Erythropoietin (Epo), a glycoprotein produced primarily by the kidney, is the principal factor that regulates erythropoiesis by stimulating the proliferation and differentiation of erythroid progenitor cells. The biological effects of Epo are mediated by the erythropoietin receptor (Epo R). A member of the hematopoietic growth factor receptor superfamily which includes IL-2, IL-3, IL-4, IL-5, IL-6, IL-7, GM-CSF, G-CSF, thrombopoietin, LIF, CNTF, growth hormone, and prolactin, Epo R is expressed not only by erythroid cells but also by embryonic stem cells, endothelial cells, and neural cells (1). Mouse Epo R cDNA encode a type I membrane‑spanning protein with 507 amino acid (aa) residues. Mouse Epo R has a 24 aa hydrophobic signal peptide, a 225 aa extracellular domain, a 22 aa transmembrane domain, and a 236 aa intracellular domain. At the protein sequence level, the human Epo R is approximately 82% identical to the mouse protein (2). Mouse and human Epo R both contain 11 cysteine residues and an N-linked glycosylation site. Mouse Epo R, however, contains two disulfide bridges not found with human Epo R. In common with other hematopoietic growth factor receptor superfamily members, mouse Epo R has 4 positionally conserved cysteines in its extracellular domain, a tryptophan-serine-X-tryptophan-serine (WSXWS) motif or its homolog located near the transmembrane region, and lacks kinase motifs in its intracellular domain. Based on its amino acid composition the molecular weight of Epo R would be 55 kDa but after post translational modification including glycosylation and tyrosine and serine‑threonine phosphorylation the molecular weight can be as high as 78 kDa (1). As a result of alternative splicing of the Epo R gene, cDNA clones encoding a truncated form of the Epo R as well as a soluble form of Epo R has been found (2, 3). The presence of a soluble form of the Epo R has also been detected in human sera. Recombinant soluble Epo R binds Epo with high affinity and is a potent Epo antagonist (3).

Key Product Details
Species Reactivity
Validated:
Mouse
Cited:
Mouse
Applications
Validated:
Western Blot, Immunocytochemistry
Cited:
Immunohistochemistry, Immunohistochemistry-Frozen, Immunocytochemistry
Label
Unconjugated
Antibody Source
Polyclonal Goat IgG
Loading...
Product Specifications
Immunogen
Mouse myeloma cell line NS0-derived recombinant mouse Erythropoietin R
Ala25-Pro249
Accession # P14753
Ala25-Pro249
Accession # P14753
Specificity
Detects mouse Erythropoietin R in direct ELISAs and Western blots. In direct ELISAs, approximately 50% cross-reactivity with recombinant human Erythropoietin R is observed.
Clonality
Polyclonal
Host
Goat
Isotype
IgG
Scientific Data Images for Mouse Erythropoietin R Antibody
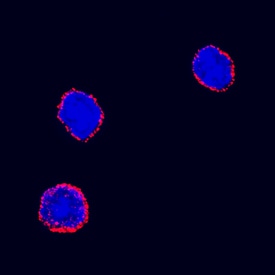
Erythropoietin R in Mouse Bone Marrow Cells.
