RNF31 (RING [Really INteresting Gene] Finger Protein 31; also HOIL-1-interacting protein/HOIP, and Zn in-between-RING-finger ubiquitin-associated domain protein/ZIBRA) is a cytoplasmic E3 ubiquitin-protein ligase that is found in breast epithelium and multiple cancer types. Although its predicted MW is 102 kDa, it runs anomalously at 95-135 kDa in SDS-Page. Ubiquitin (Ub) chains are typically thought of as 9 kDa additions to Lys residues of target molecules. The activity associated with Ub addition depends upon the location of the attachment, and the monomeric vs. polymeric nature of the chains. Ub can also be added to N-terminal Met residues by an intracellular complex called LUBAC (Linear Ub chain Assembly Complex). This complex is key to NF kappa B pathway activation. Following exposure of cells to cytokines, LUBAC ubiquitinates NEMO, which subsequently induces IKK beta phosphorylation, I kappa B alpha degradation, and NF kappa B translocation into the nucleus with gene activation. The LUBAC complex contains RNF31, HOIL-1L and sharpin, and it is now known that RNF31 is the catalyst for linear Ub chain formation. Human RNF31 is 1072 amino acids (aa) in length. It contains three consecutive RanBP2-type Zn finger domains (aa 299-438), a utilized phosphoryation site at Ser466, one UBA domain (aa 564-615), and two RING-type Zn finger domains (aa 699-747 and 860-909) with an intervening IBR-type Zn finger region (aa 779-841). There are at least two isoform variants, one that contains a 13 aa substitution for aa 1-164, and another that shows a deletion of aa 73-630 coupled to a deletion of aa 833-841. Human RNF31 shares 87% aa sequence identity with mouse RNF31.
Human HOIP/RNF31 Antibody
R&D Systems | Catalog # MAB8039

Key Product Details
Validated by
Biological Validation
Species Reactivity
Validated:
Human
Cited:
Human
Applications
Validated:
Western Blot, Immunocytochemistry, Simple Western
Cited:
Western Blot, Flow Cytometry, Immunocytochemistry, Immunoprecipitation
Label
Unconjugated
Antibody Source
Monoclonal Mouse IgG1 Clone # 875227
Loading...
Product Specifications
Immunogen
E. coli-derived recombinant human HOIP/RNF31
Arg970-Lys1072
Accession # Q96EP0
Arg970-Lys1072
Accession # Q96EP0
Specificity
Detects human HOIP/RNF31 in direct ELISA and Western Blot.
Clonality
Monoclonal
Host
Mouse
Isotype
IgG1
Scientific Data Images for Human HOIP/RNF31 Antibody
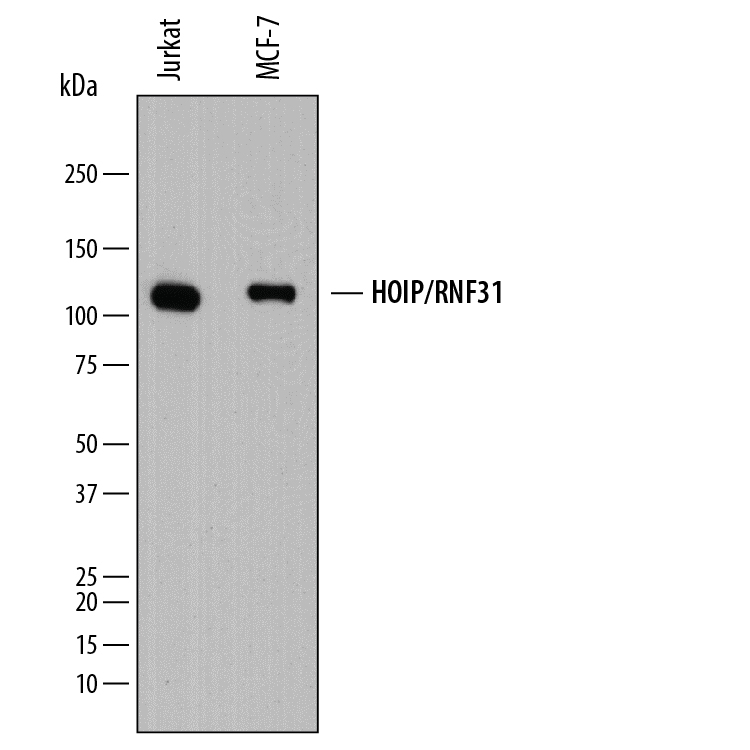
Detection of Human HOIP/RNF31 by Western Blot.
