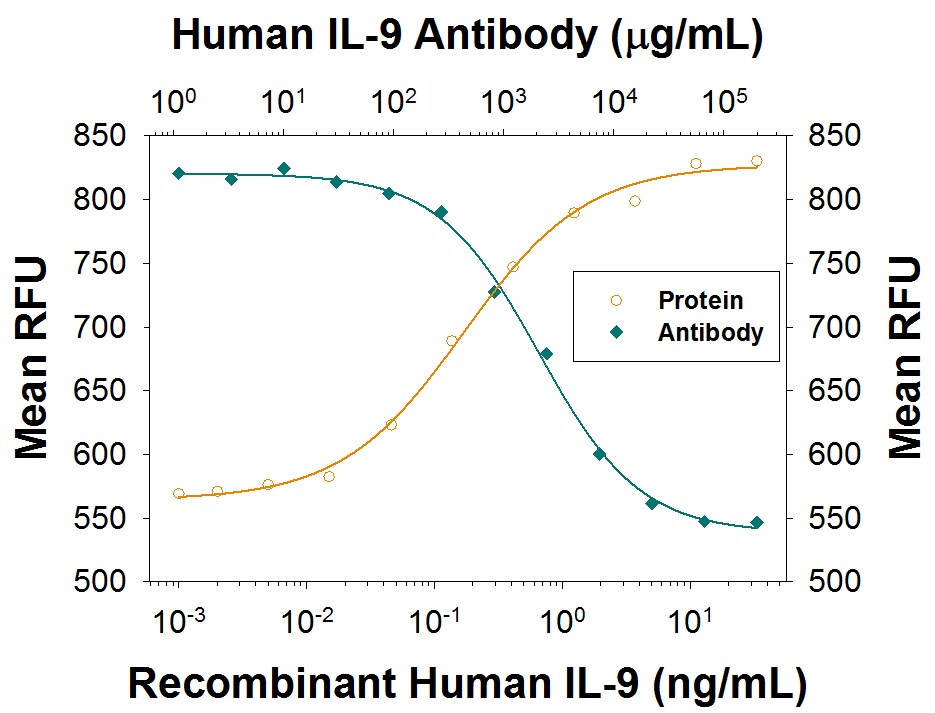
Human IL-9 was originally identified as a cytokine found in the conditioned medium of a human T cell leukemia virus type I (HTLV-I) transformed T cell line that is mitogenic for the factor-dependent human megakaryoblastic leukemic cell line, M07e. The cDNA encoding this cytokine was subsequently isolated by functional expression cloning and found to be similar to the mouse T cell growth factor III/P40. This human cytokine and its murine homologue are now designated as human and mouse IL-9. Besides HTLV-I or -II transformed T cell lines, rhIL-9 is also produced by activated human PBLs. Human IL-9 was also reported to be expressed by primary and cultured Hodgkin and Reed-Sternberg (H-RS) cells derived from Hodgkin’s disease patients, suggesting a possible role for rhIL-9 in the development of the pathophysiology of Hodgkin’s disease.
Human and murine IL-9 are also capable of enhancing in vitro survival of human T cell lines as well as synergizing with Epo to support erythroid colony formation in vitro. However, the mast cell enhancing activity associated with rmIL-9 has not yet been demonstrated in the human system and no human IL-9-dependent T cell clones have been identified.
The gene for rhIL-9 has been mapped to human chromosome 5. As in the mouse system, the human IL-9 cDNA encodes a 144 amino acid residue precursor protein with an 18 amino acid signal peptide that is cleaved to form the mature cysteine-rich protein with a predicted molecular mass of 14 kDa. Human IL-9 contains four potential N-linked glycosylation sites and the native rhIL-9 is a highly glycosylated protein. Human and mouse IL-9 share 56% and 67% homology at the amino acid and nucleotide levels, respectively. Although murine IL-9 is active on human cells, human IL-9 is not active on mouse cells.