Neuropilin-1 (Npn-1, previously neuropilin; also CD304) is a 130‑140 kDa type I transmembrane (TM) glycoprotein that regulates axon guidance and angiogenesis (1‑4). The full-length 923 amino acid (aa) human Npn‑1 contains a 623 aa extracellular domain (ECD) that shows 92‑95% aa identity with mouse, rat, bovine and canine
Npn‑1 (3, 4). The ECD contains two N-terminal CUB domains (termed a1a2), two domains with homology to coagulation factors V and VIII (b1b2) and a MAM (meprin) domain (c). C-terminally divergent splice variants with 704, 644, 609, and 551 aa lack the MAM and TM domains and are demonstrated or presumed to be soluble antagonists (1, 5‑7). A 906 aa form lacks a TM segment, but secretion has not been found (8). The sema domains of Class III secreted semaphorins such as Sema3A bind Npn-1 a1a2 (9). Heparin, the heparin-binding forms of VEGF (VEGF165, VEGF-B and VEGF-E), PlGF (PlGF2), and the C-terminus of Sema3 bind the b1b2 region (9, 10). Npn-1 and Npn-2 share 48% aa identity within the ECD and can form homo- and hetero-oligomers via interaction of their MAM domains (1). Neuropilins show partially overlapping expression in neuronal and endothelial cells during development (1, 2). Both neuropilins act as co‑receptors with plexins, mainly plexin A3 and A4, to bind class III semaphorins that mediate axon repulsion (11). However, only Npn-1 binds Sema3A, and only Npn-2 binds Sema3F (1). Both are co-receptors with VEGF R2 (also called KDR or Flk-1) for VEGF165 binding (1). Sema3A signaling can be blocked by VEGF165, which has higher affinity for Npn-1 (12). Npn-1 is preferentially expressed in arteries during development or those undergoing remodeling (1, 2). Npn-1 is also expressed on dendritic cells and mediates DC-induced T cell proliferation (13).

Key Product Details
Species Reactivity
Validated:
Human
Cited:
Human, Mouse
Applications
Validated:
Flow Cytometry, CyTOF-ready
Cited:
Neutralization, Flow Cytometry, Immunoprecipitation
Label
Unconjugated
Antibody Source
Monoclonal Mouse IgG2A Clone # 446921
Loading...
Product Specifications
Immunogen
Mouse myeloma cell line NS0-derived recombinant human Neuropilin‑1
Phe22-Lys644
Accession # NP_001019799
Phe22-Lys644
Accession # NP_001019799
Specificity
Detects human Neuropilin-1 in direct ELISAs.
Clonality
Monoclonal
Host
Mouse
Isotype
IgG2A
Scientific Data Images for Human Neuropilin‑1 Antibody
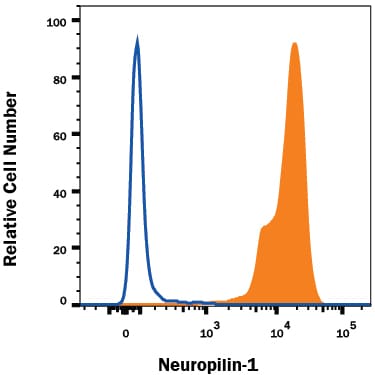
Detection of Neuropilin‑1 in HUVEC Human Cells by Flow Cytometry.
HUVEC human umbilical vein endothelial cells were stained with Human Neuropilin-1 Monoclonal Antibody (Catalog # MAB3870, filled histogram) or isotype control antibody (Catalog # MAB003, open histogram), followed by Phycoerythrin-conjugated Anti-Mouse IgG F(ab')2Secondary Antibody (Catalog # F0102B).Applications for Human Neuropilin‑1 Antibody
Application
Recommended Usage
CyTOF-ready
Ready to be labeled using established conjugation methods. No BSA or other carrier proteins that could interfere with conjugation.
Flow Cytometry
2.5 µg/106 cells
Sample: HUVEC human umbilical vein endothelial cells
Sample: HUVEC human umbilical vein endothelial cells
Reviewed Applications
Read 1 review rated 5 using MAB3870 in the following applications:
Flow Cytometry Panel Builder
Bio-Techne Knows Flow Cytometry
Save time and reduce costly mistakes by quickly finding compatible reagents using the Panel Builder Tool.
Advanced Features
- Spectra Viewer - Custom analysis of spectra from multiple fluorochromes
- Spillover Popups - Visualize the spectra of individual fluorochromes
- Antigen Density Selector - Match fluorochrome brightness with antigen density
Formulation, Preparation, and Storage
Purification
Protein A or G purified from hybridoma culture supernatant
Reconstitution
Reconstitute at 0.5 mg/mL in sterile PBS. For liquid material, refer to CoA for concentration.