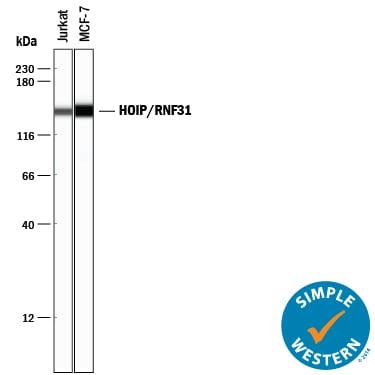
Western blot shows lysates of Jurkat human acute T cell leukemia cell line and MCF-7 human breast cancer cell line. PVDF membrane was probed with 0.5 µg/mL of Mouse Anti-Human HOIP/RNF31 Monoclonal Antibody (Catalog # MAB8039) followed by HRP-conjugated Anti-Mouse IgG Secondary Antibody (Catalog # HAF018). A specific band was detected for HOIP/RNF31 at approximately 120 kDa (as indicated). This experiment was conducted under reducing conditions and using Immunoblot Buffer Group 1.HOIP/RNF31 in MCF‑7 Human Cell Line.
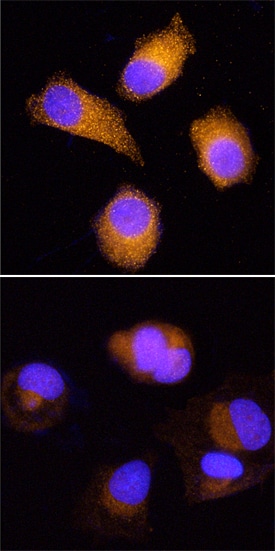
HOIP/RNF31 was detected in immersion fixed MCF-7 human breast cancer cell line, untreated (upper panel) or treated with Proteasome Inhibitor I (Tocris Bioscience, Catalog # 4045, lower panel), using Mouse Anti-Human HOIP/RNF31 Monoclonal Antibody (Catalog # MAB8039) at 25 µg/mL for 3 hours at room temperature. Cells were stained using the NorthernLights™ 557-conjugated Anti-Mouse IgG Secondary Antibody (red; Catalog # NL007) and counterstained with DAPI (blue). Specific staining was localized to cytoplasm. View our protocol for Fluorescent ICC Staining of Cells on Coverslips.Detection of Human HOIP/RNF31 by Simple WesternTM.
Simple Western lane view shows lysates of Jurkat human acute T cell leukemia cell line and MCF‑7 human breast cancer cell line, loaded at 0.5 mg/mL. A specific band was detected for HOIP/RNF31 at approximately 144 kDa (as indicated) using 5 µg/mL of Mouse Anti-Human HOIP/RNF31 Monoclonal Antibody (Catalog # MAB8039). This experiment was conducted under reducing conditions and using the 12-230 kDa separation system.Detection of HOIP/RNF31 by Western Blot
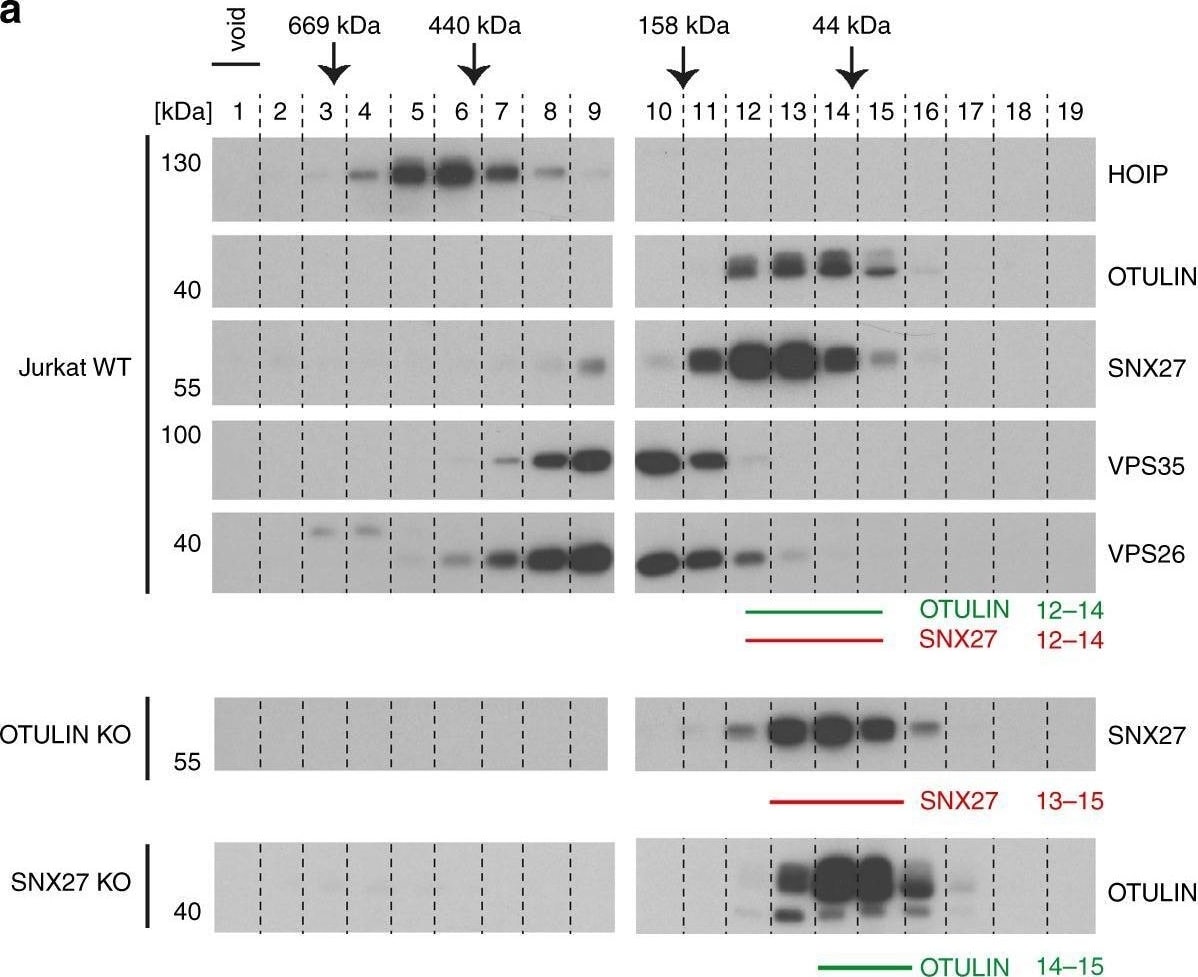
