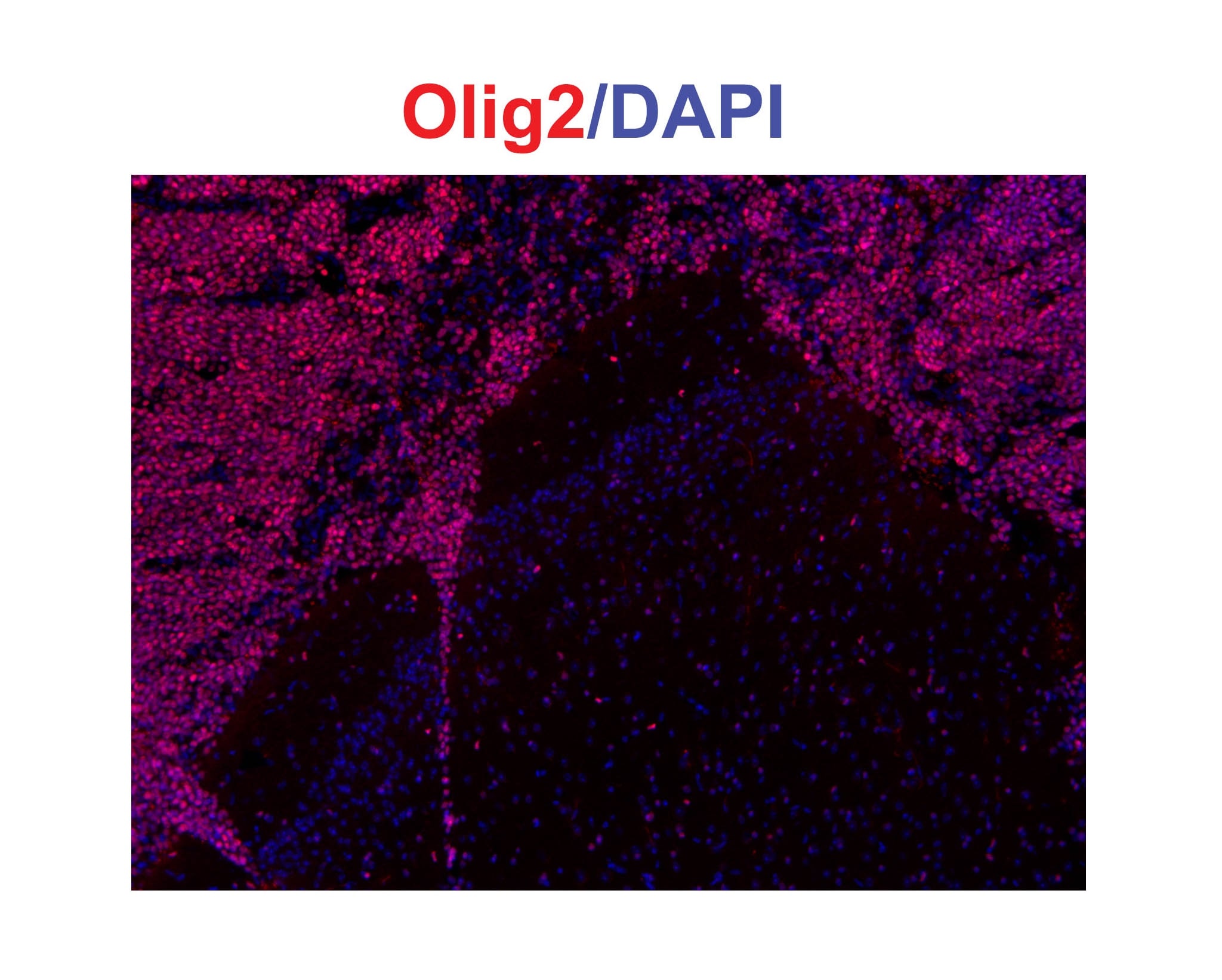
Early onset of oligodendrocyte differentiation in Ptprz-deficient mice.A, Immunohistochemistry of cultured oligodendrocyte precursor cells (OPCs, NG2-postive cells, red) and oligodendrocytes (OLs, MBP-postive cells, green) from Ptprz-deficient mice and wild-type mice. Scale bars, 100 µm. The percentages of OPCs and OLs among total cells (DAPI-positive nuclei, blue) are shown at the right of each panel. Data are the mean ± SEM from five independent experiments. *p<0.05 and **p<0.01 (Student's t-test). B, Morphological assessment of cultured OLs at DIV6. The MBP-postive cells were classified into four categories. Representative images are shown in lower panels; Stage 1, three or less primary processes longer than a cell body with minimal development of secondary and tertiary processes; Stage 2, three or more primary processess with moderate secondary and tertiary processes; Stage 3, five or more primary processes with extensive secondry and teriary processes; Stage 4, extending myelin-like membrane structures and branched processes. Scale bars, 50 µm. Data are the mean ± SEM from three independent experiments. *p<0.05 (Mann-Whitney U-test). C, Ptprz expression in oligodendrocyte lineage cells. Cultured cells at DIV10 were triple stained with anti-RPTP beta (specific for Ptprz receptor isoforms, green), anti-Olig2 (red), and DAPI (blue). Scale bars, 50 µm. Image collected and cropped by CiteAb from the following publication (https://dx.plos.org/10.1371/journal.pone.0048797), licensed under a CC-BY license. Not internally tested by R&D Systems.Detection of Mouse Olig2 by Immunocytochemistry/Immunofluorescence
Pathological analysis of l2hgdh-/- mice.The figure compares the histological and ultrastructural appearance of the brain of l2hgdh+/+ (left panels: A, B, E, G, H, K, L) and l2hgdh-/- mice (right panels: C, D, F, I, J, M, N). Hematoxylin and eosin staining shows low-magnification of the lateral part of the brain (A, C) at the