Human Cadherin‑13 Antibody
R&D Systems | Catalog # AF3264

Key Product Details
Species Reactivity
Validated:
Cited:
Applications
Validated:
Cited:
Label
Antibody Source
Product Specifications
Immunogen
Glu23-Ala692
Accession # P55290
Specificity
Clonality
Host
Isotype
Scientific Data Images for Human Cadherin‑13 Antibody
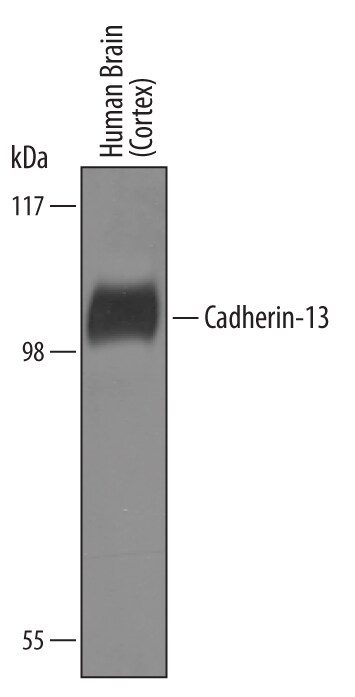
Detection of Human Cadherin‑13 by Western Blot.
Western blot shows lysates of human brain (cortex) tissue. PVDF Membrane was probed with 1 µg/mL of Goat Anti-Human Cadherin-13 Antigen Affinity-purified Polyclonal Antibody (Catalog # AF3264) followed by HRP-conjugated Anti-Goat IgG Secondary Antibody (Catalog # HAF019). A specific band was detected for Cadherin-13 at approximately 105 kDa (as indicated). This experiment was conducted under reducing conditions and using Immunoblot Buffer Group 8.Cadherin‑13 in NCI-H460 Human Cell Line.
Cadherin-13 was detected in immersion fixed NCI-H460 human large cell lung carcinoma cell line using 10 µg/mL Goat Anti-Human Cadherin-13 Antigen Affinity-purified Polyclonal Antibody (Catalog # AF3264) for 3 hours at room temperature. Cells were stained with the NorthernLights™ 557-conjugated Anti-Goat IgG Secondary Antibody (red; Catalog # NL001) and counter-stained with DAPI (blue). View our protocol for Fluorescent ICC Staining of Cells on Coverslips.Cadherin‑13 in Human Skin.
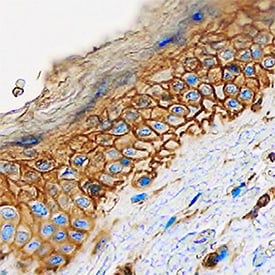
Cadherin-13 was detected in immersion fixed paraffin-embedded sections of human skin using Goat Anti-Human Cadherin-13 Antigen Affinity-purified Polyclonal Antibody (Catalog # AF3264) at 0.1 µg/mL overnight at 4 °C. Tissue was stained using the Anti-Goat HRP-DAB Cell & Tissue Staining Kit (brown; Catalog # CTS008) and counterstained with hematoxylin (blue). Specific staining was localized to plasma membranes of keratinocytes. View our protocol for Chromogenic IHC Staining of Paraffin-embedded Tissue Sections.Detection of Human Cadherin‑13 by Simple WesternTM.
