187 results for "AHR" in Products
AHR Products
AHR (Aryl-hydrocarbon receptor; also bHLHE76) is a 110 kDa member of the bHLH/PAS transcription factor family. It is widely expressed (breast, lung, liver), and serves many functions. First, it binds multiple xenobiotic chemicals in the cytoplasm. This induces dimerization with ARNT, translocation to the nucleus, and activation of P450 genes such as CYP1A1 and UGT1A6. Second, it appears to block cell cycle progression, possibly via a down-regulation of CDK proteins. And third, it blocks ...
| Reactivity: | Human, Mouse, Rat |
| Details: | Goat Polyclonal |
| Applications: | IHC, WB, ICC/IF, ChIP, GS |
| Reactivity: | Human, Mouse, Rat, Guinea Pig |
| Details: | Rabbit IgG Polyclonal |
| Applications: | IHC, WB, ELISA, Flow, ICC/IF, + 3 More |
| Reactivity: | Human, Mouse, Rat |
| Details: | Mouse IgG3 Kappa Monoclonal Clone #RPT9 |
| Applications: | IHC, WB, ELISA, Flow, ICC/IF, + 3 More |
| Reactivity: | Mouse |
| Details: | Sheep IgG Polyclonal |
| Applications: | WB, ICC/IF |
| Reactivity: | Human, Mouse |
| Details: | Mouse IgG1 kappa Monoclonal Clone #3B12 |
| Applications: | WB, ELISA, ICC/IF, IP, ChIP, + 1 More |
| Reactivity: | Human, Mouse, Rat |
| Details: | Mouse IgG1 Monoclonal Clone #RPT1 |
| Applications: | IHC, WB, ICC/IF, IP |
| Reactivity: | Human |
| Details: | Sheep IgG Polyclonal |
| Applications: | KO, WB, Flow, ICC/IF, Simple Western, + 1 More |
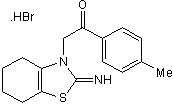
p53 inhibitor. Also aryl hydrocarbon receptor agonist
| Chemical Name: | 1-(4-Methylphenyl)-2-(4,5,6,7-tetrahydro-2-imino-3(2H)-benzothiazolyl)ethanone hydrobromide |
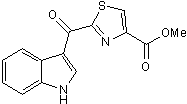
Endogenous agonist for the transcription factor aryl hydrocarbon receptor
| Alternate Names: | Aryl hydrocarbon receptor ligand |
| Chemical Name: | 2-(1H-Indol-3-ylcarbonyl)-4-thiazolecarboxylic acid methyl ester |
| Purity: | ≥98% |
Potent aryl hydrocarbon receptor (AhR) antagonist
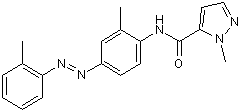
| Chemical Name: | 1-Methyl-N-[2-methyl-4-[2-(2-methylphenyl)diazenyl]phenyl-1H-pyrazole-5-carboxamide |
| Purity: | ≥98% |
| Reactivity: | Human |
| Details: | Mouse IgG1 Monoclonal Clone #684217 |
| Applications: | IHC |
Aryl hydrocarbon receptor (AhR) antagonist; promotes HSC expansion and engraftment
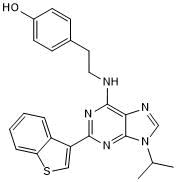
| Chemical Name: | 4-[2-[[2-Benzo[b]thien-3-yl-9-(1-methylethyl)-9H-purin-6-yl]amino]ethyl]phenol |
| Purity: | ≥98% |
| Reactivity: | Human |
| Details: | Rabbit IgG Polyclonal |
| Applications: | IHC |
High affinity aryl hydrocarbon receptor (AhR) agonist
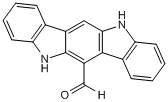
| Chemical Name: | 5,11-Dihydroindolo[3,2-b]carbazole-6-carboxaldehyde |
| Purity: | ≥95% |
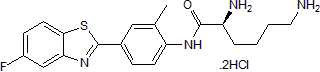
Prodrug of the antitumor agent 5F 203
| Chemical Name: | (2S)-2,6-Diamino-N-[4-(5-fluoro-2-benzothiazolyl)-2-methylphenyl]hexanamide dihydrochloride |
| Purity: | ≥98% |
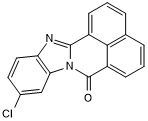
Potent aryl hydrocarbon receptor (AhR) agonist; orally bioavailable
| Chemical Name: | 10-Chloro-7H-benzimidazo[2,1-a]benz[de]isoquinolin-7-one |
| Purity: | ≥97% |
| Reactivity: | Human |
| Details: | Rabbit IgG Polyclonal |
| Applications: | IHC, ICC/IF |
| Reactivity: | Human |
| Details: | Sheep IgG Polyclonal |
| Applications: | KO, WB, Flow, ICC/IF, CyTOF-ready |
| Reactivity: | Human |
| Details: | Sheep IgG Polyclonal |
| Applications: | KO, WB, Flow, ICC/IF, CyTOF-ready |
| Reactivity: | Human |
| Details: | Sheep IgG Polyclonal |
| Applications: | KO, WB, Flow, ICC/IF, CyTOF-ready |
| Reactivity: | Human |
| Details: | Sheep IgG Polyclonal |
| Applications: | KO, WB, Flow, ICC/IF, CyTOF-ready |
| Reactivity: | Human |
| Details: | Sheep IgG Polyclonal |
| Applications: | KO, WB, Flow, ICC/IF, CyTOF-ready |
| Reactivity: | Human |
| Details: | Sheep IgG Polyclonal |
| Applications: | KO, WB, Flow, ICC/IF, CyTOF-ready |
| Reactivity: | Human |
| Details: | Sheep IgG Polyclonal |
| Applications: | KO, WB, Flow, ICC/IF, CyTOF-ready |
| Reactivity: | Human |
| Details: | Sheep IgG Polyclonal |
| Applications: | KO, WB, Flow, ICC/IF, CyTOF-ready |


