Mouse IL-17RD/SEF Antibody
R&D Systems | Catalog # AF2276

Key Product Details
Species Reactivity
Validated:
Cited:
Applications
Validated:
Cited:
Label
Antibody Source
Product Specifications
Immunogen
Gly28-Arg299
Accession # Q8JZL1
Specificity
Clonality
Host
Isotype
Scientific Data Images for Mouse IL-17RD/SEF Antibody
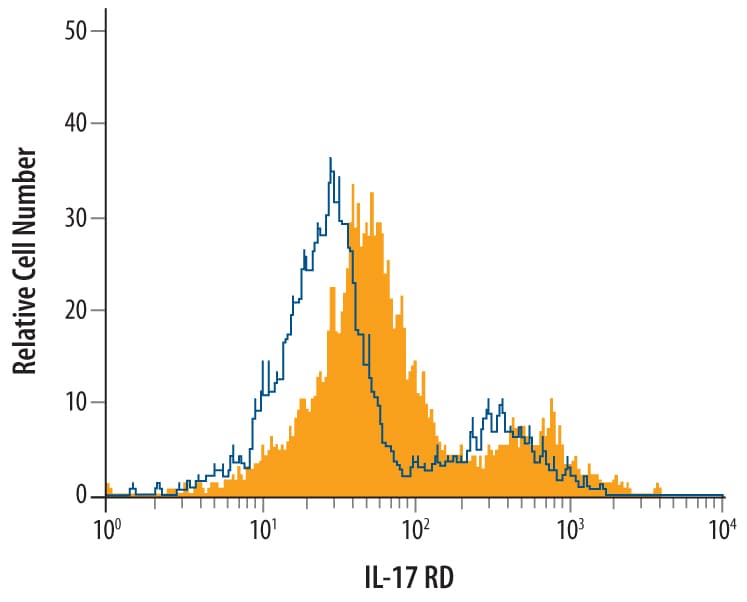
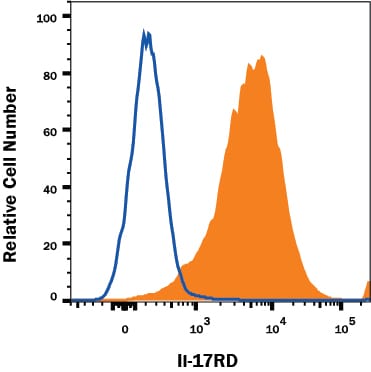
Detection of IL‑17 RD/SEF in HEK293 Human Cell Line transfected with Mouse IL-17 RD/SEF by Flow Cytometry.
HEK293 human cell line transfected with mouse IL-17 RD/SEF was stained with Goat Anti-MouseIL-17 RD/SEF Antigen Affinity-purified Polyclonal Antibody (Catalog # AF2276, filled histogram) or isotype control antibody (Catalog # AB-108-C, open histogram), followed by Phycoerythrin-conjugated Anti-Goat IgG Secondary Antibody (Catalog # F0107). View our protocol for Staining Membrane-associated Proteins.Detection of IL‑17 RD/SEF in bEnd.3 Mouse Cell Line by Flow Cytometry.
bEnd.3 mouse endothelioma cell line was stained with Goat Anti-Mouse IL-17 RD/SEF Antigen Affinity-purified Polyclonal Antibody (Catalog # AF2276, filled histogram) or isotype control antibody (Catalog # AB-108-C, open histogram), followed by Phycoerythrin-conjugated Anti-Goat IgG Secondary Antibody (Catalog # F0107).Applications for Mouse IL-17RD/SEF Antibody
CyTOF-ready
Flow Cytometry
Sample: bEnd.3 mouse endothelioma cell line and HEK293 human cell line transfected with mouse IL-17 RD/SEF
Immunohistochemistry
Sample: Perfusion fixed frozen sections of mouse lung and thymus
Western Blot
Sample: Recombinant Mouse IL‑17 RD/SEF (Catalog # 2276-ML)
Flow Cytometry Panel Builder
Bio-Techne Knows Flow Cytometry
Save time and reduce costly mistakes by quickly finding compatible reagents using the Panel Builder Tool.
Advanced Features
- Spectra Viewer - Custom analysis of spectra from multiple fluorochromes
- Spillover Popups - Visualize the spectra of individual fluorochromes
- Antigen Density Selector - Match fluorochrome brightness with antigen density
Formulation, Preparation, and Storage
Purification
Reconstitution
Reconstitute at 0.2 mg/mL in sterile PBS. For liquid material, refer to CoA for concentration.
Formulation
Shipping
Stability & Storage
- 12 months from date of receipt, -20 to -70 °C as supplied.
- 1 month, 2 to 8 °C under sterile conditions after reconstitution.
- 6 months, -20 to -70 °C under sterile conditions after reconstitution.
Calculators
Background: IL-17RD/SEF
References
- Furthauer, M. et al. (2002) Nat. Cell Biol. 4:170.
- Xiong, S. et al. (2003) J. Biol. Chem. 278:50273.
- Yang, R-B. et al. (2003) J. Biol. Chem. 278:33232.
- Preger, E. et al. (2003) Proc. Natl. Acad. Sci. USA 101:1229.
- Lin, W. et al. (2002) Mech. Dev. 113:163.
- Tsang, M. et al. (2002) Nat. Cell Biol. 4:165.
- Kovalenko, D. et al. (2003) J. Biol. Chem. 278:14087.
- Torii, S. et al. (2004) Dev. Cell 7:33.
