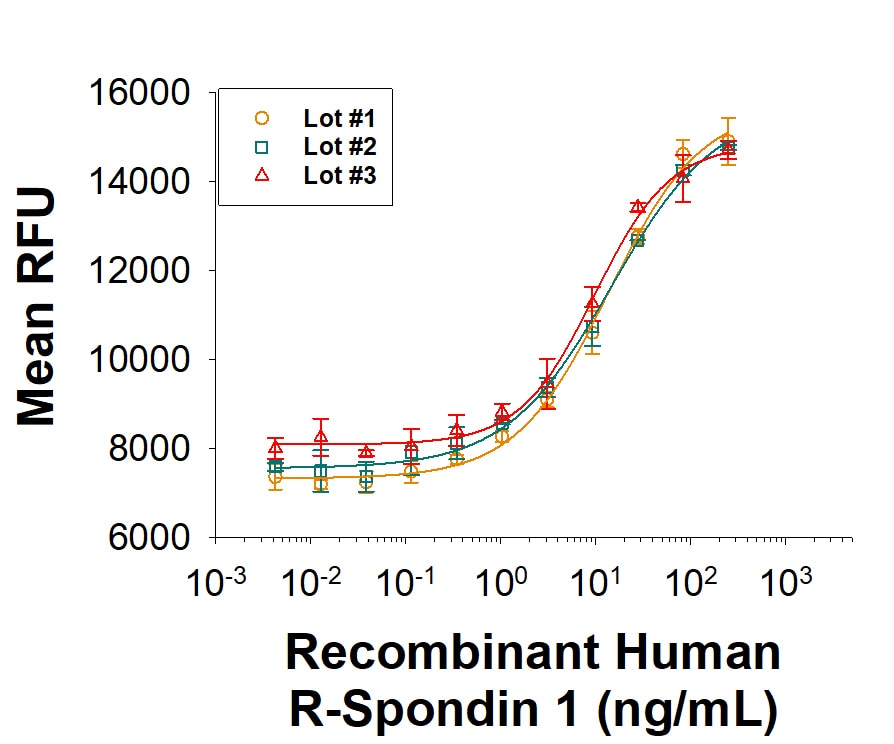
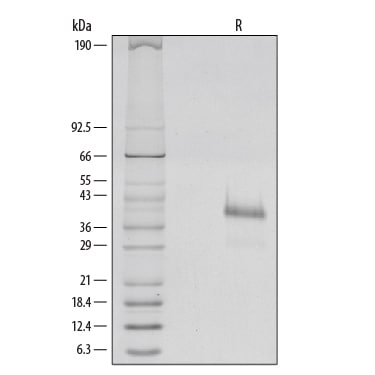
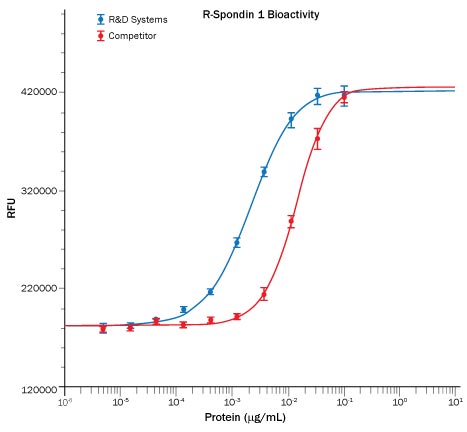
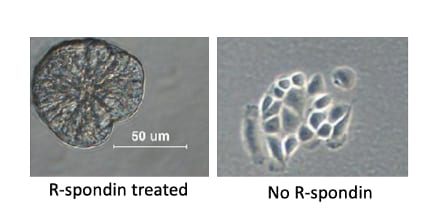
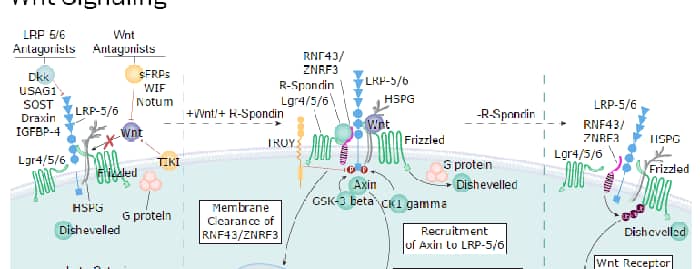
R-Spondin 1 (RSPO1, Roof plate-specific Spondin 1), also known as cysteine-rich and single thrombospondin domain containing protein 3 (Cristin 3), is a 27 kDa secreted protein that shares ~40% amino acid (aa) identity with three other R-Spondin family members (1, 2). All R-Spondins regulate Wnt/ beta-Catenin signaling but have distinct expression patterns (1-3). R-Spondin 1 competes with the Wnt antagonist DKK-1 for binding to the Wnt co-receptors, Kremen and LRP-6, reducing their DKK-1-mediated internalization (4). However, reports are mixed on whether R-Spondin 1 binds LRP-6 directly (4-6). R-Spondin 1 is expressed in early development at the roof plate boundary and is thought to contribute to dorsal neural tube development (3, 7). In humans, rare disruptions of the R-Spondin 1 gene are associated with tendencies for XX sex reversal (phenotypic male) or hermaphroditism, indicating a role for R-Spondin 1 in gender-specific differentiation (7, 8). Mutations in R-Spondin 1 are also linked with palmoplantar keratoderma, abnormal thickening of the skin on the palms of the hands and soles of the feet (7, 8). Postnatally, R-Spondin 1 is expressed by neuroendocrine cells in the intestine, adrenal gland and pancreas, and by epithelia in kidney and prostate (9). Injection of recombinant R-Spondin 1 in mice causes activation of beta-catenin and proliferation of intestinal crypt epithelial cells, and ameliorates experimental colitis (9, 10). Interest in R-Spondin 1 as a cell culture supplement has grown with the expansion of the organoid field. R-Spondin 1 is widely used in organoid cell culture workflows as a vital component that promotes both growth and survival of 3D organoids (11).
Structurally similar to other R-Spondins, R-Spondin 1 contains two adjacent cysteine-rich furin-like domains (aa 34-135) with one potential N-glycosylation site, followed by a thrombospondin (TSP-1) motif (aa 147-207) and a region rich in basic residues (aa 211-263). Only the furin-like domains are needed for beta-catenin stabilization (2, 12). A putative nuclear localization signal at the C-terminus may allow some expression in the nucleus (13). Potential isoforms of 200 and 236 aa have an alternate, shorter N-terminus or are missing aa 146-208, respectively (14). Over aa 21-263, human R-Spondin 1 shares 89%, 87%, 92%, 91%, 91% and 89% aa identity with mouse, rat, horse, dog, goat, and cow RSPO-1, respectively.