TIM-3 (T cell immunoglobulin and mucin domain-3) is a 60 kDa member of the TIM family of immune regulating molecules. TIMs are type I transmembrane glycoproteins with one Ig-like V-type domain and a Ser/Thr-rich mucin stalk (1-3). There are three TIM genes in human and eight in mouse. Mature human TIM-3 consists of a 181 amino acid (aa) extracellular domain (ECD), a 21 aa transmembrane segment, and a 78 aa cytoplasmic tail (4). An alternately spliced isoform is truncated following a short substitution after the Ig-like domain. Within the ECD, human TIM-3 shares 58% aa sequence identity with mouse and rat TIM-3. TIM-3 is expressed on the surface of effector T cells (CD4+ Th1 and CD8+ Tc1) but not on helper T cells (CD4+ Th2 and CD8+ Tc2) (4, 5). NK cells appear to transcribe the highest amounts of Tim-3 among lymphocytes, and when Tim-3 was cross-linked with antibodies it suppressed NK cell-mediated cytotoxicity (6). In chronic inflammation, autoimmune disorders, and some cancers, TIM-3 is upregulated on several other hematopoietic cell types. It also occurs on hippocampal neurons (7-10). The Ig domain of TIM-3 interacts with a ligand on resting but not activated Th1 and Th2 cells (5, 11). The glycosylated Ig domain of TIM-3 binds cell-associated galectin-9. This induces TIM-3 Tyr phosphorylation and pro-apoptotic signaling (8, 12). TIM-3 functions as a negative regulator of Th1 cell activity. Its blockade results in increased IFN-gamma production, Th1 cell proliferation and cytotoxicity (5, 10, 11), and regulatory T cell development (5). TIM-3 inhibits the antitumor efficacy of DNA vaccines and chemotherapy
by binding to the damage-associated molecular pattern molecule, HMGB1 (13).
Human TIM‑3 Antibody
R&D Systems | Catalog # MAB23652

Key Product Details
Species Reactivity
Validated:
Cited:
Applications
Validated:
Cited:
Label
Antibody Source
Product Specifications
Immunogen
Met1-Arg200
Accession # Q8TDQ0
Specificity
Clonality
Host
Isotype
Scientific Data Images for Human TIM‑3 Antibody
Detection of Human TIM‑3 by Western Blot.
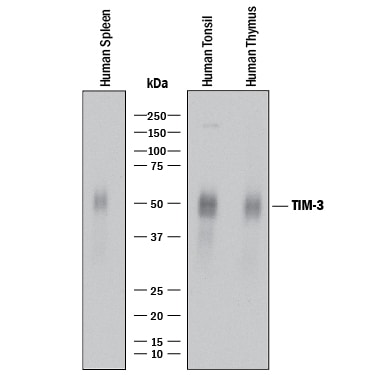
Western blot shows lysates of human spleen tissue, human tonsil tissue, and human thymus tissue. PVDF membrane was probed with 2 µg/mL of Rabbit Anti-Human TIM-3 Monoclonal Antibody (Catalog # MAB23652) followed by HRP-conjugated Anti-Rabbit IgG Secondary Antibody (Catalog # HAF008). A specific band was detected for TIM-3 at approximately 45-70 kDa (as indicated). This experiment was conducted under reducing conditions and using Immunoblot Buffer Group 1.Detection of Tim-3 in Human PBMC by Flow Cytometry.
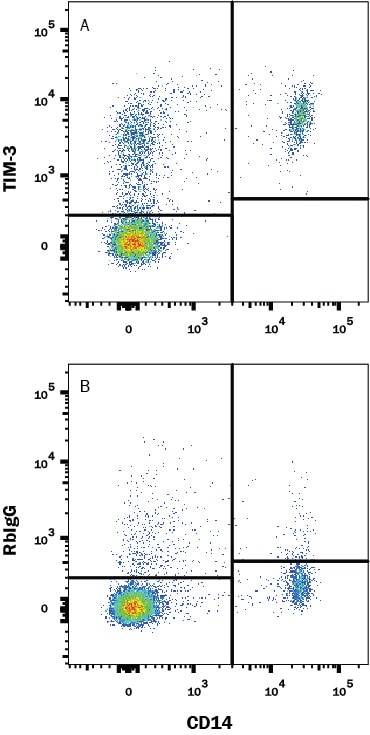
Human PBMC were stained with (A) Rabbit Anti-Human Tim-3 Monoclonal Antibody (Catalog # MAB23652) or (B) Normal Rabbit IgG Control Antibody (Catalog # MAB1050) followed by PE-conjugated Goat anti-Rabbit IgG Secondary Antibody (Catalog # F0110) and Mouse anti-Human CD14 APC-conjugated Monoclonal Antibody (Catalog # FAB3832A). View our protocol for Staining Membrane-associated Proteins.TIM‑3 in Human Tonsil.
