Epidermal growth factor (EGF) is a small, potent growth factor capable of inducing cell proliferation, differentiation, and survival. EGF is the founding member of the EGF family that also includes TGF-alpha, amphiregulin (AR), betacellulin (BTC), epiregulin (EPR), heparin‑binding EGF‑like growth factor (HB‑EGF), epigen, and the neuregulins (NRG)-1 through -6 (1). Members of The EGF family are characterized by a shared structural motif, the EGF‑like domain, which contains three intramolecular disulfide bonds that are formed by six similarly spaced, conserved cysteine residues (2). These disulfide bonds are essential for proper protein conformation and receptor binding. All EGF family members are synthesized as type I transmembrane precursor proteins that may contain several EGF domains in the extracellular region. The mature proteins are released from the cell surface by regulated proteolysis (1). The full length EGF protein is 1207 amino acids (aa) (EGF precursor) containing nine EGF domains and nine LDLR class B repeats. However, the mature protein is much smaller, only 53 aa, and is generated by proteolytic cleavage of the EGF domain proximal to the transmembrane region (3). EGF is well conserved across mammals with mature human EGF 70% identical to mature mouse and rat EGF. Physiologically, EGF is found in various body fluids, including blood, milk, urine, saliva, seminal fluid, pancreatic juice, cerebrospinal fluid, and amniotic fluid (4). EGF is a high affinity ligand of the EGF receptor (ErbB). Four ErbB (HER) family receptor tyrosine kinases including EGFR/ErbB1, ErbB2, ErbB3 and ErbB4, mediate responses to EGF family members (5). EGF binding induces dimerization of the EGF receptor resulting in activation of the protein tyrosine kinase signaling pathway. These receptors undergo a complex pattern of ligand-induced homo- or hetero-dimerization to transduce EGF family signals (6, 7). EGF binds ErbB1 and depending on the context, induces the formation of homodimers or heterodimers containing ErbB2. Dimerization results in autophosphorylation of the receptor at specific tyrosine residues to create docking sites for a variety of signaling molecules (5, 8). Biological activities ascribed to EGF include epithelial development, angiogenesis, inhibition of gastric acid secretion, fibroblast proliferation, and colony formation of epidermal cells in culture.
GMP
Best Seller
Recombinant Human EGF GMP Protein, CF
R&D Systems | Catalog # 236-GMP
Animal-Free.

Loading...
Key Product Details
- A DMF is on file for this protein
Source
E. coli
Accession Number
Applications
Bioactivity
Loading...
Product Specifications
Source
E. coli-derived human EGF protein
Asn971-Arg1023, with an N-terminal Met
Produced using non-animal reagents in an animal-free laboratory.
Manufactured and tested under cGMP guidelines.
Asn971-Arg1023, with an N-terminal Met
Produced using non-animal reagents in an animal-free laboratory.
Manufactured and tested under cGMP guidelines.
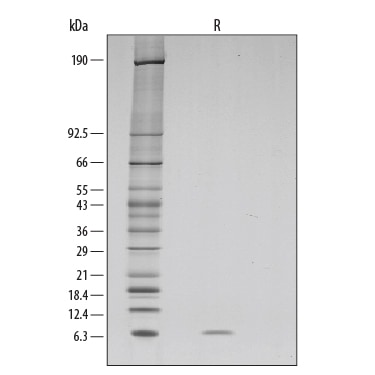
Purity
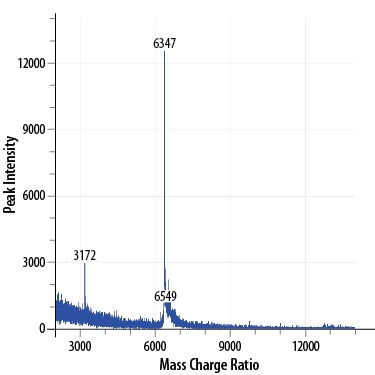
>97%, by SDS-PAGE under reducing conditions and visualized by silver stain. The result of the major peak from mass spectrometry analysis is 6347 Da, which corresponds to the calculated molecular mass of 6353 Da.
Endotoxin Level
<0.10 EU per 1 μg of the protein by the LAL method.
N-terminal Sequence Analysis
Met-Asn971-Ser-Asp-Ser-Glu-(Cys)-Pro-Leu-Ser
Predicted Molecular Mass
6 kDa
SDS-PAGE
6 kDa, reducing conditions
Activity
Measured in a cell proliferation assay using Balb/3T3 mouse embryonic fibroblast cells. Rubin, J.S. et al. (1991) Proc. Natl. Acad. Sci. USA 88:415.
