148 results for "cIAP-1/HIAP-2" in Products
cIAP-1/HIAP-2 Products
Cellular inhibitor of apoptosis protein1 (cIAP-1, also known as BIRC2, MIHB, and HIAP2) and 2 (cIAP-2, also known as BIRC3, MIHC, and HIAP1), are members of the inhibitor of apoptosis (IAP) family of proteins that inhibit the proteolytic activity of mature caspases. Structurally, cIAP-1 and cIAP-2 are each comprised of 3 BIR (baculovirus inhibitor of apoptosis) domains, a RING finger domain, and a caspase recruitment domain (CARD). Human cIAP-1 and cIAP-2 share 70% sequence identity. ...
| Reactivity: | Human |
| Details: | Goat IgG Polyclonal |
| Applications: | KO, IHC, WB, Simple Western |
| Reactivity: | Human, Mouse |
| Details: | Mouse IgG2A Monoclonal Clone #315301 |
| Applications: | WB, Simple Western |
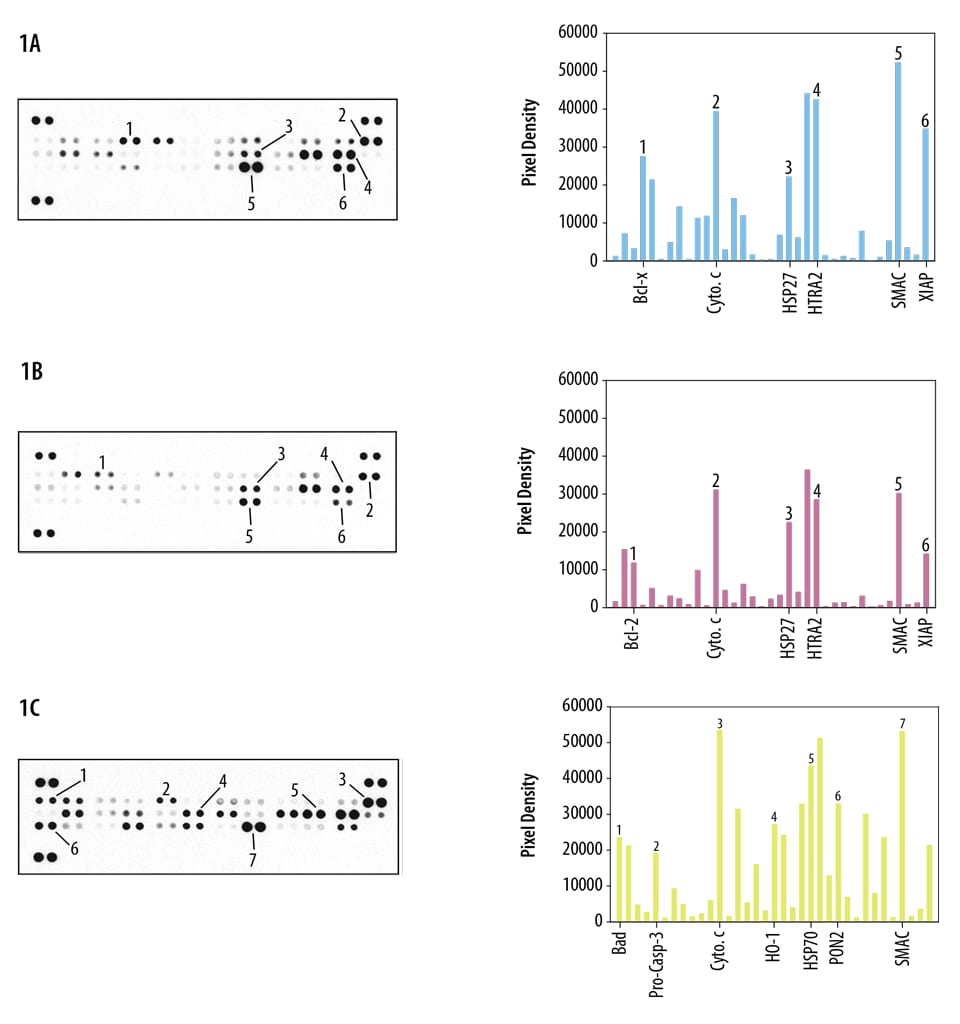
Contains 4 membranes - each spotted in duplicate with 35 different apoptosis antibodies
| Reactivity: | Human |
| Reactivity: | Human |
| Details: | Mouse IgG1 Monoclonal Clone #681732 |
| Applications: | KO, WB, Simple Western |
| Reactivity: | Human |
| Source: | E. coli |
| Accession #: | Q13490 |
| Applications: | Inhibition Activity |
| Reactivity: | Human |
| Details: | Rabbit IgG Polyclonal |
| Applications: | IHC, WB, IP |
| Reactivity: | Human, Mouse, Rat |
| Details: | Rabbit IgG Polyclonal |
| Applications: | IHC, WB, ELISA, ICC/IF |
| Reactivity: | Human |
| Details: | Mouse IgG1 Monoclonal Clone #681724 |
| Applications: | IHC |
Recombinant Monoclonal Antibody
| Reactivity: | Human |
| Details: | Rabbit IgG Monoclonal Clone #HL1045 |
| Applications: | WB |
| Reactivity: | Human, Rat |
| Details: | Rabbit IgG Polyclonal |
| Applications: | IHC, WB |
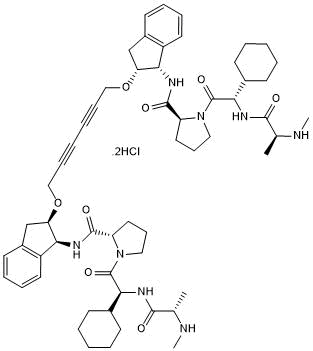
