Wnt-3a is one of 19 vertebrate members of the Wingless-type MMTV integration site (Wnt) family of highly conserved cysteine-rich secreted glycoproteins important for normal developmental processes (1). Wnts bind to the cell surface Frizzled family receptors in conjunction with low-density lipoprotein receptor-related protein family receptors (LRP5 or 6) resulting in the stabilization of intracellular beta -catenin levels (2). As intracellular beta -catenin levels rise, beta -catenin binds to TCF/LEF transcription factors leading to expression of Wnt target genes (3). Endo-IWR 1 (Catalog # 3532, # PSM1324) is a cell-permeant small molecule inhibitor of Axin turnover that suppresses Wnt signal transduction by stabilizing the beta -catenin destruction complex (4). Wnt-3a is a 44 kDa secreted hydrophobic glycoprotein containing a conserved pattern of 24 cysteine residues (5). Wnt-3a has two N-linked glycosylation sites (Asn 87, Asn 298), and Ser 209 is modified with palmitoleic acid (6). Glycosylation and acylation are essential for efficient Wnt secretion and biological activity, respectively (6, 7). Human Wnt-3a shares 96% amino acid (aa) identity with mouse, bovine and canine Wnt-3a, and 89%, 86% and 84% aa identity with chicken, Xenopus and zebrafish Wnt-3a, respectively. It also shares 87% aa identity with Wnt3. During embryonic development, Wnt-3a is necessary for proper development of the hippocampus, anterior-posterior patterning, somite development, and tailbud formation (9-12). Wnt-3a also promotes self-renewal of hematopoietic stem cells, neural stem cells, and embryonic stem cells (13, 14).
GMP
Recombinant Human Wnt-3a GMP Protein, CF
R&D Systems | Catalog # 5036-GMP

Loading...
Key Product Details
Source
CHO
Accession Number
Applications
Bioactivity
Loading...
Product Specifications
Source
Chinese Hamster Ovary cell line, CHO-derived human Wnt-3a protein
Ser19-Lys352
Manufactured and tested under cGMP guidelines.
Ser19-Lys352
Manufactured and tested under cGMP guidelines.
Purity
>75%, by SDS-PAGE with silver staining, under reducing conditions.
Endotoxin Level
<0.10 EU per 1 μg of the protein by the LAL method.
N-terminal Sequence Analysis
Ser19-Tyr-Pro-Ile-Trp-Trp-Ser-Leu-Ala-Val
Predicted Molecular Mass
37.4 kDa
SDS-PAGE
40 kDa, reducing conditions
Activity
Measured by its ability to induce alkaline phosphatase production by MC3T3‑E1 mouse preosteoblast cells.
The ED50 for this effect is ≤ 25.0 ng/mL.
Measured by its ability to induce Topflash reporter activity in HEK293T human embryonic kidney cells.
The ED50 for this effect is <500 ng/mL. Protein concentrations should be titrated based on cell type and if appropriate, passage number of the cell line.
Optimal concentrations should be determined by each laboratory for each application.
The ED50 for this effect is ≤ 25.0 ng/mL.
Measured by its ability to induce Topflash reporter activity in HEK293T human embryonic kidney cells.
The ED50 for this effect is <500 ng/mL. Protein concentrations should be titrated based on cell type and if appropriate, passage number of the cell line.
Optimal concentrations should be determined by each laboratory for each application.
Host Cell Protein
40 ng per µg of protein when tested by ELISA.
Mycoplasma
Negative for Mycoplasma.