Erythropoietin R was detected in immersion fixed mouse bone marrow cells using Goat Anti-Mouse Erythropoietin R Antigen Affinity-purified Polyclonal Antibody (Catalog # AF1390) at 15 µg/mL for 3 hours at room temperature. Cells were stained using the NorthernLights™ 557-conjugated Anti-Goat IgG Secondary Antibody (red; Catalog # NL001) and counterstained with DAPI (blue). Specific staining was localized to cell surfaces. View our protocol for Fluorescent ICC Staining of Non-adherent Cells.Detection of Erythropoietin R by Immunocytochemistry/ Immunofluorescence
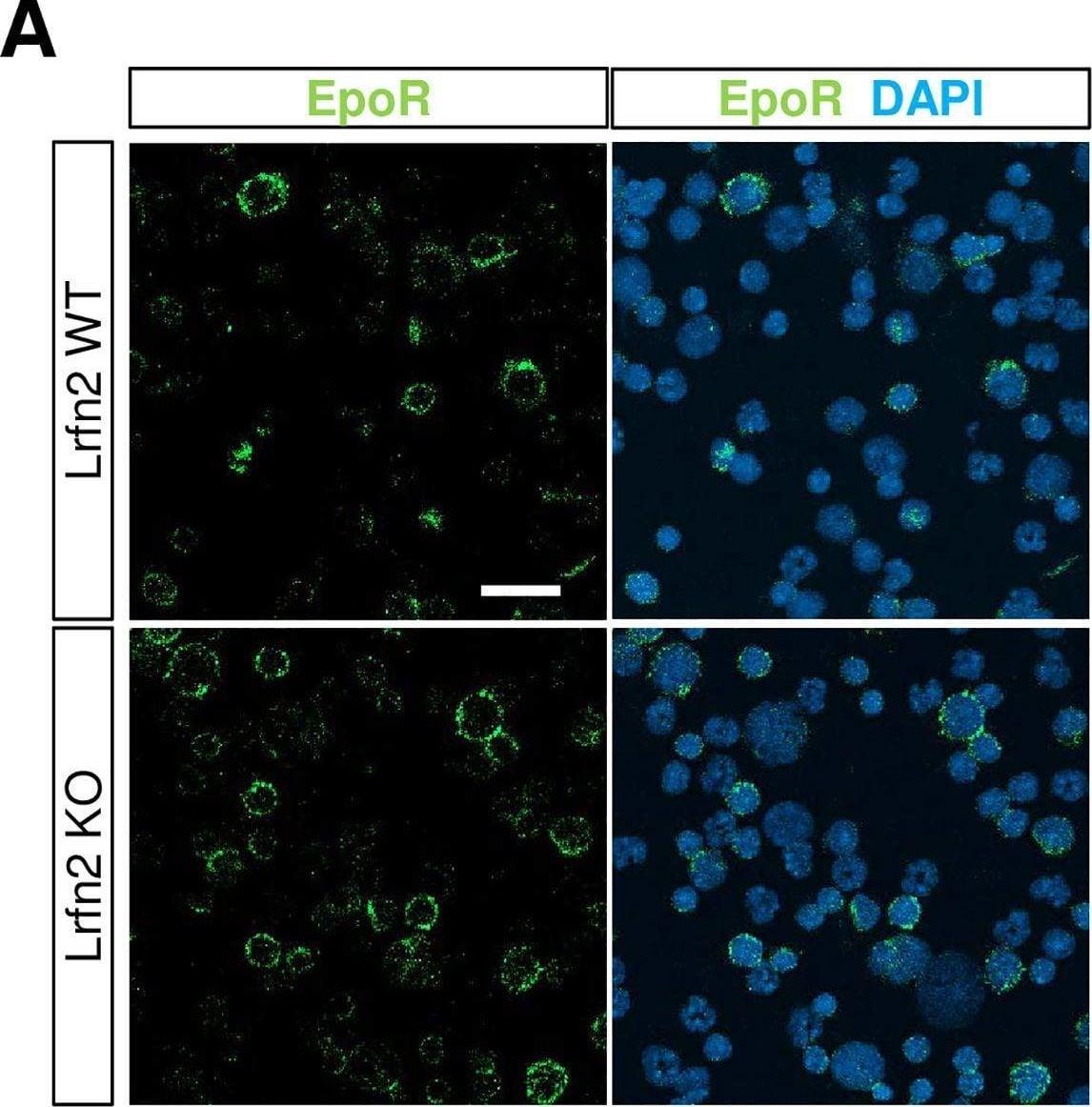
Immunostaining of EPO receptor (EpoR).BM cells were immunostained with anti-EpoR antibody (green). Nuclei were stained with DAPI (blue). (A) Representative images. Scale bar, 20 μm. (B) Quantification of the EpoR-immunopositive signals (WT, n = 6 mice; KO, n = 5 mice). Open bar, WT; closed bar, KO; error bar, SD. Each value from a mouse is indicated by circles. P values were obtained by two tailed t-tests between WT and KO. Image collected and cropped by CiteAb from the following open publication (https://pubmed.ncbi.nlm.nih.gov/33481887), licensed under a CC-BY license. Not internally tested by R&D Systems.Applications for Mouse Erythropoietin R Antibody
Application
Recommended Usage
Immunocytochemistry
5-15 µg/mL
Sample: Immersion fixed mouse bone marrow cells
Sample: Immersion fixed mouse bone marrow cells
Western Blot
0.1 µg/mL
Sample: Recombinant Mouse Erythropoietin R Fc Chimera (Catalog # 1390-ER)
Sample: Recombinant Mouse Erythropoietin R Fc Chimera (Catalog # 1390-ER)
Reviewed Applications
Read 1 review rated 4 using AF1390 in the following applications:
Formulation, Preparation, and Storage
Purification
Antigen Affinity-purified
Reconstitution
Reconstitute at 0.2 mg/mL in sterile PBS. For liquid material, refer to CoA for concentration.