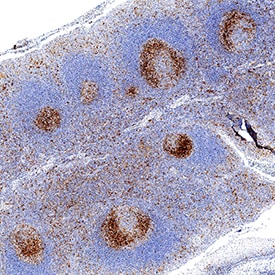
TIM-3 was detected in immersion fixed paraffin-embedded sections of human tonsil using Rabbit Anti-Human TIM-3 Monoclonal Antibody (Catalog # MAB23652) at 3 µg/mL for 1 hour at room temperature followed by incubation with the Anti-Rabbit IgG VisUCyte™ HRP Polymer Antibody (Catalog # VC003). Before incubation with the primary antibody, tissue was subjected to heat-induced epitope retrieval using Antigen Retrieval Reagent-Basic (Catalog # CTS013). Tissue was stained using DAB (brown) and counterstained with hematoxylin (blue). Specific staining was localized to cytoplasm in lymphocytes. View our protocol for IHC Staining with VisUCyte HRP Polymer Detection Reagents.Detection of TIM-3 by Western Blot
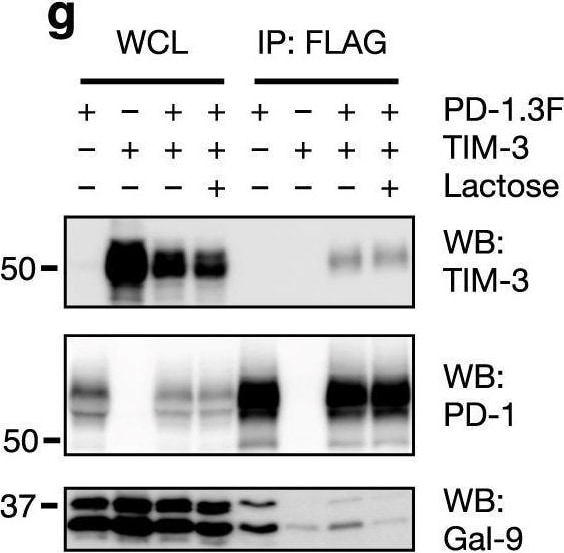
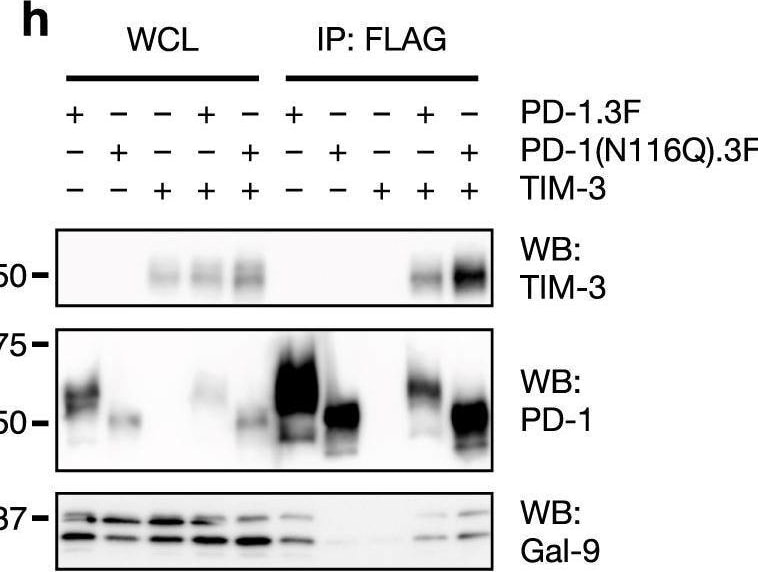
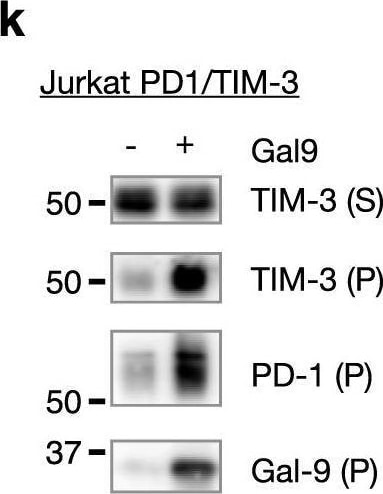
Characterization of TIM-3/Gal-9/PD-1 tri-molecular interaction.a, b TIM-3 ECD binding to plate-immobilized GST-Gal-9C (a) or GST-Gal-9N (b) in the presence of increasing concentrations of PD-1 ECD. c PD-1 ECD binding to plate-immobilized TIM-3 ECD or Gal-9. d TIM-3 ECD binding to plate-immobilized PD-1 in the presence of increasing concentrations of Gal-9. e Duolink assay of PD-1 and TIM-3 association in Gal-9 KO Jurkat cells co-expressing the two receptors with or without Gal-9. Scale bar: 10 μm. Dashed lines represent mean values; error bars represent SD. Statistical differences were assessed using unpaired two-tailed t-tests. n = 254 cells examined for each group over two independent experiments. ****P < 0.0001. f Jurkat cells expressing PD-1 (myc tagged) and TIM-3 (3xFlag tagged) individually or together were incubated with or without 2 μg/ml exogenous Gal-9 followed by IP/western blotting with indicated antibodies. g, h IP/Western analysis of Jurkat cells expressing TIM-3 and 3xFlag tagged wildtype PD-1 or PD-1(N116Q) mutant, individually or in indicated combinations, in the presence or absence of lactose. i–k Jurkat cells expressing PD-1 (i) or TIM-3 (j) or both (k) were incubated with or without Gal-9, and then lysed in a detergent buffer and centrifuged. Protein levels in the supernatants (S) and pellets (P) were determined by western blotting with the indicated antibodies. l Schematic diagram showing TIM-3/Gal-9/PD-1 tri-molecular interactions. TIM-3 and PD-1 dimerize through their intracellular domains. Gal-9 crosslinks TIM-3/PD-1 dimers with its N-CRD (green) and C-CRD (orange) to form galectin/glycoprotein lattices. Data are representative of two (a–i) or three (j, k) independent experiments. Source data are provided as a Source data file. Image collected and cropped by CiteAb from the following open publication (https://pubmed.ncbi.nlm.nih.gov/33547304), licensed under a CC-BY license. Not internally tested by R&D Systems.Detection of TIM-3 by Western Blot