Binding of OTULIN antagonizes SNX27 association with early endosomes. a Co-elution of endogenous OTULIN and SNX27 was analyzed by size exclusion chromatography. Upper panels: cell lysates of parental Jurkat T cells were fractionated using a Superdex 200 column and elution profiles of endogenous proteins (OTULIN, SNX27, HOIP, VPS35, and VPS26) were determined by WB. Lower panels: determination of elution profiles of endogenous SNX27 and OTULIN in OTULIN and SNX27 KO Jurkat T cells, respectively. Peak elution of molecular weight standards is depicted at the top. b HEK293 cells virally transduced with GFP-SNX27 (green) were stained for early endosomes using anti-EEA1 antibody (red) and co-localization was analyzed by confocal microscopy. c HEK293 cells were co-transduced with GFP-SNX27 (green) and RFP-OTULIN WT, delta ETSL or C129A (red). Localization of proteins was analyzed by confocal fluorescence microscopy. d HEK293 cells virally transduced with RFP-OTULIN WT, delta ETSL or C129A (gray) were stained for endogenous SNX27 (green) and EEA1 (red) and localization was analyzed by confocal fluorescence microscopy. e WT or OTULIN KO HEK293 cells were stained for endogenous SNX27 (green) and EEA1 (red) and localization was analyzed by confocal fluorescence microscopy. Co-localization in d and e was quantified by determining Pearson’s correlation using at least 12 pictures and imaging more than 100 cells for each condition. Graphs depict the mean ± SD. Two-tailed p-values: ns not significant, *p ≤ 0.05, ***p ≤ 0.001 by unpaired t-test. Scale bars: 10 µm. Source data are provided as a Source Data file Image collected and cropped by CiteAb from the following open publication (https://pubmed.ncbi.nlm.nih.gov/31541095), licensed under a CC-BY license. Not internally tested by R&D Systems.Detection of HOIP/RNF31 by Western Blot
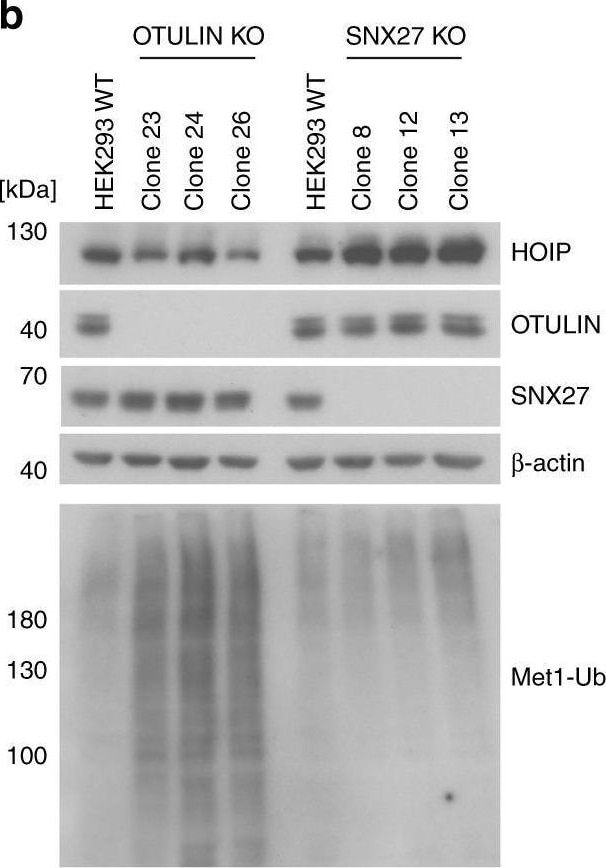
OTULIN counteracts SNX27 cargo loading. a Formation of a ternary HOIP-OTULIN-SNX27 complex was analyzed after co-expression of GFP-SNX27, HA-HOIP, and Flag-OTULIN in HEK293 cells. Ternary complex via Flag-OTULIN was verified using OTULIN Y56F (PIM mutation) and delta ETSL (PDZbm deletion) mutants. GFP-SNX27 was precipitated by GFP-Traps and association of OTULIN and HOIP was