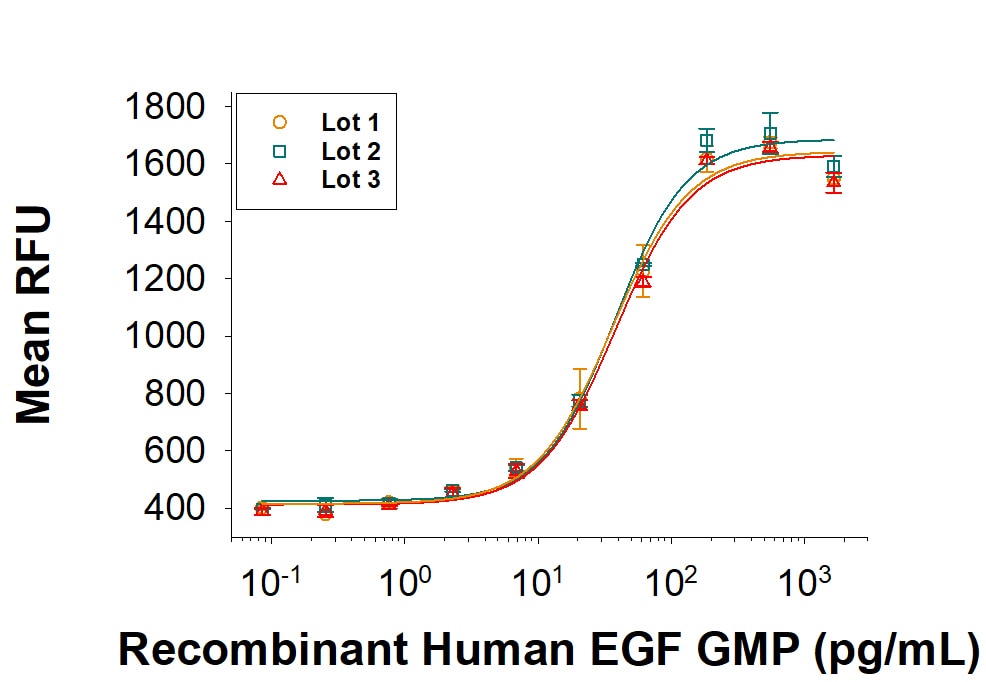
The ED50 for this effect is 20‑100 pg/mL.
The specific activity of Recombinant Human EGF is >8.0 x 105 IU/mg, which is calibrated against the human EGF WHO International Standard (NIBSC code: 91/530).
The ED50 for this effect is 20‑100 pg/mL.
The specific activity of Recombinant Human EGF is >8.0 x 105 IU/mg, which is calibrated against the human EGF WHO International Standard (NIBSC code: 91/530).
Host Cell Protein
<0.5 ng per µg of protein when tested by ELISA.
Mycoplasma
Negative for Mycoplasma.
Host Cell DNA
<0.0015 ng per µg of protein when tested by PCR.
Reviewed Applications
Read 2 reviews rated 5 using 236-GMP in the following applications:
Scientific Data Images for Recombinant Human EGF GMP Protein, CF
Recombinant Human EGF GMP Protein Bioactivity
GMP-grade Recombinant Human EGF (Catalog # 236‑GMP) stimulates proliferation of the Balb/3T3 mouse embryonic fibroblast cell line. The ED50 for this effect is 20‑100 pg/mL. Three independent lots were tested for activity and plotted on the same graph to show lot-to-lot consistency of GMP EGF.Recombinant Human EGF GMP Protein SDS-PAGE
1 µg/lane of GMP-grade Recombinant Human EGF (Catalog # 236-GMP) was resolved by SDS-PAGE and visualized by silver staining under reducing (R) conditions, showing a single band at 6 kDa.Recombinant Human EGF GMP Protein Mass Spectrometry
MALDI‑TOF analysis of GMP‑grade Recombinant Human EGF (Catalog # 236‑GMP). The major peak corresponds to the calculated molecular mass, 6353 Da. The minor peak at 6549 Da is a matrix-associated artifact of the MALDI-TOF.Formulation, Preparation, and Storage
236-GMP
| Formulation | Lyophilized from a 0.2 μm filtered solution in PBS. |
| Reconstitution | Reconstitute at 200 μg/mL in PBS. |
| Shipping | The product is shipped with polar packs. Upon receipt, store it immediately at the temperature recommended below. |
| Stability & Storage | Use a manual defrost freezer and avoid repeated freeze-thaw cycles.
|
Calculators
Background: EGF
References
- Harris, R.C. et al. (2003) Exp. Cell Res. 284:2.