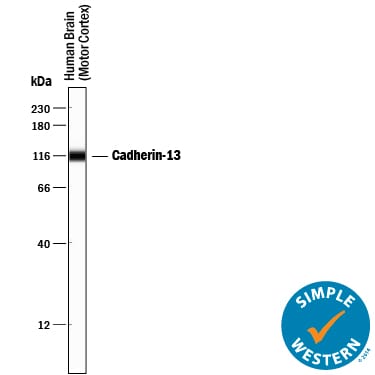
Simple Western lane view shows lysates of human brain (motor cortex) tissue, loaded at 0.2 mg/mL. A specific band was detected for Cadherin-13 at approximately 114 kDa (as indicated) using 10 µg/mL of Goat Anti-Human Cadherin-13 Antigen Affinity-purified Polyclonal Antibody (Catalog # AF3264) followed by 1:50 dilution of HRP-conjugated Anti-Goat IgG Secondary Antibody (Catalog # HAF109). This experiment was conducted under reducing conditions and using the 12-230 kDa separation system.Detection of Human Cadherin-13 by Western Blot
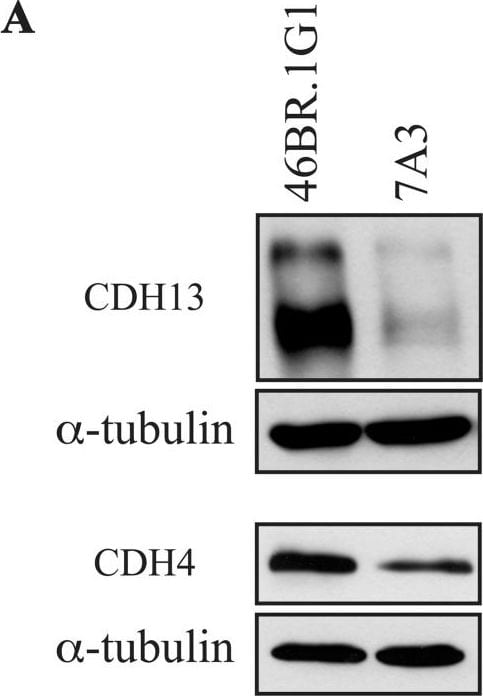
Differential expression of cadherin 13 and cadherin 4 proteins in 46BR.1G1 and 7A3 cells.(A) Cell lysates from 46BR.1G1 and 7A3 cells were analyzed by Western blotting with anti-cadherin 13, anti-cadherin 4, and anti-alpha -tubulin antibodies. (B) Quantification of the assay was performed by densitometric analysis with NIH ImageJ 1.43 program. Bars show mean ± SEM of three independent experiments. Image collected and cropped by CiteAb from the following publication (https://dx.plos.org/10.1371/journal.pone.0130561), licensed under a CC-BY license. Not internally tested by R&D Systems.Detection of Human Cadherin-13 by Western Blot
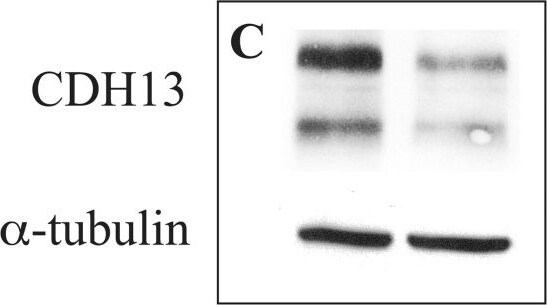
Differential expression of cadherin 13 and cadherin 4 proteins in 46BR.1G1 and 31W cells.Cell lysates from 46BR.1G1 and 31W cells were analyzed by Western blotting with antibodies against the indicated proteins. Image collected and cropped by CiteAb from the following publication (https://dx.plos.org/10.1371/journal.pone.0130561), licensed under a CC-BY license. Not internally tested by R&D Systems.Detection of Cadherin-13 by Western Blot
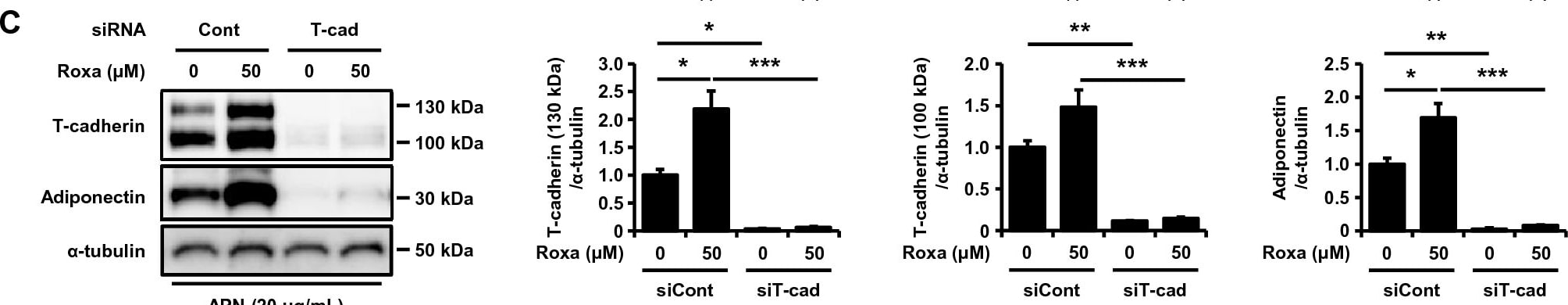
HIF-1 activation-induced T-cadherin upregulation increased adiponectin accumulation and EV production. (A) Western blot analysis of total cell lysates. UV-F2 cells cultured in DMEM containing 5% serum from adiponectin knockout mice were treated with or without high molecular weight adiponectin (HMW-APN) (10 μg/mL) and roxadustat (Roxa) (50 μM) for 48 h (n = 3 for each group). (B) Western blot analysis of EVs isolated from cell culture medium by differential ultracentrifugation. UV-F2 cells cultured in FBS-free Advanced DMEM were treated with or without HMW-APN (20 μg/mL) and roxadustat (50 μM) for 48 h (n = 3 for each group). Alix, TSG101, and syntenin were evaluated as EV markers. (C) Western blot analysis of total cell lysates. UV-F2 cells transfected control (Cont) or T-cadherin (T-cad) siRNA were cultured in DMEM containing 5% serum from adiponectin knockout mice with or without HMW-APN (20 μg/mL) and