Characterization of TIM-3/Gal-9/PD-1 tri-molecular interaction.a, b TIM-3 ECD binding to plate-immobilized GST-Gal-9C (a) or GST-Gal-9N (b) in the presence of increasing concentrations of PD-1 ECD. c PD-1 ECD binding to plate-immobilized TIM-3 ECD or Gal-9. d TIM-3 ECD binding to plate-immobilized PD-1 in the presence of increasing concentrations of Gal-9. e Duolink assay of PD-1 and TIM-3 association in Gal-9 KO Jurkat cells co-expressing the two receptors with or without Gal-9. Scale bar: 10 μm. Dashed lines represent mean values; error bars represent SD. Statistical differences were assessed using unpaired two-tailed t-tests. n = 254 cells examined for each group over two independent experiments. ****P < 0.0001. f Jurkat cells expressing PD-1 (myc tagged) and TIM-3 (3xFlag tagged) individually or together were incubated with or without 2 μg/ml exogenous Gal-9 followed by IP/western blotting with indicated antibodies. g, h IP/Western analysis of Jurkat cells expressing TIM-3 and 3xFlag tagged wildtype PD-1 or PD-1(N116Q) mutant, individually or in indicated combinations, in the presence or absence of lactose. i–k Jurkat cells expressing PD-1 (i) or TIM-3 (j) or both (k) were incubated with or without Gal-9, and then lysed in a detergent buffer and centrifuged. Protein levels in the supernatants (S) and pellets (P) were determined by western blotting with the indicated antibodies. l Schematic diagram showing TIM-3/Gal-9/PD-1 tri-molecular interactions. TIM-3 and PD-1 dimerize through their intracellular domains. Gal-9 crosslinks TIM-3/PD-1 dimers with its N-CRD (green) and C-CRD (orange) to form galectin/glycoprotein lattices. Data are representative of two (a–i) or three (j, k) independent experiments. Source data are provided as a Source data file. Image collected and cropped by CiteAb from the following open publication (https://pubmed.ncbi.nlm.nih.gov/33547304), licensed under a CC-BY license. Not internally tested by R&D Systems.Detection of TIM-3 by Western Blot
Characterization of TIM-3/Gal-9/PD-1 tri-molecular interaction.a, b TIM-3 ECD binding to plate-immobilized GST-Gal-9C (a) or GST-Gal-9N (b) in the presence of increasing concentrations of PD-1 ECD. c PD-1 ECD binding to plate-immobilized TIM-3 ECD or Gal-9. d TIM-3 ECD binding to plate-immobilized PD-1 in the presence of increasing concentrations of Gal-9. e Duolink assay of PD-1 and TIM-3 association in Gal-9 KO Jurkat cells co-expressing the two receptors with or without Gal-9. Scale bar: 10 μm. Dashed lines represent mean values; error bars represent SD. Statistical differences were assessed using unpaired two-tailed t-tests. n = 254 cells examined for each group over two independent experiments. ****P < 0.0001. f Jurkat cells expressing PD-1 (myc tagged) and TIM-3 (3xFlag tagged) individually or together were incubated with or without 2 μg/ml exogenous Gal-9 followed by IP/western blotting with indicated antibodies. g, h IP/Western analysis of Jurkat cells expressing TIM-3 and 3xFlag tagged wildtype PD-1 or PD-1(N116Q) mutant, individually or in indicated combinations, in the presence or absence of lactose. i–k Jurkat cells expressing PD-1 (i) or TIM-3 (j) or both (k) were incubated with or without Gal-9, and then lysed in a detergent buffer and centrifuged. Protein