Dimeric Smac mimetic; potent IAP inhibitor
| Chemical Name: | 3,3'-[2,4-Hexadiyne-1,6-diylbis[oxy[(1S,2R)-2,3-dihydro-1H-indene-2,1-diyl]]]bis[N-methyl-L-alanyl-(2S)-2-cyclohexylglycyl-L-prolinamide |
| Purity: | ≥97% |
Recombinant Monoclonal Antibody
| Reactivity: | Human, Rat |
| Details: | Rabbit IgG Monoclonal Clone #2L0W9 |
| Applications: | IHC, WB |
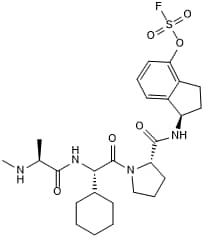
Potent Lys-covalent pan-IAP inhibitor
| Chemical Name: | (R)-1-((S)-1-((S)-2-Cyclohexyl-2-((S)-2-(methylamino)propanamido)acetyl)pyrrolidine-2-carboxamido)-2,3-dihydro-1H-inden-4-yl sulfurofluoridate |
| Purity: | ≥95% |
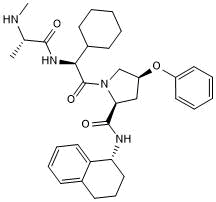
Monovalent IAP antagonist
| Chemical Name: | (2S,4S)-1-((S)-2-Cyclohexyl-2-((S)-2-(methylamino)propanamido)acetyl)-4-phenoxy-N-((R)-1,2,3,4-tetrahydronaphthalen-1-yl)pyrrolidine-2-carboxamide |
| Purity: | ≥97% |
Recombinant Monoclonal Antibody
| Reactivity: | Human |
| Details: | Rabbit IgG Monoclonal Clone #S01-1C5 |
| Applications: | WB, ICC/IF |
| Reactivity: | Human |
| Details: | Goat IgG Polyclonal |
| Applications: | KO, IHC, WB |
| Reactivity: | Human |
| Details: | Goat IgG Polyclonal |
| Applications: | KO, IHC, WB |
| Reactivity: | Human |
| Details: | Goat IgG Polyclonal |
| Applications: | KO, IHC, WB |
| Reactivity: | Human |
| Details: | Goat IgG Polyclonal |
| Applications: | KO, IHC, WB |
| Reactivity: | Human |
| Details: | Goat IgG Polyclonal |
| Applications: | KO, IHC, WB |
| Reactivity: | Human |
| Details: | Goat IgG Polyclonal |
| Applications: | KO, IHC, WB |
| Reactivity: | Human |
| Details: | Goat IgG Polyclonal |
| Applications: | KO, IHC, WB |
| Reactivity: | Human |
| Details: | Goat IgG Polyclonal |
| Applications: | KO, IHC, WB |