- Yang, X. et al. (2004) J. Biol. Chem. 279:38099.
Long Name
Alternate Names
Gene Symbol
UniProt
Additional IL-17RD/SEF Products
Product Documents for Mouse IL-17RD/SEF Antibody
Certificate of Analysis
To download a Certificate of Analysis, please enter a lot or batch number in the search box below.
Note: Certificate of Analysis not available for kit components.
Product Specific Notices for Mouse IL-17RD/SEF Antibody
For research use only
Citations for Mouse IL-17RD/SEF Antibody
Customer Reviews for Mouse IL-17RD/SEF Antibody
There are currently no reviews for this product. Be the first to review Mouse IL-17RD/SEF Antibody and earn rewards!
Have you used Mouse IL-17RD/SEF Antibody?
Submit a review and receive an Amazon gift card!
$25/€18/£15/$25CAN/¥2500 Yen for a review with an image
$10/€7/£6/$10CAN/¥1110 Yen for a review without an image
Submit a review
Protocols
Find general support by application which include: protocols, troubleshooting, illustrated assays, videos and webinars.
- 7-Amino Actinomycin D (7-AAD) Cell Viability Flow Cytometry Protocol
- Antigen Retrieval Protocol (PIER)
- Antigen Retrieval for Frozen Sections Protocol
- Appropriate Fixation of IHC/ICC Samples
- Cellular Response to Hypoxia Protocols
- Chromogenic IHC Staining of Formalin-Fixed Paraffin-Embedded (FFPE) Tissue Protocol
- Chromogenic Immunohistochemistry Staining of Frozen Tissue
- ClariTSA™ Fluorophore Kits
- Detection & Visualization of Antibody Binding
- Extracellular Membrane Flow Cytometry Protocol
- Flow Cytometry Protocol for Cell Surface Markers
- Flow Cytometry Protocol for Staining Membrane Associated Proteins
- Flow Cytometry Staining Protocols
- Flow Cytometry Troubleshooting Guide
- Fluorescent IHC Staining of Frozen Tissue Protocol
- Graphic Protocol for Heat-induced Epitope Retrieval
- Graphic Protocol for the Preparation and Fluorescent IHC Staining of Frozen Tissue Sections
- Graphic Protocol for the Preparation and Fluorescent IHC Staining of Paraffin-embedded Tissue Sections
- Graphic Protocol for the Preparation of Gelatin-coated Slides for Histological Tissue Sections
- IHC Sample Preparation (Frozen sections vs Paraffin)
- Immunofluorescent IHC Staining of Formalin-Fixed Paraffin-Embedded (FFPE) Tissue Protocol
- Immunohistochemistry (IHC) and Immunocytochemistry (ICC) Protocols
- Immunohistochemistry Frozen Troubleshooting
- Immunohistochemistry Paraffin Troubleshooting
- Intracellular Flow Cytometry Protocol Using Alcohol (Methanol)
- Intracellular Flow Cytometry Protocol Using Detergents
- Intracellular Nuclear Staining Flow Cytometry Protocol Using Detergents
- Intracellular Staining Flow Cytometry Protocol Using Alcohol Permeabilization
- Intracellular Staining Flow Cytometry Protocol Using Detergents to Permeabilize Cells
- Preparing Samples for IHC/ICC Experiments
- Preventing Non-Specific Staining (Non-Specific Binding)
- Primary Antibody Selection & Optimization
- Propidium Iodide Cell Viability Flow Cytometry Protocol
- Protocol for Heat-Induced Epitope Retrieval (HIER)
- Protocol for Liperfluo
- Protocol for Making a 4% Formaldehyde Solution in PBS
- Protocol for VisUCyte™ HRP Polymer Detection Reagent
- Protocol for the Characterization of Human Th22 Cells
- Protocol for the Characterization of Human Th9 Cells
- Protocol for the Preparation & Fixation of Cells on Coverslips
- Protocol for the Preparation and Chromogenic IHC Staining of Frozen Tissue Sections
- Protocol for the Preparation and Chromogenic IHC Staining of Frozen Tissue Sections - Graphic
- Protocol for the Preparation and Chromogenic IHC Staining of Paraffin-embedded Tissue Sections
- Protocol for the Preparation and Chromogenic IHC Staining of Paraffin-embedded Tissue Sections - Graphic
- Protocol for the Preparation and Fluorescent IHC Staining of Frozen Tissue Sections
- Protocol for the Preparation and Fluorescent IHC Staining of Paraffin-embedded Tissue Sections
- Protocol for the Preparation of Gelatin-coated Slides for Histological Tissue Sections
- Protocol: Annexin V and PI Staining by Flow Cytometry
- Protocol: Annexin V and PI Staining for Apoptosis by Flow Cytometry
- R&D Systems Quality Control Western Blot Protocol
- TUNEL and Active Caspase-3 Detection by IHC/ICC Protocol
- The Importance of IHC/ICC Controls
- Troubleshooting Guide: Fluorokine Flow Cytometry Kits
- Troubleshooting Guide: Immunohistochemistry
- Troubleshooting Guide: Western Blot Figures
- Western Blot Conditions
- Western Blot Protocol
- Western Blot Protocol for Cell Lysates
- Western Blot Troubleshooting
- Western Blot Troubleshooting Guide
- View all Protocols, Troubleshooting, Illustrated assays and Webinars
Associated Pathways
 MAPK Signaling Pathway: Mitogen Stimulation Pathway
MAPK Signaling Pathway: Mitogen Stimulation Pathway