![Immunocytochemistry/ Immunofluorescence: AHR Antibody [NB100-128] Immunocytochemistry/ Immunofluorescence: AHR Antibody [NB100-128]](https://resources.rndsystems.com/images/products/AHR-Antibody-Immunocytochemistry-Immunofluorescence-NB100-128-img0004.jpg)
![Western Blot: AHR Antibody - BSA Free [NB100-2289] Knockdown Validated: AHR Antibody - BSA Free [NB100-2289]](https://resources.rndsystems.com/images/products/AHR-Antibody-Knockdown-Validated-NB100-2289-img0014.jpg)
![Western Blot: AHR Antibody (RPT9)BSA Free [NB300-515] Western Blot: AHR Antibody (RPT9)BSA Free [NB300-515]](https://resources.rndsystems.com/images/products/AHR-Antibody-RPT9-Western-Blot-NB300-515-img0018.jpg)

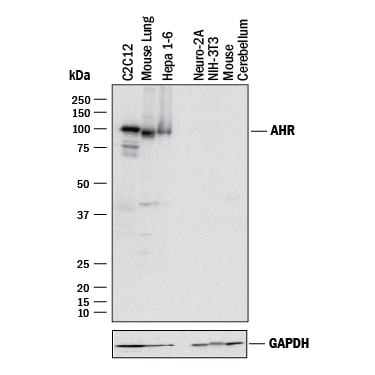
![Western Blot: AHR Antibody (3B12) [H00000196-M02] Western Blot: AHR Antibody (3B12) [H00000196-M02]](https://resources.rndsystems.com/images/products/AHR-Antibody-3B12-Western-Blot-H00000196-M02-img0016.jpg)
![Western Blot: AHR Antibody (RPT1) [NB300-530] Western Blot: AHR Antibody (RPT1) [NB300-530]](https://resources.rndsystems.com/images/products/AHR-Antibody-RPT1-Western-Blot-NB300-530-img0013.jpg)
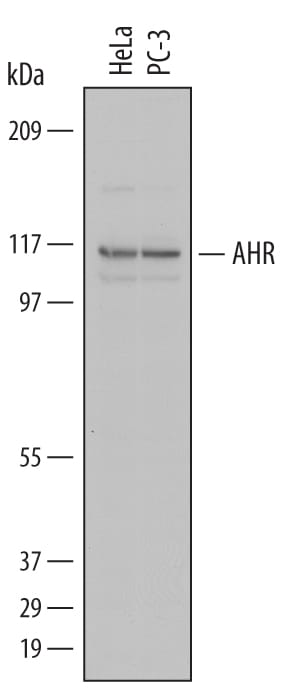

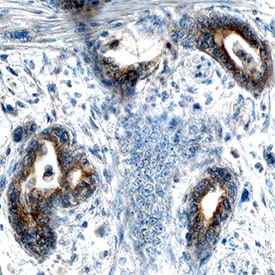
![Immunohistochemistry-Paraffin: AHR Antibody - BSA Free [NBP1-89975] AHR Antibody - BSA Free Immunohistochemistry-Paraffin: AHR Antibody - BSA Free [NBP1-89975]](https://resources.rndsystems.com/images/products/nbp1-89975_rabbit-polyclonal-ahr-antibody-15520258564345.jpg)
![Immunocytochemistry/ Immunofluorescence: AHR Antibody [NBP1-89974] Immunocytochemistry/ Immunofluorescence: AHR Antibody [NBP1-89974]](https://resources.rndsystems.com/images/products/AHR-Antibody-Immunocytochemistry-Immunofluorescence-NBP1-89974-img0004.jpg)