Scientific Data Images for Recombinant Human Wnt-3a GMP Protein, CF
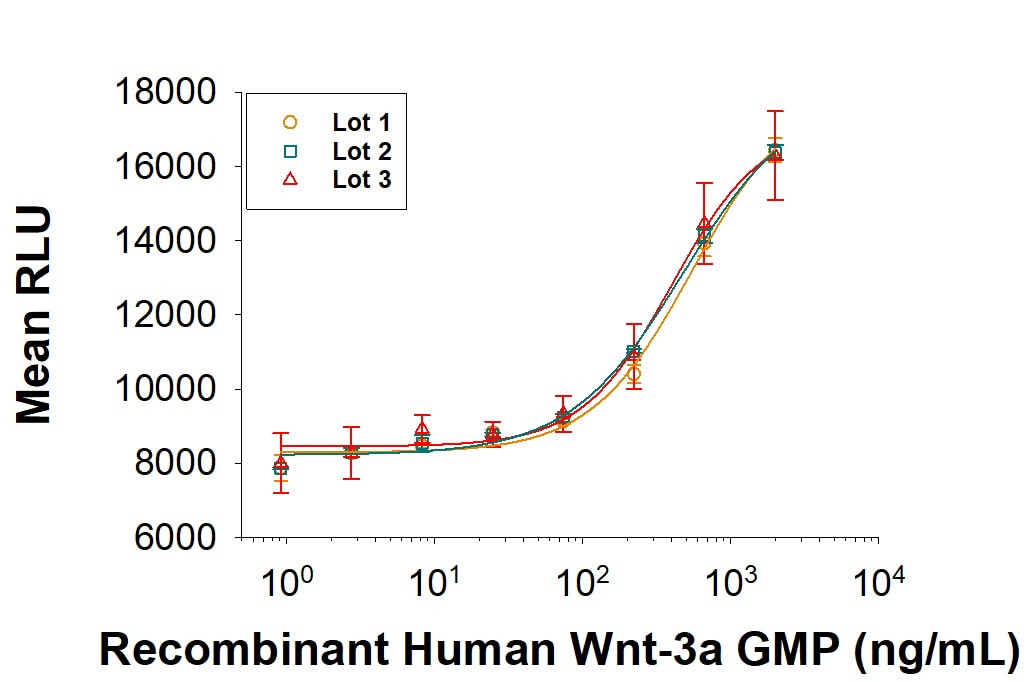
Recombinant Human Wnt-3a GMP Protein Bioactivity
GMP-grade Recombinant Human Wnt-3a (Catalog # 5036-GMP) induces alkaline phosphatase production by the MC3T3‑E1 mouse preosteoblast cell line. The ED50 for this effect is ≤ 25.0 ng/mL. Three independent lots were tested for activity and plotted on the same graph to show lot-to-lot consistency of GMP Wnt-3a.Recombinant Human Wnt-3a GMP Protein Bioactivity
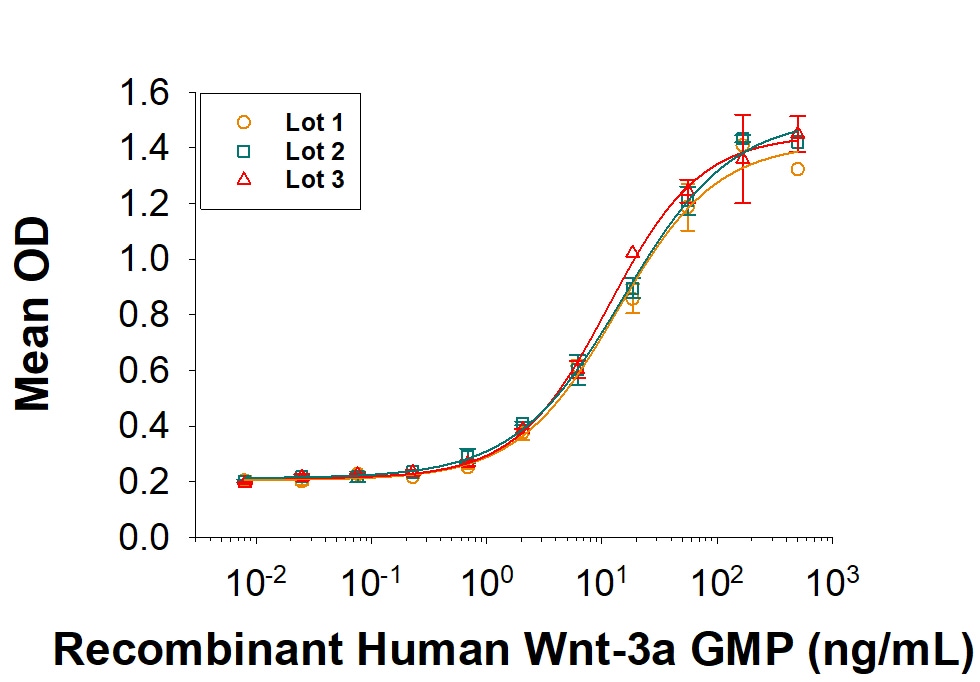
GMP-grade Recombinant Human Wnt-3a (Catalog # 5036-GMP) induces beta -catenin-responsive transcriptional activation in a Topflash Luciferase assay using the HEK293T human embryonic kidney cell line. The ED50 for this effect is <500 ng/mL. Three independent lots were tested for activity and plotted on the same graph to show lot-to-lot consistency of GMP Wnt-3a.Formulation, Preparation, and Storage
5036-GMP
| Formulation | Lyophilized from a 0.2 μm filtered solution in PBS, EDTA and CHAPS. |
| Reconstitution | Reconstitute at 200 μg/mL in PBS. |
| Shipping | The product is shipped with polar packs. Upon receipt, store it immediately at the temperature recommended below. |
| Stability & Storage | Use a manual defrost freezer and avoid repeated freeze-thaw cycles.
|
Calculators
Background: Wnt-3a
References
- Willert, K. and Nusse, R. (2012) Cold Spring Harb. Perspect. Biol. 4:a007864.
