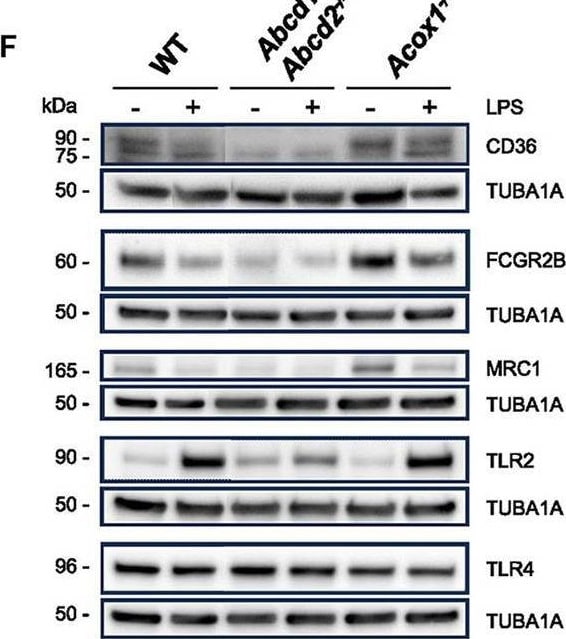
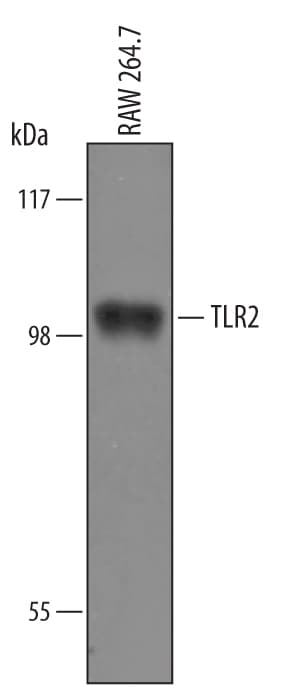
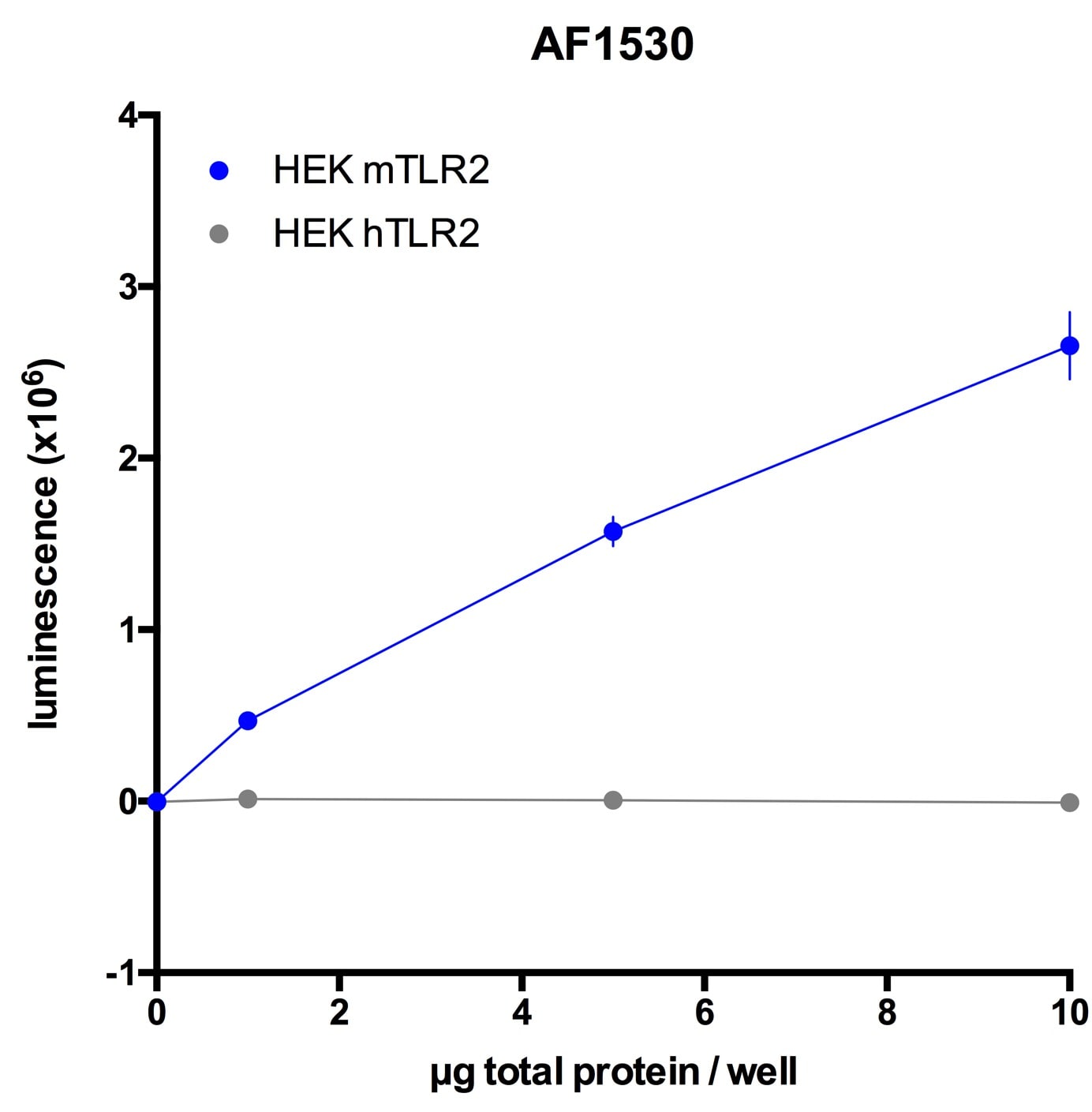
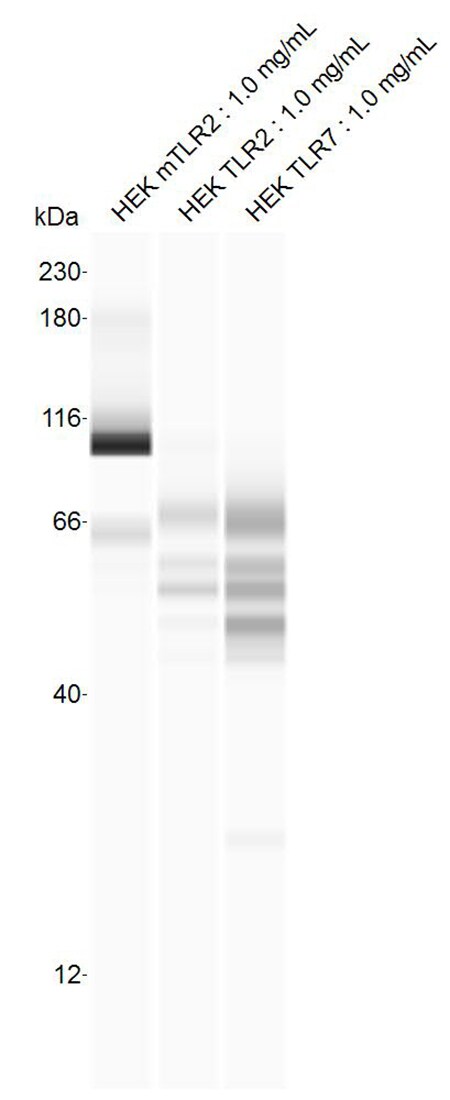
The Toll-like family of molecules are a group of integral membrane proteins that serve as pattern recognition receptors for microbial pathogens (1‑4). To date, there are at least eleven mouse and ten human members that activate the innate immune system following exposure to a variety of microbial species (1, 3). All Toll-like receptors (TLRs) are type I transmembrane (TM) proteins that exist either in the plasma membrane or in the membranes of endosomal structures (where they bind intracellular nucleic acids) (3). All TLRs also contain a large number of extracellular leucine-rich repeats (LRRs) and a cytoplasmic tail with a Toll/IL-1 receptor (TIR) domain. Mouse Toll-like receptor-2 (TLR2) is a 97 kDa, 760 amino acid (aa) glycoprotein that contains a 563 aa extracellular region, a 21 aa TM segment, and a 176 aa cytoplasmic domain (5, 6). The extracellular region contains 16 leucine-rich repeats, while the cytoplasmic tail shows one 146 aa TIR domain. The receptor is expressed on a number of cell types including T cells ( alpha