Loading...
Formulation
Lyophilized from a 0.2 μm filtered solution in PBS with Trehalose. *Small pack size (SP) is supplied either lyophilized or as a 0.2 µm filtered solution in PBS.
Shipping
Lyophilized product is shipped at ambient temperature. Liquid small pack size (-SP) is shipped with polar packs. Upon receipt, store immediately at the temperature recommended below.
Stability & Storage
Use a manual defrost freezer and avoid repeated freeze-thaw cycles.
- 12 months from date of receipt, -20 to -70 °C as supplied.
- 1 month, 2 to 8 °C under sterile conditions after reconstitution.
- 6 months, -20 to -70 °C under sterile conditions after reconstitution.
Calculators
Background: Neuropilin-1
References
- Bielenberg, D.R. et al. (2006) Exp. Cell Res. 312:584.
- Gu, C. et al. (2003) Dev. Cell 5:45.
- He, Z. and M. Tessier-Lavigne (1997) Cell 90:739.
- Soker, S. et al. (1998) Cell 92:735.
- Gagnon, M.L. et al. (2000) Proc. Natl. Acad. Sci. USA 97:2573.
- Cackowski, F.C. et al. (2004) Genomics 84:82.
- Rossignol, M. et al. (2000) Genomics 70:211.
- Tao, Q. et al. (2003) Angiogenesis 6:39.
- Gu, C. et al. (2002) J. Biol. Chem. 277:18069.
- Mamluk, R. et al. (2002) J. Biol. Chem. 277:24818.
- Yaron, A. et al. (2005) Neuron 45:513.
- Narazaki, M. and G. Tosato (2006) Blood 107:3892.
- Tordjman, R. et al. (2002) Nat. Immunol. 3477.
Alternate Names
BDCA-4, CD304, Neuropilin1, NRP1
Gene Symbol
NRP1
UniProt
Additional Neuropilin-1 Products
Product Documents for Human Neuropilin‑1 Antibody
Certificate of Analysis
To download a Certificate of Analysis, please enter a lot or batch number in the search box below.
Note: Certificate of Analysis not available for kit components.
Product Specific Notices for Human Neuropilin‑1 Antibody
For research use only
Related Research Areas
Citations for Human Neuropilin‑1 Antibody
Customer Reviews for Human Neuropilin‑1 Antibody (1)
5 out of 5
1 Customer Rating
Have you used Human Neuropilin‑1 Antibody?
Submit a review and receive an Amazon gift card!
$25/€18/£15/$25CAN/¥2500 Yen for a review with an image
$10/€7/£6/$10CAN/¥1110 Yen for a review without an image
Submit a review
Showing
1
-
1 of
1 review
Showing All
Filter By:
-
Application: ImmunofluorescenceSample Tested: See PMID 22986738Species: HumanVerified Customer | Posted 02/20/2015
There are no reviews that match your criteria.
Protocols
Find general support by application which include: protocols, troubleshooting, illustrated assays, videos and webinars.
- 7-Amino Actinomycin D (7-AAD) Cell Viability Flow Cytometry Protocol
- Extracellular Membrane Flow Cytometry Protocol
- Flow Cytometry Protocol for Cell Surface Markers
- Flow Cytometry Protocol for Staining Membrane Associated Proteins
- Flow Cytometry Staining Protocols
- Flow Cytometry Troubleshooting Guide
- Intracellular Flow Cytometry Protocol Using Alcohol (Methanol)
- Intracellular Flow Cytometry Protocol Using Detergents
- Intracellular Nuclear Staining Flow Cytometry Protocol Using Detergents
- Intracellular Staining Flow Cytometry Protocol Using Alcohol Permeabilization
- Intracellular Staining Flow Cytometry Protocol Using Detergents to Permeabilize Cells
- Propidium Iodide Cell Viability Flow Cytometry Protocol
- Protocol for Liperfluo
- Protocol for the Characterization of Human Th22 Cells
- Protocol for the Characterization of Human Th9 Cells
- Protocol: Annexin V and PI Staining by Flow Cytometry
- Protocol: Annexin V and PI Staining for Apoptosis by Flow Cytometry
- Troubleshooting Guide: Fluorokine Flow Cytometry Kits
- View all Protocols, Troubleshooting, Illustrated assays and Webinars
Loading...
Associated Pathways
 VEGF - VEGF R2 Signaling Pathways
VEGF - VEGF R2 Signaling Pathways


