Tenascin C, also known as hexabrachion, cytotactin, neuronectin, GMEM, JI, myotendinous antigen, glioma-associated-extracellular matrix antigen, and GP 150‑225, is a member of the Tenascin family of extracellular matrix proteins. It is secreted as a disulfide-linked homohexamer whose subunits can vary in size from approximately 200 kDa to over 300 kDa due to differences in glycosylation (1). Rotary-shadowed electron micrographs of the purified molecule show six strands joined to one another at one end in a globular domain with each arm terminating in a knob-like structure (2, 3). The human Tenascin C monomer is synthesized as a precursor with a 22 amino acid (aa) signal sequence and a 2179 aa mature chain. The mature chain consists of a coiled-coil region (aa 118‑145), followed by 15 EGF‑like domains, 15 fibronectin type-III domains, and a fibrinogen C-terminal domain. In addition, there are 23 potential sites of N-linked glycosylation. Alternative splicing within the fibronectin type-III repeats produces six isoforms for human Tenascin C. Mature human Tenascin C (isoform 1) shares 84% aa sequence identity with mature mouse Tenascin C. In the developing embryo, Tenascin C is expressed during neural, skeletal, and vascular morphogenesis (1, 2). In the adult, it virtually disappears with continued basal expression detectable only in tendon-associated tissues (1, 2). However, great up‑regulation in expression occurs in tissues undergoing remodeling processes seen during wound repair and neovascularization or in pathological states such as inflammation or tumorigenesis (1, 4, 5). Biologically, Tenascin C functions as an adhesion-modulatory extracellular matrix protein (1, 4‑8). Specifically, it antagonizes the adhesive effects of fibronectin, and impacts the ability of fibroblasts to deposit and contract the matrix by affecting the morphology and signaling pathways of adherent cells (5‑7). Tenascin C acts by blocking syndecan-4 binding at the edges of the wound and by suppressing fibronectin-mediated activation of RhoA and focal adhesion kinase (FAK) (4‑8). Tenascin C thus promotes epidermal cell migration and proliferation during wound repair.


Key Product Details
Species Reactivity
Validated:
Human
Cited:
Human, Mouse, Transgenic Mouse
Applications
Validated:
Western Blot, Immunocytochemistry
Cited:
Western Blot
Label
Unconjugated
Antibody Source
Polyclonal Goat IgG
Loading...
Product Specifications
Immunogen
Mouse myeloma cell line NS0-derived recombinant human Tenascin C
Ser186-Pro625
Accession # P24821
Ser186-Pro625
Accession # P24821
Specificity
Detects human Tenascin C in direct ELISAs and Western blots.
Clonality
Polyclonal
Host
Goat
Isotype
IgG
Scientific Data Images for Human Tenascin C Antibody
Detection of Human Tenascin C by Western Blot.
Western blot shows lysates of U-87 MG human glioblastoma/astrocytoma cell line. PVDF membrane was probed with 1 µg/mL of Goat Anti-Human Tenascin C Antigen Affinity-purified Polyclonal Antibody (Catalog # AF3358) followed by HRP-conjugated Anti-Goat IgG Secondary Antibody (Catalog # HAF019). A specific band was detected for Tenascin C at approximately 250 kDa (as indicated). This experiment was conducted under reducing conditions and using Immunoblot Buffer Group 1.Tenascin C in U‑87 MG Human Cell Line.
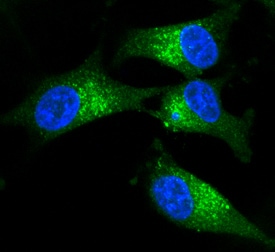
Tenascin C was detected in immersion fixed U-87 MG human glioblastoma/astrocytoma cell line using Goat Anti-Human Tenascin C Antigen Affinity-purified Polyclonal Antibody (Catalog # AF3358) at 10 µg/mL for 3 hours at room temperature. Cells were stained using the NorthernLights™ 493-conjugated Anti-Goat IgG Secondary Antibody (green; Catalog # NL003) and counterstained with DAPI(blue). Specific staining was localized to cytoplasm. View our protocol for Fluorescent ICC Staining of Cells on Coverslips.Detection of Human Tenascin C by Immunohistochemistry
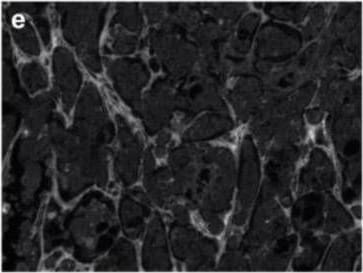
Breast cancer brain metastases exhibit remodeled extracellular matrix. Non-cancerous control brain resections (a–c) and breast cancer brain metastases resections (d–f) were stained for collagen I (anti-collagen I), tenascin C (anti-tenascin C), and hyaluronan (hyaluronic acid binding protein). Representative images were selected across six patients (n = 6). Scale bar = 200 μm. Image collected and cropped by CiteAb from the following open publication (https://pubmed.ncbi.nlm.nih.gov/35200398), licensed under a CC-BY license. Not internally tested by R&D Systems.Detection of Human Tenascin C by Immunohistochemistry
Breast cancer brain metastases exhibit remodeled extracellular matrix. Non-cancerous control brain resections (a–c) and breast cancer brain metastases resections (d–f) were stained for collagen I (anti-collagen I), tenascin C (anti-tenascin C), and hyaluronan (hyaluronic acid binding protein). Representative images were selected across six patients (n = 6). Scale bar = 200 μm. Image collected and cropped by CiteAb from the following open publication (https://pubmed.ncbi.nlm.nih.gov/35200398), licensed under a CC-BY license. Not internally tested by R&D Systems.Applications for Human Tenascin C Antibody
Application
Recommended Usage
Immunocytochemistry
5-15 µg/mL
Sample: Immersion fixed U-87 MG human glioblastoma/astrocytoma cell line
Sample: Immersion fixed U-87 MG human glioblastoma/astrocytoma cell line
Western Blot
1 µg/mL
Sample: U‑87 MG human glioblastoma/astrocytoma cell line
Sample: U‑87 MG human glioblastoma/astrocytoma cell line
Reviewed Applications
Read 1 review rated 4 using AF3358 in the following applications:
Formulation, Preparation, and Storage
Purification
Antigen Affinity-purified
Reconstitution
Reconstitute at 0.2 mg/mL in sterile PBS. For liquid material, refer to CoA for concentration.
