C-Reactive Protein (CRP) is a member of the pentraxin family of proteins that are characterized by a cyclic pentameric structure. The rat CRP gene encodes a 230 amino acid (aa) precursor with a signal peptide of 19 aa and the mature polypeptide of 211 aa. Rat CRP shares 64% and 70% aa sequence homology with human and mouse CRP respectively. Human, mouse and rabbit CRP are non-glycosylated proteins, and the units are non-covalently linked to form the pentameter. In contrast, rat CRP is a glycoprotein and contains a covalently linked dimer in the pentameter. CRP exhibits Ca++-dependent binding to ligands. Phosphocholine (PCh), a constituent of many bacterial and fungal walls, is a principal ligand of CRP. CRP also binds to the membrane of injured cells, the membrane and nuclear components of necrotic and apoptotic cells. Upon binding with the ligands, CRP is recognized by C1q and initiates the activation of complement cascade. Ligand bound CRP also binds to Fc gamma RI and Fc gamma RIIa on phagocytes and activates phagocytotic responses. In addition to phagocytosis, CRP also induces the production of hydrogen peroxide and inflammatory cytokines, such as IL-1, IL-6 and TNF-alpha. In human and rabbits, CRP is an important acute-phase protein that plays a role in the first line of host innate defense. The level of plasma CRP at basal conditions in human and rabbits is very low, and can increase 1,000-fold within 24‑48 hours in response to infection, inflammation or tissue damage. In rats, CRP exists at a high level at basal conditions and only increases about 2-fold in response to injury. CRP is not a typical acute-phase protein in rat and is a minor component in response to injury. In mice, CRP is expressed at very low levels and is not an acute phase reactant. Serum amyloid P component (SAP), another pentraxin, is an acute phase serum protein in mice.
Rat C‑Reactive Protein/CRP Antibody
R&D Systems | Catalog # AF1744

Key Product Details
Species Reactivity
Validated:
Rat
Cited:
Rat
Applications
Validated:
Immunohistochemistry, Western Blot, Simple Western
Cited:
Western Blot
Label
Unconjugated
Antibody Source
Polyclonal Goat IgG
Loading...
Product Specifications
Immunogen
Mouse myeloma cell line NS0-derived recombinant rat C-Reactive Protein/CRP
His20-Ser230
Accession # P48199
His20-Ser230
Accession # P48199
Specificity
Detects rat C‑Reactive Protein/CRP in direct ELISAs and Western blots. In Western blots, approximately 20% cross-reactivity with recombinant mouse CRP is observed and 10% cross-reactivity with recombinant human CRP is observed.
Clonality
Polyclonal
Host
Goat
Isotype
IgG
Scientific Data Images for Rat C‑Reactive Protein/CRP Antibody
Detection of Rat C‑Reactive Protein/CRP by Western Blot.
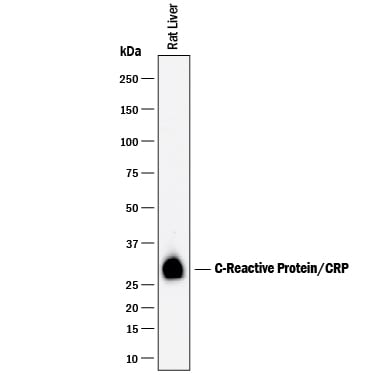
Western blot shows lysates of rat liver tissue. PVDF membrane was probed with 1 µg/mL of Goat Anti-Rat C-Reactive Protein/CRP Antigen Affinity-purified Polyclonal Antibody (Catalog # AF1744) followed by HRP-conjugated Anti-Goat IgG Secondary Antibody (Catalog # HAF017). A specific band was detected for C-Reactive Protein/CRP at approximately 28 kDa (as indicated). This experiment was conducted under reducing conditions and using Immunoblot Buffer Group 1.Detection of Rat C‑Reactive Protein/CRP by Simple WesternTM.
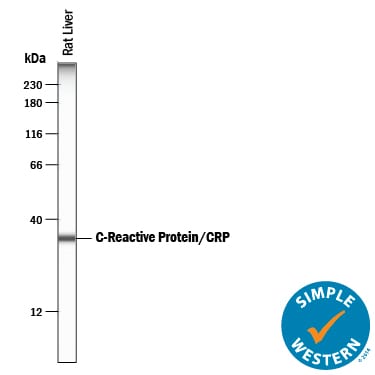
Simple Western lane view shows lysates of rat liver tissue, loaded at 0.2 mg/mL. A specific band was detected for C-Reactive Protein/CRP at approximately 34 kDa (as indicated) using 10 µg/mL of Goat Anti-Rat C-Reactive Protein/CRP Antigen Affinity-purified Polyclonal Antibody (Catalog # AF1744) followed by 1:50 dilution of HRP-conjugated Anti-Goat IgG Secondary Antibody (Catalog # HAF109). This experiment was conducted under reducing conditions and using the 12-230 kDa separation system.C-Reactive Protein/CRP in Rat Liver.
C-Reactive Protein/CRP was detected in perfusion fixed frozen sections of rat liver using Goat Anti-Rat C-Reactive Protein/CRP Antigen Affinity-purified Polyclonal Antibody (Catalog # AF1744) at 5 µg/mL overnight at 4 °C. Tissue was stained using the Anti-Goat HRP-DAB Cell & Tissue Staining Kit (brown; Catalog # CTS008) and counterstained with hematoxylin (blue). Specific labeling was localized to the cytoplasm of hepatocytes. View our protocol for Chromogenic IHC Staining of Frozen Tissue Sections.Applications for Rat C‑Reactive Protein/CRP Antibody
Application
Recommended Usage
Immunohistochemistry
5-15 µg/mL
Sample: Perfusion fixed frozen sections of rat liver
Sample: Perfusion fixed frozen sections of rat liver
Simple Western
10 µg/mL
Sample: Rat liver tissue
Sample: Rat liver tissue
Western Blot
1 µg/mL
Sample: Rat liver tissue
Sample: Rat liver tissue
Formulation, Preparation, and Storage
Purification
Antigen Affinity-purified
Reconstitution
Reconstitute at 0.2 mg/mL in sterile PBS. For liquid material, refer to CoA for concentration.