Loading...
Formulation
Lyophilized from a 0.2 μm filtered solution in PBS with Trehalose. *Small pack size (SP) is supplied either lyophilized or as a 0.2 µm filtered solution in PBS.
Shipping
Lyophilized product is shipped at ambient temperature. Liquid small pack size (-SP) is shipped with polar packs. Upon receipt, store immediately at the temperature recommended below.
Stability & Storage
Use a manual defrost freezer and avoid repeated freeze-thaw cycles.
- 12 months from date of receipt, -20 to -70 °C as supplied.
- 1 month, 2 to 8 °C under sterile conditions after reconstitution.
- 6 months, -20 to -70 °C under sterile conditions after reconstitution.
Calculators
Background: Erythropoietin R
References
- Spivak, J.L. (2001) in Cytokine Reference, Oppenhiem, J.J. and M. Feldmann, eds. Academic Press, New York, p. 941.
- Kuramochi, S., Y. Ikawa and K. Todokoro (1990) J. Mol. Biol. 216:567.
- Baynes, R.D. et al. (1993) Blood 82:2088.
Long Name
Erythropoietin Receptor
Alternate Names
EpoR
Gene Symbol
EPOR
UniProt
Additional Erythropoietin R Products
Product Documents for Mouse Erythropoietin R Antibody
Certificate of Analysis
To download a Certificate of Analysis, please enter a lot or batch number in the search box below.
Note: Certificate of Analysis not available for kit components.
Product Specific Notices for Mouse Erythropoietin R Antibody
For research use only
Related Research Areas
Citations for Mouse Erythropoietin R Antibody
Customer Reviews for Mouse Erythropoietin R Antibody (1)
4 out of 5
1 Customer Rating
Have you used Mouse Erythropoietin R Antibody?
Submit a review and receive an Amazon gift card!
$25/€18/£15/$25CAN/¥2500 Yen for a review with an image
$10/€7/£6/$10CAN/¥1110 Yen for a review without an image
Submit a review
Showing
1
-
1 of
1 review
Showing All
Filter By:
-
Application: Western BlotSample Tested: See PMID 23966665Species: MouseVerified Customer | Posted 01/05/2015
There are no reviews that match your criteria.
Protocols
Find general support by application which include: protocols, troubleshooting, illustrated assays, videos and webinars.
- Appropriate Fixation of IHC/ICC Samples
- Cellular Response to Hypoxia Protocols
- ClariTSA™ Fluorophore Kits
- Detection & Visualization of Antibody Binding
- ICC Cell Smear Protocol for Suspension Cells
- ICC Immunocytochemistry Protocol Videos
- ICC for Adherent Cells
- Immunocytochemistry (ICC) Protocol
- Immunocytochemistry Troubleshooting
- Immunofluorescence of Organoids Embedded in Cultrex Basement Membrane Extract
- Immunohistochemistry (IHC) and Immunocytochemistry (ICC) Protocols
- Preparing Samples for IHC/ICC Experiments
- Preventing Non-Specific Staining (Non-Specific Binding)
- Primary Antibody Selection & Optimization
- Protocol for VisUCyte™ HRP Polymer Detection Reagent
- Protocol for the Fluorescent ICC Staining of Cell Smears - Graphic
- Protocol for the Fluorescent ICC Staining of Cultured Cells on Coverslips - Graphic
- Protocol for the Preparation and Fluorescent ICC Staining of Cells on Coverslips
- Protocol for the Preparation and Fluorescent ICC Staining of Non-adherent Cells
- Protocol for the Preparation and Fluorescent ICC Staining of Stem Cells on Coverslips
- Protocol for the Preparation of a Cell Smear for Non-adherent Cell ICC - Graphic
- R&D Systems Quality Control Western Blot Protocol
- TUNEL and Active Caspase-3 Detection by IHC/ICC Protocol
- The Importance of IHC/ICC Controls
- Troubleshooting Guide: Western Blot Figures
- Western Blot Conditions
- Western Blot Protocol
- Western Blot Protocol for Cell Lysates
- Western Blot Troubleshooting
- Western Blot Troubleshooting Guide
- View all Protocols, Troubleshooting, Illustrated assays and Webinars
Loading...
Associated Pathways