analyzed by WB. b SNX27 deficiency does not affect accumulation of Met1-ubiquitin chains. Extracts of WT, OTULIN-deficient, or SNX27-deficient HEK293 cells were analyzed for the abundance of Met1-ubiquitin chains and the expression of HOIP, OTULIN, and SNX27 by WB. c GFP or GFP-SNX27 constructs (WT or H114A) were expressed in HEK293 cells and cargo loading was analyzed by GFP-Traps by WB. d GFP-SNX27 WT was expressed in HEK293 cells alone or in the presence of HA-OTULIN WT, delta ETSL (PDZbm deletion), C129A (catalytically inactive), or C129A/L259E (catalytically inactive ubiquitin-binding mutant) and cargo loading was analyzed by GFP-Traps from cell lysates by WB. e Cargo-loading to endogenous SNX27 was assessed after anti-SNX27-IP in parental HEK293 cells as well as SNX27 KO or OTULIN KO cells. Binding of SNX27 to cargos and OTULIN was analyzed by WB. f Cargo-loading onto endogenous SNX27 was assessed by WB after anti-SNX27-IP in OTULIN KO HEK293 cells reconstituted with mock, OTULIN WT or OTULIN delta ETSL. Binding of each cargo to GFP-SNX27 (d) or SNX27 (e, f) was quantified from four independent experiments (see Supplementary Fig. 7a, b, e). A cumulative score for relative binding of all cargos to SNX27 was calculated and depicted as mean ± SD below the WB. Two-tailed p-values: ns not significant, ***p ≤ 0.001 by unpaired t-test. Source data are provided as a Source Data file Image collected and cropped by CiteAb from the following open publication (https://pubmed.ncbi.nlm.nih.gov/31541095), licensed under a CC-BY license. Not internally tested by R&D Systems.Detection of Human HOIP/RNF31 by Western Blot
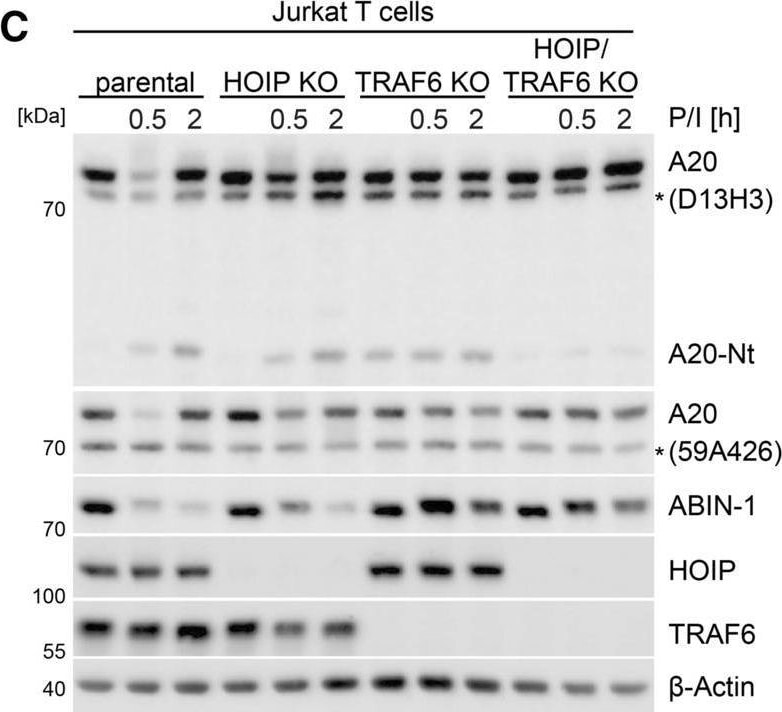
A post-translational crosstalk controls stability of A20 and ABIN-1 after stimulation in Jurkat T cells. A Stability of ABIN-1 and A20 in Jurkat T cells after P/I stimulation treated with proteasome inhibitor MG132 (25 µM) analyzed by Western blot. B, C Stability of ABIN-1 and A20 in parental, CARD11 KO TRAF6 KO, HOIP KO and