level of the lateral ventricle (LV), the caudate putamen (striatum; CPu) and the anterior commissure, anterior part (aca). Higher magnification of pencils of Wilson are shown in panels B and D. Notice the satellite oligodendrocyte at B (arrow). Luxol fast blue staining of myelin is shown in E and F, highlighting the predominant presence of vacuoles in the pencils of Wilson of the striatum of the l2hgdh-/- mouse. Panels G-J are microphotographs from the junction between cortex and white matter in the vicinity of corpus callosum. Immunostaining of NeuN (G and I) shows that most vacuoles in the l2hgdh-/- mice are at distance from the nucleus of neurons (notice the abundance of vacuoles in the lower part of panel I, devoid of neurons) although a few vacuoles indent the nucleus of some neurons (arrowheads). However, the nucleus of satellite oligodendrocytes is also present in close contact to the vacuole indenting the nucleus of some neurons (arrows), preventing to identify clearly which cell type actually contains the peri-neuronal vacuole. In contrast, immunostaining of Olig2 (H and J) clearly shows that most vacuoles are in close contact to the nucleus of oligodendrocytes in the l2hgdh-/- mouse brain. Notice the small size of the nucleus of oligodendrocytes compared to the nucleus of neurons. Panels K-N are Transmission Electron Microscopy photographs of striatum tissue. Ultrastructural analysis (K-N) shows many empty-looking cell processes (asterisks in panels M and N), containing dilated cytoplasmic organelles, in the glia of l2hgdh-/- mice. The myelin sheath of many axons appears focally altered or disrupted (arrows at M and N), sometimes at the vicinity of empty-looking spaces. C, capillary lumen. The length of the scale bar is indicated in each panel. Image collected and cropped by CiteAb from the following publication (https://pubmed.ncbi.nlm.nih.gov/25763823), licensed under a CC-BY license. Not internally tested by R&D Systems.Detection of Olig2 in Human Cortex.