roxadustat (50 μM) for 48 h (n = 3 for each group). (D) Western blot analysis of EVs isolated from cell culture medium. UV-F2 cells transfected Cont or T-cad siRNA were cultured in FBS-free Advanced DMEM with or without HMW-APN (20 μg/mL) and roxadustat (50 μM) for 48 h (n = 3). Data are means ± SEMs. *p < 0.05, **p < 0.01, and ***p < 0.001 (Tukey–Kramer test). Image collected and cropped by CiteAb from the following open publication (https://www.nature.com/articles/s41598-024-51935-6), licensed under a CC-BY license. Not internally tested by R&D Systems.Detection of Cadherin-13 by Western Blot
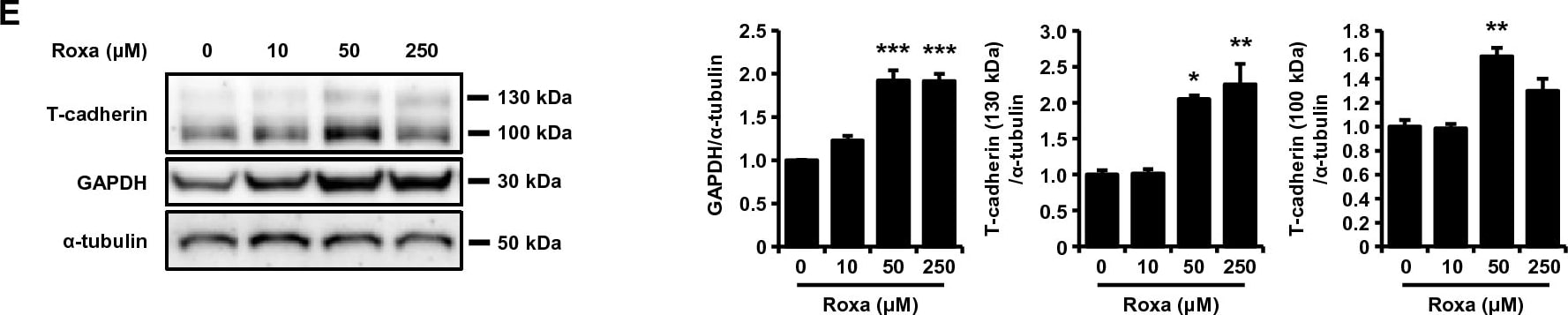
HIF-1 activation upregulated transcription of the T-cadherin mRNA and increased T-cadherin protein in UV-F2 endothelial cells. (A) Quantitative PCR analysis of murine endothelial UV-F2 cells treated with roxadustat (Roxa) for 24 h, daprodustat (Dapro) for 48 h, or deferoxamine (DFO) for 24 h at the indicated concentrations (n = 3 for each group). Data are means ± SEMs. *p < 0.05, **p < 0.01, and ***p < 0.001 versus control (Dunnett's test). (B) Quantitative PCR analysis of UV-F2 cells treated with or without roxadustat (50 μM) and 1 μM of 2-methoxyestradiol (2-ME), an inhibitor of HIF-1, for 24 h (n = 3 for each group). Data are means ± SEMs. *p < 0.05, **p < 0.01, and ***p < 0.001 (Tukey–Kramer test). (C) The constructs used for reporter assay were summarized (left). Luciferase reporter assay using the promoter region of the mouse T-cadherin gene (mCdh13) in HEK293 cells. Luciferase activities 24 h after treatment with or without roxadustat (50 μM) were quantified by calculating Firefly luciferase activity against an internal standard, Renilla luciferase activity (n = 3 for each group). Data are means ± SEMs. *p < 0.05 (unpaired t test). (D) mRNA stability assay using actinomycin D. UV-F2 cells were incubated with or without roxadustat (50 μM) and 5 μg/mL of actinomycin D for the time indicated (n = 3 for each group). (E) and (F) Western blot analysis of total cell lysates. UV-F2 cells were treated with roxadustat (E) or daprodustat (F) for 48 h at the indicated concentrations (n = 3 for each group). Data are means ± SEMs. *p < 0.05, **p < 0.01, and ***p < 0.001 versus control (Dunnett's test or unpaired t test). Image collected and cropped by CiteAb from the following open publication (https://www.nature.com/articles/s41598-024-51935-6), licensed under a CC-BY license. Not internally tested by R&D Systems.Detection of Cadherin‑13 in NCI-H460 cells by Flow Cytometry
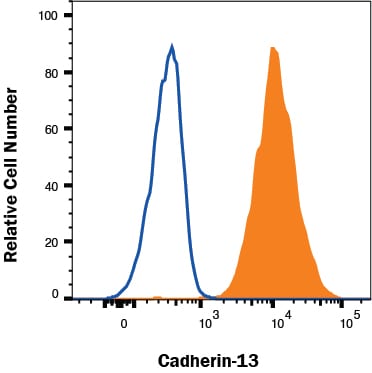