levels in the supernatants (S) and pellets (P) were determined by western blotting with the indicated antibodies. l Schematic diagram showing TIM-3/Gal-9/PD-1 tri-molecular interactions. TIM-3 and PD-1 dimerize through their intracellular domains. Gal-9 crosslinks TIM-3/PD-1 dimers with its N-CRD (green) and C-CRD (orange) to form galectin/glycoprotein lattices. Data are representative of two (a–i) or three (j, k) independent experiments. Source data are provided as a Source data file. Image collected and cropped by CiteAb from the following open publication (https://pubmed.ncbi.nlm.nih.gov/33547304), licensed under a CC-BY license. Not internally tested by R&D Systems.Applications for Human TIM‑3 Antibody
CyTOF-ready
Flow Cytometry
Sample: Human PBMC
Immunohistochemistry
Sample: Immersion fixed paraffin-embedded sections of human tonsil
Western Blot
Sample: Human spleen tissue, Human tonsil tissue, and Human thymus tissue
Flow Cytometry Panel Builder
Bio-Techne Knows Flow Cytometry
Save time and reduce costly mistakes by quickly finding compatible reagents using the Panel Builder Tool.
Advanced Features
- Spectra Viewer - Custom analysis of spectra from multiple fluorochromes
- Spillover Popups - Visualize the spectra of individual fluorochromes
- Antigen Density Selector - Match fluorochrome brightness with antigen density
Formulation, Preparation, and Storage
Purification
Reconstitution
Reconstitute at 0.5 mg/mL in sterile PBS. For liquid material, refer to CoA for concentration.
Formulation
Shipping
Stability & Storage
- 12 months from date of receipt, -20 to -70 °C as supplied.
- 1 month, 2 to 8 °C under sterile conditions after reconstitution.
- 6 months, -20 to -70 °C under sterile conditions after reconstitution.
Calculators
Background: TIM-3
References
- Anderson, A.C. and D.E. Anderson (2006) Curr. Opin. Immunol. 18:665.
- Mariat, C. et al. (2005) Phil. Trans. R. Soc. B. 360:1681.
- Meyers, J.H. et al. (2005) Trends Mol. Med. 11:362.
- Monney, L. et al. (2002) Nature 415:536.
- Sanchez-Fueyo, A. et al. (2003) Nat. Immunol. 4:1093.
- Ndhlovu, L. et al. (2012) Blood 119:3734.
- Wiener, Z. et al. (2007) J. Invest. Dermatol. 127:906.
- van de Weyer, P.S. et al. (2006) Biochem. Biophys. Res. Commun. 351:571.
- Gielen, A.W. et al. (2005) J. Neuroimmunol. 164:93.
- Oikawa, T. et al. (2006) J. Immunol. 177:4281.
- Sabatos, C.A. et al. (2003) Nat. Immunol. 4:1102.
- Zhu, C. et al. (2005) Nat. Immunol. 6:1245.
- Chiba, S. et al. (2012) Nat. Immunol. 13:832.
Long Name
Alternate Names
Entrez Gene IDs
Gene Symbol
UniProt
Additional TIM-3 Products
Product Documents for Human TIM‑3 Antibody
Certificate of Analysis
To download a Certificate of Analysis, please enter a lot or batch number in the search box below.
Note: Certificate of Analysis not available for kit components.
Product Specific Notices for Human TIM‑3 Antibody
For research use only
Citations for Human TIM‑3 Antibody
Customer Reviews for Human TIM‑3 Antibody
There are currently no reviews for this product. Be the first to review Human TIM‑3 Antibody and earn rewards!