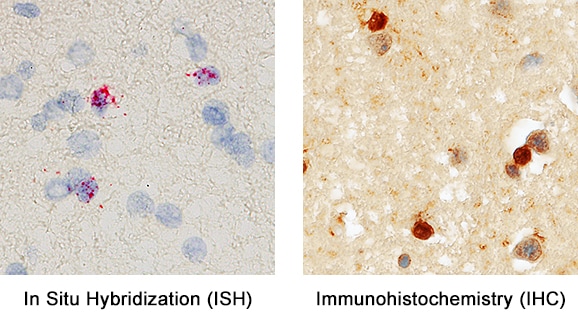
Formalin-fixed paraffin-embedded tissue sections of human cerebral cortex were probed for Olig2 mRNA (ACD RNAScope Probe, catalog #424198; Fast Red chromogen, ACD catalog # 322750). Adjacent tissue section was processed for immunohistochemistry using goat anti-human Olig2 polyclonal antibody (R&D Systems catalog # AF2418) at 3ug/mL with overnight incubation at 4 degrees Celsius followed by incubation with anti-goat IgG VisUCyte HRP Polymer Antibody (Catalog # VC004) and DAB chromogen (yellow-brown). Tissue was counterstained with hematoxylin (blue). Specific staining was localized to glial cells.Detection of Olig2 by Western Blot
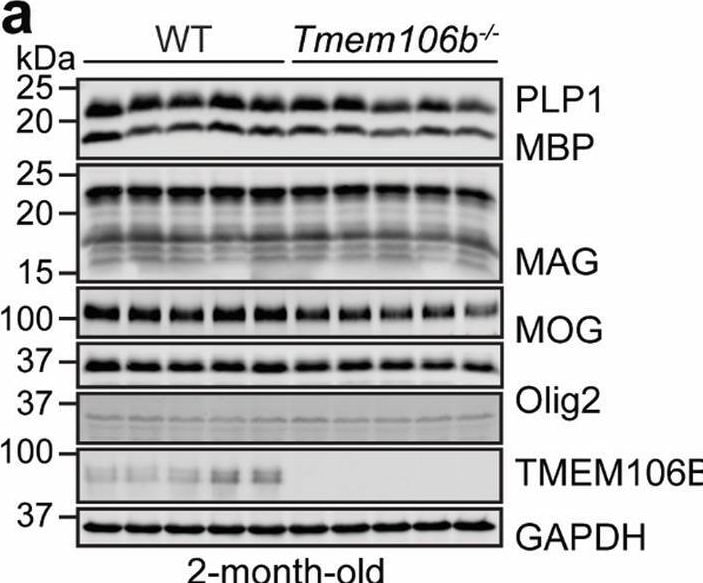
Myelination defects, axonal degeneration of Purkinje cells and disruption of cerebellar cortico-nuclear connection in young Tmem106b−/− mice. a, b Western blot analysis of myelin proteins and GAPDH in 2-month-old WT and Tmem106b−/− cerebellar lysates. Protein levels were quantified and normalized to GAPDH in b. n = 5, *, p < 0.05, **, p < 0.01, unpaired t-test. c–e Cerebellar sections from 5‐month‐old WT and Tmem106b−/− mice were co-stained with anti-calbindin, myelin basic protein (MBP), and NF-H antibodies. MBP intensity around Purkinje cell axon and the number of giant torpedos in the axon of Purkinje cells were quantified in d and e, respectively. Scale bar = 10 µm. n = 3–4, ***, p < 0.001, unpaired t-test. f Cerebellar sections from 5‐month‐old Tmem106b−/− mice were co-stained with anti-calbindin and Cath D antibodies. Scale bar = 10 µm. g, h Cerebellar sections from 2‐month‐old WT and Tmem106b−/− mice were immunostained with antibodies of calbindin, synaptophysin (SYN, presynaptic marker), and MAP2. The intensity of SYN around MAP2-positive soma in the DCN region was quantified in h. Scale bar = 10 µm. n = 3, ***, p < 0.001, non-parametric test (Mann Whitney test) Image collected and cropped by CiteAb from the following open publication (https://pubmed.ncbi.nlm.nih.gov/35287730), licensed under a CC-BY license. Not internally tested by R&D Systems.Applications for Human/Mouse/Rat Olig2 Antibody
Application
Recommended Usage
Chromatin Immunoprecipitation (ChIP)
5 µg/5 x 106 cells
Sample: A172 human glioblastoma cell line chromatin, p21 promoter detected by standard PCR.
Sample: A172 human glioblastoma cell line chromatin, p21 promoter detected by standard PCR.