NCI-H460 cells were stained with Goat Anti-Human Cadherin‑13 Antigen Affinity-purified Polyclonal Antibody (Catalog # AF3264, filled histogram) or isotype control antibody (Catalog # AB-108-C, open histogram) followed by Allophycocyanin-conjugated Anti-Goat IgG Secondary Antibody (Catalog # F0108). View our protocol for Staining Membrane-associated Proteins.Applications for Human Cadherin‑13 Antibody
CyTOF-ready
Flow Cytometry
Sample: NCI‑H460 human large cell lung carcinoma cell line
Immunocytochemistry
Sample: Immersion fixed NCI-H460 human large cell lung carcinoma cell line
Immunohistochemistry
Sample: Immersion fixed paraffin-embedded sections of human skin
Simple Western
Sample: Human brain (motor cortex) tissue
Western Blot
Sample: Human brain (cortex) tissue
Flow Cytometry Panel Builder
Bio-Techne Knows Flow Cytometry
Save time and reduce costly mistakes by quickly finding compatible reagents using the Panel Builder Tool.
Advanced Features
- Spectra Viewer - Custom analysis of spectra from multiple fluorochromes
- Spillover Popups - Visualize the spectra of individual fluorochromes
- Antigen Density Selector - Match fluorochrome brightness with antigen density
Formulation, Preparation, and Storage
Purification
Reconstitution
Reconstitute at 0.2 mg/mL in sterile PBS. For liquid material, refer to CoA for concentration.
Formulation
*Small pack size (-SP) is supplied either lyophilized or as a 0.2 µm filtered solution in PBS.
Shipping
Stability & Storage
- 12 months from date of receipt, -20 to -70 °C as supplied.
- 1 month, 2 to 8 °C under sterile conditions after reconstitution.
- 6 months, -20 to -70 °C under sterile conditions after reconstitution.
Calculators
Background: Cadherin-13
References
- Tanihara, H. et al. (1994) Cell Adhes. Commun. 2:15.
- Philippova, M. et al. (2009) Cell. Signal. 21:1035.
Alternate Names
Gene Symbol
UniProt
Additional Cadherin-13 Products
Product Documents for Human Cadherin‑13 Antibody
Certificate of Analysis
To download a Certificate of Analysis, please enter a lot or batch number in the search box below.
Note: Certificate of Analysis not available for kit components.
Product Specific Notices for Human Cadherin‑13 Antibody
For research use only
Related Research Areas
Citations for Human Cadherin‑13 Antibody
Customer Reviews for Human Cadherin‑13 Antibody
There are currently no reviews for this product. Be the first to review Human Cadherin‑13 Antibody and earn rewards!
Have you used Human Cadherin‑13 Antibody?
Submit a review and receive an Amazon gift card!
$25/€18/£15/$25CAN/¥2500 Yen for a review with an image
$10/€7/£6/$10CAN/¥1110 Yen for a review without an image
Submit a review
Protocols
Find general support by application which include: protocols, troubleshooting, illustrated assays, videos and webinars.