Have you used Human TIM‑3 Antibody?
Submit a review and receive an Amazon gift card!
$25/€18/£15/$25CAN/¥2500 Yen for a review with an image
$10/€7/£6/$10CAN/¥1110 Yen for a review without an image
Submit a review
Protocols
Find general support by application which include: protocols, troubleshooting, illustrated assays, videos and webinars.
- 7-Amino Actinomycin D (7-AAD) Cell Viability Flow Cytometry Protocol
- Antigen Retrieval Protocol (PIER)
- Antigen Retrieval for Frozen Sections Protocol
- Appropriate Fixation of IHC/ICC Samples
- Cellular Response to Hypoxia Protocols
- Chromogenic IHC Staining of Formalin-Fixed Paraffin-Embedded (FFPE) Tissue Protocol
- Chromogenic Immunohistochemistry Staining of Frozen Tissue
- ClariTSA™ Fluorophore Kits
- Detection & Visualization of Antibody Binding
- Extracellular Membrane Flow Cytometry Protocol
- Flow Cytometry Protocol for Cell Surface Markers
- Flow Cytometry Protocol for Staining Membrane Associated Proteins
- Flow Cytometry Staining Protocols
- Flow Cytometry Troubleshooting Guide
- Fluorescent IHC Staining of Frozen Tissue Protocol
- Graphic Protocol for Heat-induced Epitope Retrieval
- Graphic Protocol for the Preparation and Fluorescent IHC Staining of Frozen Tissue Sections
- Graphic Protocol for the Preparation and Fluorescent IHC Staining of Paraffin-embedded Tissue Sections
- Graphic Protocol for the Preparation of Gelatin-coated Slides for Histological Tissue Sections
- IHC Sample Preparation (Frozen sections vs Paraffin)
- Immunofluorescent IHC Staining of Formalin-Fixed Paraffin-Embedded (FFPE) Tissue Protocol
- Immunohistochemistry (IHC) and Immunocytochemistry (ICC) Protocols
- Immunohistochemistry Frozen Troubleshooting
- Immunohistochemistry Paraffin Troubleshooting
- Intracellular Flow Cytometry Protocol Using Alcohol (Methanol)
- Intracellular Flow Cytometry Protocol Using Detergents
- Intracellular Nuclear Staining Flow Cytometry Protocol Using Detergents
- Intracellular Staining Flow Cytometry Protocol Using Alcohol Permeabilization
- Intracellular Staining Flow Cytometry Protocol Using Detergents to Permeabilize Cells
- Preparing Samples for IHC/ICC Experiments
- Preventing Non-Specific Staining (Non-Specific Binding)
- Primary Antibody Selection & Optimization
- Propidium Iodide Cell Viability Flow Cytometry Protocol
- Protocol for Heat-Induced Epitope Retrieval (HIER)
- Protocol for Liperfluo
- Protocol for Making a 4% Formaldehyde Solution in PBS
- Protocol for VisUCyte™ HRP Polymer Detection Reagent
- Protocol for the Characterization of Human Th22 Cells
- Protocol for the Characterization of Human Th9 Cells
- Protocol for the Preparation & Fixation of Cells on Coverslips
- Protocol for the Preparation and Chromogenic IHC Staining of Frozen Tissue Sections
- Protocol for the Preparation and Chromogenic IHC Staining of Frozen Tissue Sections - Graphic
- Protocol for the Preparation and Chromogenic IHC Staining of Paraffin-embedded Tissue Sections
- Protocol for the Preparation and Chromogenic IHC Staining of Paraffin-embedded Tissue Sections - Graphic
- Protocol for the Preparation and Fluorescent IHC Staining of Frozen Tissue Sections
- Protocol for the Preparation and Fluorescent IHC Staining of Paraffin-embedded Tissue Sections
- Protocol for the Preparation of Gelatin-coated Slides for Histological Tissue Sections
- Protocol: Annexin V and PI Staining by Flow Cytometry
- Protocol: Annexin V and PI Staining for Apoptosis by Flow Cytometry
- R&D Systems Quality Control Western Blot Protocol
- TUNEL and Active Caspase-3 Detection by IHC/ICC Protocol
- The Importance of IHC/ICC Controls
- Troubleshooting Guide: Fluorokine Flow Cytometry Kits
- Troubleshooting Guide: Immunohistochemistry
- Troubleshooting Guide: Western Blot Figures
- Western Blot Conditions
- Western Blot Protocol
- Western Blot Protocol for Cell Lysates
- Western Blot Troubleshooting
- Western Blot Troubleshooting Guide
- View all Protocols, Troubleshooting, Illustrated assays and Webinars