Dual RNAscope ISH-IHC Compatible
5-15 µg/mL
Sample: Immersion fixed paraffin-embedded sections of human cortex
Sample: Immersion fixed paraffin-embedded sections of human cortex
Immunocytochemistry
5-15 µg/mL
Sample: Immersion fixed 7 day differentiated rat cortical stem cells
Sample: Immersion fixed 7 day differentiated rat cortical stem cells
Immunohistochemistry
5-15 µg/mL
Sample: Immersion fixed frozen sections of mouse embryo (E13), human brain (cortex), and rat midbrain.
Sample: Immersion fixed frozen sections of mouse embryo (E13), human brain (cortex), and rat midbrain.
Western Blot
0.1 µg/mL
Sample: Recombinant Human Olig2
Sample: Recombinant Human Olig2
Reviewed Applications
Read 6 reviews rated 4.8 using AF2418 in the following applications:
Formulation, Preparation, and Storage
Purification
Antigen Affinity-purified
Reconstitution
Reconstitute at 0.2 mg/mL in sterile PBS. For liquid material, refer to CoA for concentration.
Loading...
Formulation
Lyophilized from a 0.2 μm filtered solution in PBS with Trehalose. See Certificate of Analysis for details.
*Small pack size (-SP) is supplied either lyophilized or as a 0.2 µm filtered solution in PBS.
*Small pack size (-SP) is supplied either lyophilized or as a 0.2 µm filtered solution in PBS.
Shipping
Lyophilized product is shipped at ambient temperature. Liquid small pack size (-SP) is shipped with polar packs. Upon receipt, store immediately at the temperature recommended below.
Stability & Storage
Use a manual defrost freezer and avoid repeated freeze-thaw cycles.
- 12 months from date of receipt, -20 to -70 °C as supplied.
- 1 month, 2 to 8 °C under sterile conditions after reconstitution.
- 6 months, -20 to -70 °C under sterile conditions after reconstitution.
Calculators
Background: Olig2
Long Name
Oligodendrocyte Lineage Transcription Factor 2
Alternate Names
BHLHB1, PRKCBP2, RACK17
Entrez Gene IDs
10215 (Human)
Gene Symbol
OLIG2
UniProt
Additional Olig2 Products
Product Documents for Human/Mouse/Rat Olig2 Antibody
Certificate of Analysis
To download a Certificate of Analysis, please enter a lot or batch number in the search box below.
Note: Certificate of Analysis not available for kit components.
Product Specific Notices for Human/Mouse/Rat Olig2 Antibody
For research use only
Citations for Human/Mouse/Rat Olig2 Antibody
Customer Reviews for Human/Mouse/Rat Olig2 Antibody (6)
4.8 out of 5
6 Customer Ratings
Have you used Human/Mouse/Rat Olig2 Antibody?
Submit a review and receive an Amazon gift card!
$25/€18/£15/$25CAN/¥2500 Yen for a review with an image
$10/€7/£6/$10CAN/¥1110 Yen for a review without an image
Submit a review
Customer Images
Showing
1
-
5 of
6 reviews
Showing All
Filter By:
-
Application: Immunocytochemistry/ImmunofluorescenceSample Tested: Spinal cordSpecies: MouseVerified Customer | Posted 07/09/2019This antibody worked nicely 1:500 or 1:1000 of mouse spinal cord perfused with 4% PFA.