- 7-Amino Actinomycin D (7-AAD) Cell Viability Flow Cytometry Protocol
- Antigen Retrieval Protocol (PIER)
- Antigen Retrieval for Frozen Sections Protocol
- Appropriate Fixation of IHC/ICC Samples
- Cellular Response to Hypoxia Protocols
- Chromogenic IHC Staining of Formalin-Fixed Paraffin-Embedded (FFPE) Tissue Protocol
- Chromogenic Immunohistochemistry Staining of Frozen Tissue
- ClariTSA™ Fluorophore Kits
- Detection & Visualization of Antibody Binding
- Extracellular Membrane Flow Cytometry Protocol
- Flow Cytometry Protocol for Cell Surface Markers
- Flow Cytometry Protocol for Staining Membrane Associated Proteins
- Flow Cytometry Staining Protocols
- Flow Cytometry Troubleshooting Guide
- Fluorescent IHC Staining of Frozen Tissue Protocol
- Graphic Protocol for Heat-induced Epitope Retrieval
- Graphic Protocol for the Preparation and Fluorescent IHC Staining of Frozen Tissue Sections
- Graphic Protocol for the Preparation and Fluorescent IHC Staining of Paraffin-embedded Tissue Sections
- Graphic Protocol for the Preparation of Gelatin-coated Slides for Histological Tissue Sections
- ICC Cell Smear Protocol for Suspension Cells
- ICC Immunocytochemistry Protocol Videos
- ICC for Adherent Cells
- IHC Sample Preparation (Frozen sections vs Paraffin)
- Immunocytochemistry (ICC) Protocol
- Immunocytochemistry Troubleshooting
- Immunofluorescence of Organoids Embedded in Cultrex Basement Membrane Extract
- Immunofluorescent IHC Staining of Formalin-Fixed Paraffin-Embedded (FFPE) Tissue Protocol
- Immunohistochemistry (IHC) and Immunocytochemistry (ICC) Protocols
- Immunohistochemistry Frozen Troubleshooting
- Immunohistochemistry Paraffin Troubleshooting
- Intracellular Flow Cytometry Protocol Using Alcohol (Methanol)
- Intracellular Flow Cytometry Protocol Using Detergents
- Intracellular Nuclear Staining Flow Cytometry Protocol Using Detergents
- Intracellular Staining Flow Cytometry Protocol Using Alcohol Permeabilization
- Intracellular Staining Flow Cytometry Protocol Using Detergents to Permeabilize Cells
- Preparing Samples for IHC/ICC Experiments
- Preventing Non-Specific Staining (Non-Specific Binding)
- Primary Antibody Selection & Optimization
- Propidium Iodide Cell Viability Flow Cytometry Protocol
- Protocol for Heat-Induced Epitope Retrieval (HIER)
- Protocol for Liperfluo
- Protocol for Making a 4% Formaldehyde Solution in PBS
- Protocol for VisUCyte™ HRP Polymer Detection Reagent
- Protocol for the Characterization of Human Th22 Cells
- Protocol for the Characterization of Human Th9 Cells
- Protocol for the Fluorescent ICC Staining of Cell Smears - Graphic
- Protocol for the Fluorescent ICC Staining of Cultured Cells on Coverslips - Graphic
- Protocol for the Preparation & Fixation of Cells on Coverslips
- Protocol for the Preparation and Chromogenic IHC Staining of Frozen Tissue Sections
- Protocol for the Preparation and Chromogenic IHC Staining of Frozen Tissue Sections - Graphic
- Protocol for the Preparation and Chromogenic IHC Staining of Paraffin-embedded Tissue Sections
- Protocol for the Preparation and Chromogenic IHC Staining of Paraffin-embedded Tissue Sections - Graphic
- Protocol for the Preparation and Fluorescent ICC Staining of Cells on Coverslips
- Protocol for the Preparation and Fluorescent ICC Staining of Non-adherent Cells
- Protocol for the Preparation and Fluorescent ICC Staining of Stem Cells on Coverslips
- Protocol for the Preparation and Fluorescent IHC Staining of Frozen Tissue Sections
- Protocol for the Preparation and Fluorescent IHC Staining of Paraffin-embedded Tissue Sections
- Protocol for the Preparation of Gelatin-coated Slides for Histological Tissue Sections
- Protocol for the Preparation of a Cell Smear for Non-adherent Cell ICC - Graphic
- Protocol: Annexin V and PI Staining by Flow Cytometry
- Protocol: Annexin V and PI Staining for Apoptosis by Flow Cytometry
- R&D Systems Quality Control Western Blot Protocol
- TUNEL and Active Caspase-3 Detection by IHC/ICC Protocol
- The Importance of IHC/ICC Controls
- Troubleshooting Guide: Fluorokine Flow Cytometry Kits
- Troubleshooting Guide: Immunohistochemistry
- Troubleshooting Guide: Western Blot Figures
- Western Blot Conditions
- Western Blot Protocol
- Western Blot Protocol for Cell Lysates
- Western Blot Troubleshooting
- Western Blot Troubleshooting Guide
- View all Protocols, Troubleshooting, Illustrated assays and Webinars