| Reactivity: | Human |
| Details: | Mouse IgG1 Monoclonal Clone #681724 |
| Applications: | IHC |
| Reactivity: | Human |
| Details: | Mouse IgG1 Monoclonal Clone #681724 |
| Applications: | IHC |



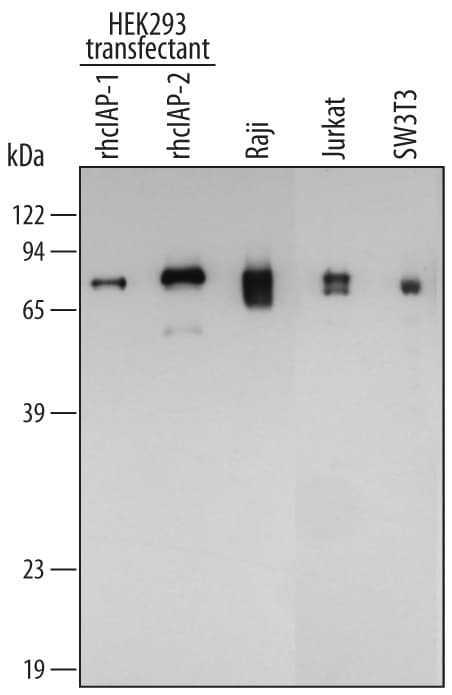
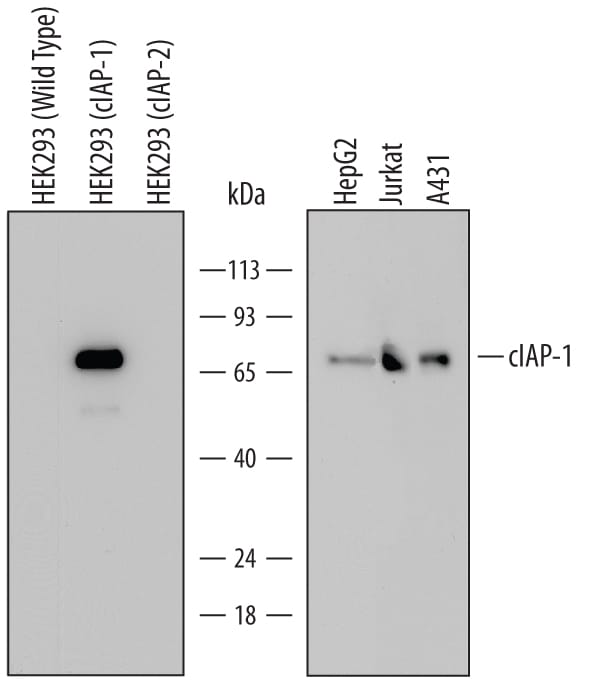


![Western Blot: cIAP-1/HIAP-2 Antibody [NB100-56128] Western Blot: cIAP-1/HIAP-2 Antibody [NB100-56128]](https://resources.rndsystems.com/images/products/cIAP-1-HIAP-2-Antibody-Western-Blot-NB100-56128-img0005.jpg)
![Western Blot: cIAP-1/HIAP-2 AntibodyBSA Free [NBP1-77193] Western Blot: cIAP-1/HIAP-2 AntibodyBSA Free [NBP1-77193]](https://resources.rndsystems.com/images/products/cIAP-1-HIAP-2-Antibody-Western-Blot-NBP1-77193-img0008.jpg)
![Western Blot: cIAP-1/HIAP-2 Antibody (HL1045) [NBP3-13700] Western Blot: cIAP-1/HIAP-2 Antibody (HL1045) [NBP3-13700]](https://resources.rndsystems.com/images/products/cIAP-1-HIAP-2-Antibody-HL1045-Western-Blot-NBP3-13700-img0002.jpg)
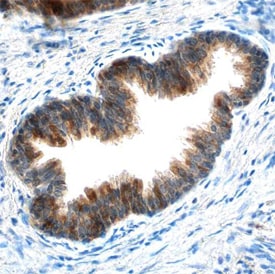
![Immunohistochemistry-Paraffin: cIAP-1/HIAP-2 Antibody [NBP1-90133] Immunohistochemistry-Paraffin: cIAP-1/HIAP-2 Antibody [NBP1-90133]](https://resources.rndsystems.com/images/products/cIAP-1-HIAP-2-Antibody-Immunohistochemistry-Paraffin-NBP1-90133-img0006.jpg)

![Western Blot: cIAP-1/HIAP-2 Antibody (2L0W9) [NBP3-15860] Western Blot: cIAP-1/HIAP-2 Antibody (2L0W9) [NBP3-15860]](https://resources.rndsystems.com/images/products/cIAP-1-HIAP-2-Antibody-2L0W9-Western-Blot-NBP3-15860-img0002.jpg)
![Western Blot: cIAP-1/HIAP-2 Antibody (S01-1C5) [NBP3-19605] Western Blot: cIAP-1/HIAP-2 Antibody (S01-1C5) [NBP3-19605]](https://resources.rndsystems.com/images/products/cIAP-1-HIAP-2-Antibody-S01-1C5-Western-Blot-NBP3-19605-img0002.jpg)