-
Application: Immunohistochemistry-FrozenSample Tested: Mouse brainSpecies: MouseVerified Customer | Posted 05/12/2019Olig2 positive cells in the mouse corpus callosum
-
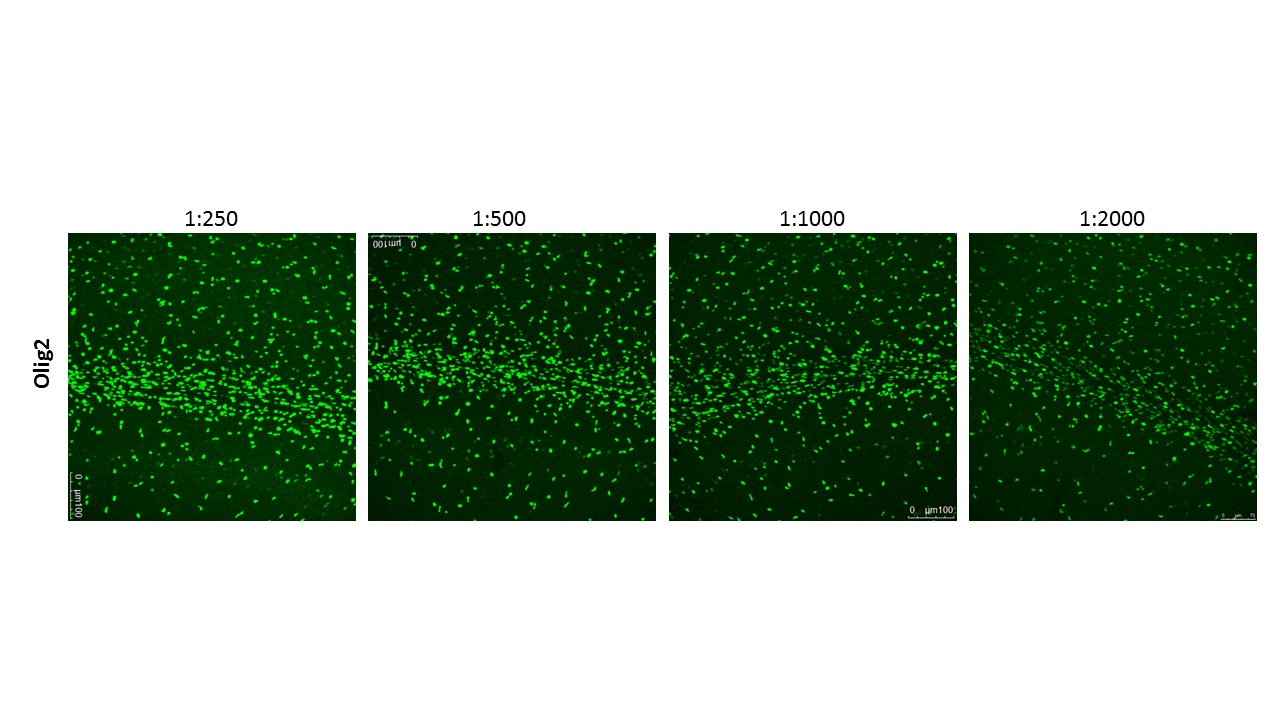
Application: ImmunohistochemistrySample Tested: Adult brainSpecies: MouseVerified Customer | Posted 06/20/2018This antibody worked beautifully at 1:2000 in 30um free-floating sections of adult (P25) mouse brain perfused with 4% PFA and cryoprotected. Region imaged: corpus callosum above hippocampus. Protocol: 1. Antigen Retrieval: 100mM Sodium Citrate pH6.0, 0.05% Tween, in microwave 2. Rinse sections PBS once 3. Block in 5% NDS in PBSTx 1% RT for 1 hour 4. Primary Antibody (1:2000) in 1% NDS in PBSTx 0.3% (Incubate RT overnight on shaker) 5. Wash 3x 10 min PBS 6. Secondary Antibody (1:600) in 1% NDS in PBSTx 0.3% (Incubate RT 2h on shaker) 7. Wash 3x 10 min PBS 8. Mount on slides in water and let dry 9. Coverslip using Prolong Gold
-
Application: Immunocytochemistry/ImmunofluorescenceSample Tested: GliomaSpecies: HumanVerified Customer | Posted 01/04/2018Glioma xenografted in NOD SCID mice -> olig2 in red
-
Application: Affinity PurificationSample Tested: Brain sectionsSpecies: Rat Sprague DawleyVerified Customer | Posted 12/07/2015Dilution 1:500
-
Application: ImmunocytochemistrySample Tested: See PMID 22504067Species: MouseVerified Customer | Posted 01/06/2015
There are no reviews that match your criteria.
Protocols
Find general support by application which include: protocols, troubleshooting, illustrated assays, videos and webinars.