Loading...
Formulation
Lyophilized from a 0.2 μm filtered solution in PBS with Trehalose. *Small pack size (SP) is supplied either lyophilized or as a 0.2 µm filtered solution in PBS.
Shipping
Lyophilized product is shipped at ambient temperature. Liquid small pack size (-SP) is shipped with polar packs. Upon receipt, store immediately at the temperature recommended below.
Stability & Storage
Use a manual defrost freezer and avoid repeated freeze-thaw cycles.
- 12 months from date of receipt, -20 to -70 °C as supplied.
- 1 month, 2 to 8 °C under sterile conditions after reconstitution.
- 6 months, -20 to -70 °C under sterile conditions after reconstitution.
Calculators
Background: Tenascin C
References
- Hsia, H.C. and J.E. Schwarzbauer (2005) J. Biol. Chem. 280:26641.
- Nies, D.E. et al. (1991) J. Biol. Chem. 266:2818.
- Erickson, H.P and J.L. Iglesias (1984) Nature 311:267.
- Orend, G. et al. (2003) Oncogene 22:3917.
- Wenk, M.B. et al. (2000) J. Cell Biol. 150:913.
- Midwood, K.S. et al. (2004) Mol. Biol. Cell 15:5670.
- Midwood, K.S. and J. E. Schwarzbauer (2002) Mol. Biol. Cell 13:3601.
- Hsia, H.C. and J.E. Schwarzbauer (2006) J. Surg. Res. 136:92.
Alternate Names
Cytotactin, HXB, Tenascin J1, TNC
Gene Symbol
TNC
UniProt
Additional Tenascin C Products
Product Documents for Human Tenascin C Antibody
Certificate of Analysis
To download a Certificate of Analysis, please enter a lot or batch number in the search box below.
Note: Certificate of Analysis not available for kit components.
Product Specific Notices for Human Tenascin C Antibody
For research use only
Related Research Areas
Citations for Human Tenascin C Antibody
Customer Reviews for Human Tenascin C Antibody (1)
4 out of 5
1 Customer Rating
Have you used Human Tenascin C Antibody?
Submit a review and receive an Amazon gift card!
$25/€18/£15/$25CAN/¥2500 Yen for a review with an image
$10/€7/£6/$10CAN/¥1110 Yen for a review without an image
Submit a review
Showing
1
-
1 of
1 review
Showing All
Filter By:
-
Application: Western BlotSample Tested: See PMID 23084751Species: MouseVerified Customer | Posted 01/07/2015
There are no reviews that match your criteria.
Protocols
Find general support by application which include: protocols, troubleshooting, illustrated assays, videos and webinars.
- Appropriate Fixation of IHC/ICC Samples
- Cellular Response to Hypoxia Protocols
- ClariTSA™ Fluorophore Kits
- Detection & Visualization of Antibody Binding
- ICC Cell Smear Protocol for Suspension Cells
- ICC Immunocytochemistry Protocol Videos
- ICC for Adherent Cells
- Immunocytochemistry (ICC) Protocol
- Immunocytochemistry Troubleshooting
- Immunofluorescence of Organoids Embedded in Cultrex Basement Membrane Extract
- Immunohistochemistry (IHC) and Immunocytochemistry (ICC) Protocols
- Preparing Samples for IHC/ICC Experiments
- Preventing Non-Specific Staining (Non-Specific Binding)
- Primary Antibody Selection & Optimization
- Protocol for VisUCyte™ HRP Polymer Detection Reagent
- Protocol for the Fluorescent ICC Staining of Cell Smears - Graphic
- Protocol for the Fluorescent ICC Staining of Cultured Cells on Coverslips - Graphic
- Protocol for the Preparation and Fluorescent ICC Staining of Cells on Coverslips
- Protocol for the Preparation and Fluorescent ICC Staining of Non-adherent Cells
- Protocol for the Preparation and Fluorescent ICC Staining of Stem Cells on Coverslips
- Protocol for the Preparation of a Cell Smear for Non-adherent Cell ICC - Graphic
- R&D Systems Quality Control Western Blot Protocol
- TUNEL and Active Caspase-3 Detection by IHC/ICC Protocol
- The Importance of IHC/ICC Controls
- Troubleshooting Guide: Western Blot Figures
- Western Blot Conditions
- Western Blot Protocol
- Western Blot Protocol for Cell Lysates
- Western Blot Troubleshooting
- Western Blot Troubleshooting Guide
- View all Protocols, Troubleshooting, Illustrated assays and Webinars
Loading...