- Carpenter, G. and Cohen, S. (1990) J. Biol. Chem. 265:7709.
- Bell, G.I. et al. (1986) Nucl. Acids Res. 14:8427.
- Carpenter, G. and Zendegui, J.G. (1986) Exp. Cell Res. 164:1.
- Jorissen, R.N. et al. (2003) Exp. Cell Res. 284:31.
- Gamett, D.C. et al. (1997) J. Biol. Chem. 272:12052.
- Qian, X. et al. (1994) Proc. Natl. Acad. Sci. 91:1500.
- Qian, X. et al. (1999) J. Biol. Chem. 274:574.
Long Name
Epidermal Growth Factor
Alternate Names
HOMG4, URG, Urogastrone
Gene Symbol
EGF
UniProt
Additional EGF Products
Product Documents for Recombinant Human EGF GMP Protein, CF
Certificate of Analysis
To download a Certificate of Analysis, please enter a lot or batch number in the search box below.
Note: Certificate of Analysis not available for kit components.
Manufacturing Specifications
GMP ProteinsR&D Systems, a Bio-Techne Brand's GMP proteins are produced according to relevant sections of the following documents: USP Chapter 1043, Ancillary Materials for Cell, Gene and Tissue-Engineered Products and Eu. Ph. 5.2.12, Raw Materials of Biological Origin for the Production of Cell-based and Gene Therapy Medicinal Products.
R&D Systems' quality focus includes:
- Designed, manufactured and tested under an ISO 9001:2015 and ISO 13485:2016 certified quality system
- Documented and controlled manufacturing process
- Control of documentation and process changes by QA
- Personnel training programs
- Raw material inspection and vendor qualification/monitoring program
- Validated equipment, processes and test methods
- Equipment calibration and maintenance schedules using a Regulatory Asset Manager
- Facility/Utilities maintenance, contamination controls, safety and pest control programs
- Material review process for variances
- Robust product stability program following relevant ICH guidelines
- N-terminal amino acid analysis
- SDS-PAGE purity analysis
- Molecular weight analysis via mass spectrometry
- Endotoxin assessment per USP <85> and Ph. Eur. 2.6.14 guidelines
- Bioassay analysis
- Microbial testing per USP <71> and Ph. Eur. 2.6.1 guidelines
- Host cell protein assessment
- Host cell DNA assessment
- Mycoplasma assessment
Production records and facilities are available for examination by appropriate personnel on-site at R&D Systems in Minneapolis and St. Paul, Minnesota USA.
R&D Systems sells GMP grade products for preclinical or clinical ex vivo use. They are not for in vivo use. Please read the following End User Terms prior to using this product.
Animal-Free Manufacturing Conditions
Our dedicated controlled-access animal-free laboratories ensure that at no point in production are the products exposed to potential contamination by animal components or byproducts. Every stage of manufacturing is conducted in compliance with R&D Systems' stringent Standard Operating Procedures (SOPs). Production and purification procedures use equipment and media that are confirmed animal-free.
Production
- All molecular biology procedures use animal-free media and dedicated labware.
- Dedicated fermentors are utilized in committed animal-free areas.
- Protein purification columns are animal-free.
- Bulk proteins are filtered using animal-free filters.
- Purified proteins are stored in animal-free containers.
Product Specific Notices for Recombinant Human EGF GMP Protein, CF
Full terms and conditions of sale can be found online in the Protein Sciences Segment T&Cs at: Terms & Conditions.
For preclinical, or clinical ex vivo use
Related Research Areas
Citations for Recombinant Human EGF GMP Protein, CF
Customer Reviews for Recombinant Human EGF GMP Protein, CF (2)
5 out of 5
2 Customer Ratings
Have you used Recombinant Human EGF GMP Protein, CF?
Submit a review and receive an Amazon gift card!
$25/€18/£15/$25CAN/¥2500 Yen for a review with an image
$10/€7/£6/$10CAN/¥1110 Yen for a review without an image
Submit a review
Customer Images
Showing
1
-
2 of
2 reviews
Showing All
Filter By:
-
Application: Cell ProliferationVerified Customer | Posted 08/17/2021EGF-GMP used to convert fibroblast into organoid in GMP condition.
-
Application: Cell ProliferationVerified Customer | Posted 07/21/20210.2ng/ml EGF was added to promote the growth of the limbal stem cells
There are no reviews that match your criteria.