- Antigen Retrieval Protocol (PIER)
- Antigen Retrieval for Frozen Sections Protocol
- Appropriate Fixation of IHC/ICC Samples
- Cellular Response to Hypoxia Protocols
- ChIP Protocol Video
- Chromatin Immunoprecipitation (ChIP) Protocol
- Chromatin Immunoprecipitation Protocol
- Chromogenic IHC Staining of Formalin-Fixed Paraffin-Embedded (FFPE) Tissue Protocol
- Chromogenic Immunohistochemistry Staining of Frozen Tissue
- ClariTSA™ Fluorophore Kits
- Detection & Visualization of Antibody Binding
- Fluorescent IHC Staining of Frozen Tissue Protocol
- Graphic Protocol for Heat-induced Epitope Retrieval
- Graphic Protocol for the Preparation and Fluorescent IHC Staining of Frozen Tissue Sections
- Graphic Protocol for the Preparation and Fluorescent IHC Staining of Paraffin-embedded Tissue Sections
- Graphic Protocol for the Preparation of Gelatin-coated Slides for Histological Tissue Sections
- ICC Cell Smear Protocol for Suspension Cells
- ICC Immunocytochemistry Protocol Videos
- ICC for Adherent Cells
- IHC Sample Preparation (Frozen sections vs Paraffin)
- ISH-IHC Protocol for Chromogenic Detection on Formalin Fixed Paraffin Embedded (FFPE) Tissue
- Immunocytochemistry (ICC) Protocol
- Immunocytochemistry Troubleshooting
- Immunofluorescence of Organoids Embedded in Cultrex Basement Membrane Extract
- Immunofluorescent IHC Staining of Formalin-Fixed Paraffin-Embedded (FFPE) Tissue Protocol
- Immunohistochemistry (IHC) and Immunocytochemistry (ICC) Protocols
- Immunohistochemistry Frozen Troubleshooting
- Immunohistochemistry Paraffin Troubleshooting
- Preparing Samples for IHC/ICC Experiments
- Preventing Non-Specific Staining (Non-Specific Binding)
- Primary Antibody Selection & Optimization
- Protocol for Heat-Induced Epitope Retrieval (HIER)
- Protocol for Making a 4% Formaldehyde Solution in PBS
- Protocol for VisUCyte™ HRP Polymer Detection Reagent
- Protocol for the Fluorescent ICC Staining of Cell Smears - Graphic
- Protocol for the Fluorescent ICC Staining of Cultured Cells on Coverslips - Graphic
- Protocol for the Preparation & Fixation of Cells on Coverslips
- Protocol for the Preparation and Chromogenic IHC Staining of Frozen Tissue Sections
- Protocol for the Preparation and Chromogenic IHC Staining of Frozen Tissue Sections - Graphic
- Protocol for the Preparation and Chromogenic IHC Staining of Paraffin-embedded Tissue Sections
- Protocol for the Preparation and Chromogenic IHC Staining of Paraffin-embedded Tissue Sections - Graphic
- Protocol for the Preparation and Fluorescent ICC Staining of Cells on Coverslips
- Protocol for the Preparation and Fluorescent ICC Staining of Non-adherent Cells
- Protocol for the Preparation and Fluorescent ICC Staining of Stem Cells on Coverslips
- Protocol for the Preparation and Fluorescent IHC Staining of Frozen Tissue Sections
- Protocol for the Preparation and Fluorescent IHC Staining of Paraffin-embedded Tissue Sections
- Protocol for the Preparation of Gelatin-coated Slides for Histological Tissue Sections
- Protocol for the Preparation of a Cell Smear for Non-adherent Cell ICC - Graphic
- R&D Systems Quality Control Western Blot Protocol
- TUNEL and Active Caspase-3 Detection by IHC/ICC Protocol
- The Importance of IHC/ICC Controls
- Troubleshooting Guide: Immunohistochemistry
- Troubleshooting Guide: Western Blot Figures
- Western Blot Conditions
- Western Blot Protocol
- Western Blot Protocol for Cell Lysates
- Western Blot Troubleshooting
- Western Blot Troubleshooting Guide
- View all Protocols, Troubleshooting, Illustrated assays and Webinars
Loading...
Associated Pathways