TRAF6/HOIP double KO Jurkat T cells after P/I stimulation analyzed by Western blot. D, E Stability of ABIN-1 and A20 in parental, ABIN-1 KO and A20 KO Jurkat T cells after CD3/CD28 and P/I stimulation analyzed by Western blot. F ABIN-1-IP and detection of A20-bound ABIN-1 in Jurkat T cells after P/I stimulation analyzed by Western blot. G ABIN-1 ubiquitination in HEK293 cells transfected with Flag-A20 WT, ZNF4/7 mt and OTU mt together with ABIN-1 and HA-K48Ub (K48-only) after ABIN-1-IP analyzed by Western blot. H A20 ubiquitination in HEK293 cells transfected with Flag-A20 WT, ZNF4/7 mt and OTU mt after HA-IP was analyzed by Western blot. I, J ABIN-1 stability and A20 cleavage in Jurkat T cells treated with MALT1 inhibitor MLT-985 (1 µM) following CD3/CD28 or P/I stimulation analyzed by Western blot Image collected and cropped by CiteAb from the following open publication (https://pubmed.ncbi.nlm.nih.gov/35099607), licensed under a CC-BY license. Not internally tested by R&D Systems.Applications for Human HOIP/RNF31 Antibody
Application
Recommended Usage
Immunocytochemistry
8-25 µg/mL
Sample: Immersion fixed MCF‑7 human breast cancer cell line treated with Proteasome Inhibitor I (Tocris Bioscience, Catalog # 4045)
Sample: Immersion fixed MCF‑7 human breast cancer cell line treated with Proteasome Inhibitor I (Tocris Bioscience, Catalog # 4045)
Simple Western
5 µg/mL
Sample: Jurkat human acute T cell leukemia cell line and MCF‑7 human breast cancer cell line
Sample: Jurkat human acute T cell leukemia cell line and MCF‑7 human breast cancer cell line
Western Blot
0.5 µg/mL
Sample: Jurkat human acute T cell leukemia cell line and MCF‑7 human breast cancer cell line
Sample: Jurkat human acute T cell leukemia cell line and MCF‑7 human breast cancer cell line
Formulation, Preparation, and Storage
Purification
Protein A or G purified from hybridoma culture supernatant
Reconstitution
Reconstitute at 0.5 mg/mL in sterile PBS. For liquid material, refer to CoA for concentration.