- MacDonald, B.T. and X. He (2012) Cold Spring Harb. Perspect. Biol. 4:a007880.
- Korinek, V. et al. (1997) Science 275:1784.
- Chen, B. et al. (2009) Nat. Chem. Biol. 5:100.
- Smolich, B.D. et al. (1993) Mol. Biol. Cell 4:1267.
- Takada, R. et al. (2006) Dev. Cell 11:791.
- Komekado, H. (2007) Genes Cells 12:521.
- Dunty Jr. W. C. et al. (2008) Development 135:85.
- Ikeya, M. and S. Takada (2001) Mech. Dev. 103:27.
- Lee, S. M. et al. (2000) Development 127:457.
- Takada, S. et al. (1994) Genes Dev. 8:174.
- Willert, K. et al. (2003) Nature 423:6938.
- Kalani, M.Y. et al. (2008) Proc. Natl. Acad. Sci. USA 105:16970.
- Ten Berge, D. et al. (2011) Nat. Cell Biol. 13:1070.
Long Name
Wingless-type MMTV Integration Site Family, Member 3a
Alternate Names
Wnt3a
Gene Symbol
WNT3A
UniProt
Additional Wnt-3a Products
Product Documents for Recombinant Human Wnt-3a GMP Protein, CF
Certificate of Analysis
To download a Certificate of Analysis, please enter a lot or batch number in the search box below.
Note: Certificate of Analysis not available for kit components.
Manufacturing Specifications
GMP ProteinsR&D Systems, a Bio-Techne Brand's GMP proteins are produced according to relevant sections of the following documents: USP Chapter 1043, Ancillary Materials for Cell, Gene and Tissue-Engineered Products and Eu. Ph. 5.2.12, Raw Materials of Biological Origin for the Production of Cell-based and Gene Therapy Medicinal Products.
R&D Systems' quality focus includes:
- Designed, manufactured and tested under an ISO 9001:2015 and ISO 13485:2016 certified quality system
- Documented and controlled manufacturing process
- Control of documentation and process changes by QA
- Personnel training programs
- Raw material inspection and vendor qualification/monitoring program
- Validated equipment, processes and test methods
- Equipment calibration and maintenance schedules using a Regulatory Asset Manager
- Facility/Utilities maintenance, contamination controls, safety and pest control programs
- Material review process for variances
- Robust product stability program following relevant ICH guidelines
- N-terminal amino acid analysis
- SDS-PAGE purity analysis
- Molecular weight analysis via mass spectrometry
- Endotoxin assessment per USP <85> and Ph. Eur. 2.6.14 guidelines
- Bioassay analysis
- Microbial testing per USP <71> and Ph. Eur. 2.6.1 guidelines
- Host cell protein assessment
- Host cell DNA assessment
- Mycoplasma assessment
Production records and facilities are available for examination by appropriate personnel on-site at R&D Systems in Minneapolis and St. Paul, Minnesota USA.
R&D Systems sells GMP grade products for preclinical or clinical ex vivo use. They are not for in vivo use. Please read the following End User Terms prior to using this product.
Product Specific Notices for Recombinant Human Wnt-3a GMP Protein, CF
Full terms and conditions of sale can be found online in the Protein Sciences Segment T&Cs at: Terms & Conditions.
For preclinical, or clinical ex vivo use
Related Research Areas
Citations for Recombinant Human Wnt-3a GMP Protein, CF
Customer Reviews for Recombinant Human Wnt-3a GMP Protein, CF
There are currently no reviews for this product. Be the first to review Recombinant Human Wnt-3a GMP Protein, CF and earn rewards!
Have you used Recombinant Human Wnt-3a GMP Protein, CF?
Submit a review and receive an Amazon gift card!
$25/€18/£15/$25CAN/¥2500 Yen for a review with an image
$10/€7/£6/$10CAN/¥1110 Yen for a review without an image
Submit a review