Loading...
Formulation
Lyophilized from a 0.2 μm filtered solution in PBS with Trehalose. *Small pack size (SP) is supplied either lyophilized or as a 0.2 µm filtered solution in PBS.
Shipping
Lyophilized product is shipped at ambient temperature. Liquid small pack size (-SP) is shipped with polar packs. Upon receipt, store immediately at the temperature recommended below.
Stability & Storage
Use a manual defrost freezer and avoid repeated freeze-thaw cycles.
- 12 months from date of receipt, -20 to -70 °C as supplied.
- 1 month, 2 to 8 °C under sterile conditions after reconstitution.
- 6 months, -20 to -70 °C under sterile conditions after reconstitution.
Calculators
Background: C-Reactive Protein/CRP
References
- Mohammad, R. et al. (1992) J. Biol. Chem. 267:2947.
- Sambasivam, H. et al. (1993) J. Biol. Chem. 268:10007.
- Padilla, N.D. et al. (2003) Immunology 109:564.
- Volanakis, J.E. (2001) Molecular Immunology 38:189.
Alternate Names
CRP
Gene Symbol
CRP
UniProt
Additional C-Reactive Protein/CRP Products
Product Documents for Rat C‑Reactive Protein/CRP Antibody
Certificate of Analysis
To download a Certificate of Analysis, please enter a lot or batch number in the search box below.
Note: Certificate of Analysis not available for kit components.
Product Specific Notices for Rat C‑Reactive Protein/CRP Antibody
For research use only
Related Research Areas
Citations for Rat C‑Reactive Protein/CRP Antibody
Customer Reviews for Rat C‑Reactive Protein/CRP Antibody
There are currently no reviews for this product. Be the first to review Rat C‑Reactive Protein/CRP Antibody and earn rewards!
Have you used Rat C‑Reactive Protein/CRP Antibody?
Submit a review and receive an Amazon gift card!
$25/€18/£15/$25CAN/¥2500 Yen for a review with an image
$10/€7/£6/$10CAN/¥1110 Yen for a review without an image
Submit a review
Protocols
Find general support by application which include: protocols, troubleshooting, illustrated assays, videos and webinars.
- Antigen Retrieval Protocol (PIER)
- Antigen Retrieval for Frozen Sections Protocol
- Appropriate Fixation of IHC/ICC Samples
- Cellular Response to Hypoxia Protocols
- Chromogenic IHC Staining of Formalin-Fixed Paraffin-Embedded (FFPE) Tissue Protocol
- Chromogenic Immunohistochemistry Staining of Frozen Tissue
- ClariTSA™ Fluorophore Kits
- Detection & Visualization of Antibody Binding
- Fluorescent IHC Staining of Frozen Tissue Protocol
- Graphic Protocol for Heat-induced Epitope Retrieval
- Graphic Protocol for the Preparation and Fluorescent IHC Staining of Frozen Tissue Sections
- Graphic Protocol for the Preparation and Fluorescent IHC Staining of Paraffin-embedded Tissue Sections
- Graphic Protocol for the Preparation of Gelatin-coated Slides for Histological Tissue Sections
- IHC Sample Preparation (Frozen sections vs Paraffin)
- Immunofluorescent IHC Staining of Formalin-Fixed Paraffin-Embedded (FFPE) Tissue Protocol
- Immunohistochemistry (IHC) and Immunocytochemistry (ICC) Protocols
- Immunohistochemistry Frozen Troubleshooting
- Immunohistochemistry Paraffin Troubleshooting
- Preparing Samples for IHC/ICC Experiments
- Preventing Non-Specific Staining (Non-Specific Binding)
- Primary Antibody Selection & Optimization
- Protocol for Heat-Induced Epitope Retrieval (HIER)
- Protocol for Making a 4% Formaldehyde Solution in PBS
- Protocol for VisUCyte™ HRP Polymer Detection Reagent
- Protocol for the Preparation & Fixation of Cells on Coverslips
- Protocol for the Preparation and Chromogenic IHC Staining of Frozen Tissue Sections
- Protocol for the Preparation and Chromogenic IHC Staining of Frozen Tissue Sections - Graphic
- Protocol for the Preparation and Chromogenic IHC Staining of Paraffin-embedded Tissue Sections
- Protocol for the Preparation and Chromogenic IHC Staining of Paraffin-embedded Tissue Sections - Graphic
- Protocol for the Preparation and Fluorescent IHC Staining of Frozen Tissue Sections
- Protocol for the Preparation and Fluorescent IHC Staining of Paraffin-embedded Tissue Sections
- Protocol for the Preparation of Gelatin-coated Slides for Histological Tissue Sections
- R&D Systems Quality Control Western Blot Protocol
- TUNEL and Active Caspase-3 Detection by IHC/ICC Protocol
- The Importance of IHC/ICC Controls
- Troubleshooting Guide: Immunohistochemistry
- Troubleshooting Guide: Western Blot Figures
- Western Blot Conditions
- Western Blot Protocol
- Western Blot Protocol for Cell Lysates
- Western Blot Troubleshooting
- Western Blot Troubleshooting Guide
- View all Protocols, Troubleshooting, Illustrated assays and Webinars
Loading...