Loading...
Formulation
Lyophilized from a 0.2 μm filtered solution in PBS with Trehalose. *Small pack size (SP) is supplied either lyophilized or as a 0.2 µm filtered solution in PBS.
Shipping
Lyophilized product is shipped at ambient temperature. Liquid small pack size (-SP) is shipped with polar packs. Upon receipt, store immediately at the temperature recommended below.
Stability & Storage
Use a manual defrost freezer and avoid repeated freeze-thaw cycles.
- 12 months from date of receipt, -20 to -70 °C as supplied.
- 1 month, 2 to 8 °C under sterile conditions after reconstitution.
- 6 months, -20 to -70 °C under sterile conditions after reconstitution.
Calculators
Background: HOIP/RNF31
Long Name
HOIL-1-Interacting Protein
Alternate Names
RNF31, ZIBRA
Gene Symbol
RNF31
UniProt
Additional HOIP/RNF31 Products
Product Documents for Human HOIP/RNF31 Antibody
Certificate of Analysis
To download a Certificate of Analysis, please enter a lot or batch number in the search box below.
Note: Certificate of Analysis not available for kit components.
Product Specific Notices for Human HOIP/RNF31 Antibody
For research use only
Related Research Areas
Citations for Human HOIP/RNF31 Antibody
Customer Reviews for Human HOIP/RNF31 Antibody
There are currently no reviews for this product. Be the first to review Human HOIP/RNF31 Antibody and earn rewards!
Have you used Human HOIP/RNF31 Antibody?
Submit a review and receive an Amazon gift card!
$25/€18/£15/$25CAN/¥2500 Yen for a review with an image
$10/€7/£6/$10CAN/¥1110 Yen for a review without an image
Submit a review
Protocols
Find general support by application which include: protocols, troubleshooting, illustrated assays, videos and webinars.
- Appropriate Fixation of IHC/ICC Samples
- Cellular Response to Hypoxia Protocols
- ClariTSA™ Fluorophore Kits
- Detection & Visualization of Antibody Binding
- ICC Cell Smear Protocol for Suspension Cells
- ICC Immunocytochemistry Protocol Videos
- ICC for Adherent Cells
- Immunocytochemistry (ICC) Protocol
- Immunocytochemistry Troubleshooting
- Immunofluorescence of Organoids Embedded in Cultrex Basement Membrane Extract
- Immunohistochemistry (IHC) and Immunocytochemistry (ICC) Protocols
- Preparing Samples for IHC/ICC Experiments
- Preventing Non-Specific Staining (Non-Specific Binding)
- Primary Antibody Selection & Optimization
- Protocol for VisUCyte™ HRP Polymer Detection Reagent
- Protocol for the Fluorescent ICC Staining of Cell Smears - Graphic
- Protocol for the Fluorescent ICC Staining of Cultured Cells on Coverslips - Graphic
- Protocol for the Preparation and Fluorescent ICC Staining of Cells on Coverslips
- Protocol for the Preparation and Fluorescent ICC Staining of Non-adherent Cells
- Protocol for the Preparation and Fluorescent ICC Staining of Stem Cells on Coverslips
- Protocol for the Preparation of a Cell Smear for Non-adherent Cell ICC - Graphic
- R&D Systems Quality Control Western Blot Protocol
- TUNEL and Active Caspase-3 Detection by IHC/ICC Protocol
- The Importance of IHC/ICC Controls
- Troubleshooting Guide: Western Blot Figures
- Western Blot Conditions
- Western Blot Protocol
- Western Blot Protocol for Cell Lysates
- Western Blot Troubleshooting
- Western Blot Troubleshooting Guide
- View all Protocols, Troubleshooting, Illustrated assays and Webinars
Loading...
Associated Pathways