beta and gamma delta ), monocytes, dendritic cells, neutrophils, B cells, endothelial cells, mast cells, NK cells, macrophages and hepatocytes (1, 4, 5, 7, 8). TLR2 functions as part of a heterodimeric complex with either TLR1 or TLR6 (1, 3, 4). These complexes recognize lipoproteins and glycolipids from gram-positive and gram-negative bacteria as well as mycoplasma and yeast. TLR2/TLR1 heterodimers recognize triacylated lipopeptides from a variety of microorganisms. The TLR2/TLR6 heterodimer preferentially recognizes diacylated lipopeptides (9). Biglycan is also known to activate TLR2, but the context is unclear (8). Notably, in human, TLR2 also dimerizes with TLR10. But the TLR10 gene in mouse (but not rat) is mutationally inactive, and thus this complex is nonfunctional (10). Upon ligand recognition, TLR2 delivers an activating signal via the associated adapter molecules, MyD88 and TIRAP (1, 11). Activation via TLR2 also results in production of a number of pro-inflammatory cytokines including TNF-alpha, IL-2, IL-6, IL-12, and MIP-2 (1, 3). The extracellular region of mouse TLR2 is 89%, 67%, 81%, and 65% aa identical to the equivalent region in rat, human, hamster and canine, respectively.